Researchers with the Universiti Tun Hussein Onn Malaysia (UTHM) recently published a paper about the challenges faced when using 3D printing to manufacture biomedical products in Malaysia. Additive manufacturing has not been widely accepted by local industries in the country, which has led to “a call for government and local practitioners to promulgate the development of this technology for various industries, particularly for biomedical products.”
“The current study intends to frame the challenges endured by biomedical industries who use 3D printing technology for their manufacturing processes. Qualitative methods, particularly in-depth interviews, were used to identify the challenges faced by manufacturing firms when producing 3D printed biomedical products,” the team wrote.
Many studies about 3D printing are centered on developments in the US and Europe, and not developing countries such as Malaysia. Biomedical products complicate things even further, since they require clinical testing and biocompatible materials. So the researchers discussed “the practices involved in manufacturing printed 3D biomaterial products” in their study, created a framework regarding biomedical product development, and interviewed three local companies in order to attain real-life perspectives from those involved in 3D printing biomedical products in Malaysia.
Quoting ASTM standard F2792, the team said there are seven classification groups for 3D printing – binder jetting, directed energy deposition, material extrusion, material jetting, powder bed fusion, sheet lamination, and vat photopolymerization. While studies have identified over 350 types of industrial 3D printers, systems for making biomedical products are mostly extrusion and inkjet-based. They also explained that 3D printing can make functional parts with over 450 materials, including thermoplastic filaments, hydrogels, aluminum alloys, carbon fiber-reinforced polymer composites, and more.
“An ideal 3D printed biomaterial should morphologically mimic living tissue, be biocompatible, and be easily printable with tuneable degradation rates,” they noted.
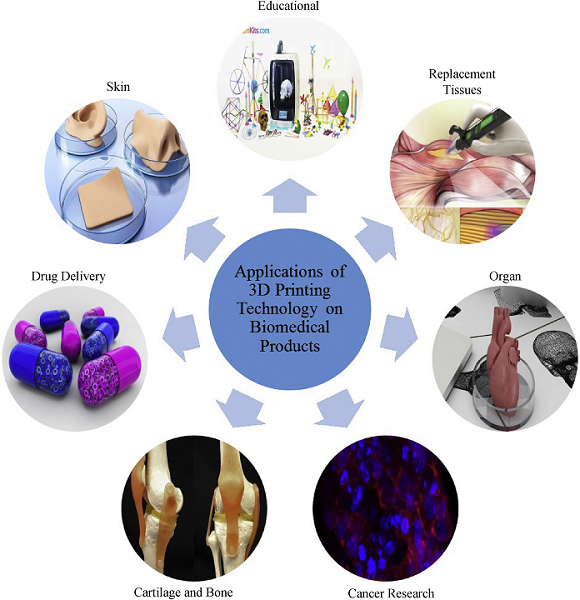
The paper lists several applications for making 3D printed biomedical products, such as:
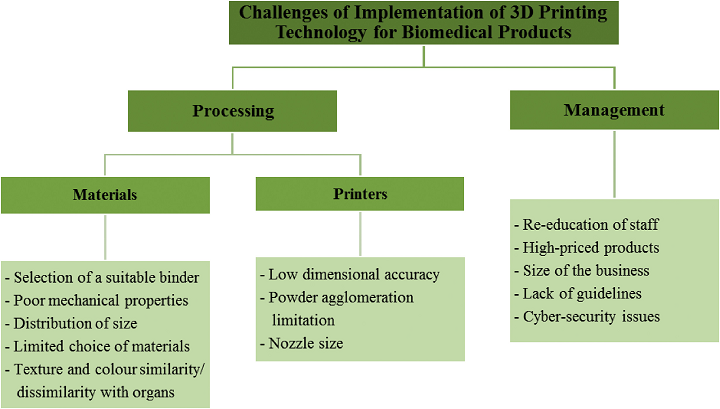
The team created “a conceptual framework” related to twelve important challenges that arise when 3D printing biomedical products, such as poor mechanical properties, material lifespan, nozzle size, binder selection, layer height, high powder agglomeration levels, and build failure. For example, choosing the proper binder fitting to 3D print bone tissue is tough, as not all binder materials can be used in sintering. Organic ones are the best for making 3D printed parts, and photopolymers are the only good choice when making bone tissue with SLA technology.
“Furthermore, the particle size of the powder influences the thickness of the printed layer [73]. Distribution of sizes and shapes of the powder also affect the quality of scaffolds,” they wrote. “Lack of pore interconnectivity affects the mechanical properties of 3D scaffolds. The powder must be biocompatible and biodegradable as scaffolds need to promote tissue regeneration after implantation [74].”
Another difficulty, outlined by Yap et al. [77], is that the color and texture of products need to be similar to the 3D printed organ when it comes to ophthalmic models.
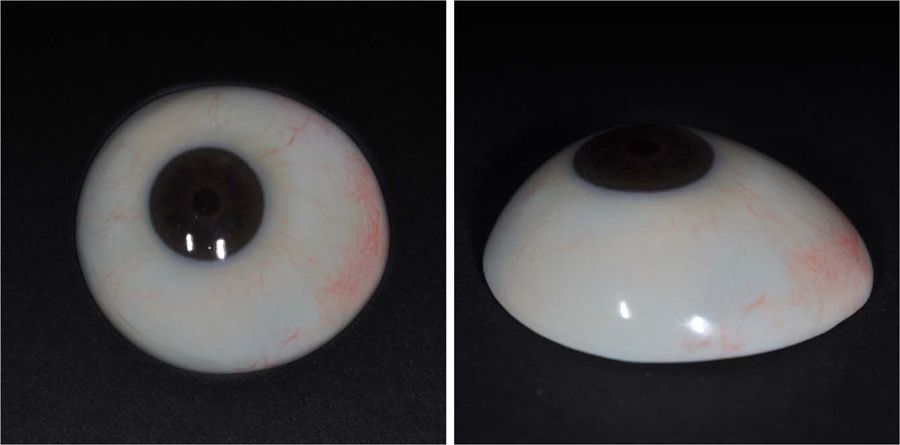
Customized ocular prosthesis fabricated using 3D printing and surface printing. (Image: Severance Hospital South Korea)
“Meanwhile, according to Chang [78], a challenge faced when producing 3D printed biomedical products is the similarity in colour to the printed product,” the team explained.
When it comes to the 3D printer itself, one needs to maintain accurate dimensions – especially with thickness – when using the technology for tissue engineering applications. Accuracy is dependent on design, since 3D printing isn’t great when it comes to unsupported long, thin features. Additionally, Scott [80] said dimensional accuracy is not easy with FDM printing, but that higher accuracy and resolution can be achieved through PolyJet and inkjet systems.
“According to Husain et al. [81], one of the challenges of current 3D printing technology for producing biomedical products is the difficulty in achieving a nanoscale resolution for clinically relevant biomedical products,” they noted. “Advanced 3D bioprinting techniques were developed to fabricate the next generation of complex biocompatible and biomimetic tissue constructs, such as vascular grafts, dermal dressing, osteochondral tissues, and neural tissues. This statement was also supported by Vasireddi & Basu [73], who said that the challenge of producing 3D scaffolds is the limited resolution of a 3D printer caused by the size of the nozzle [73].”
But materials science and powder aren’t the only issues the UTHM researchers discuss.
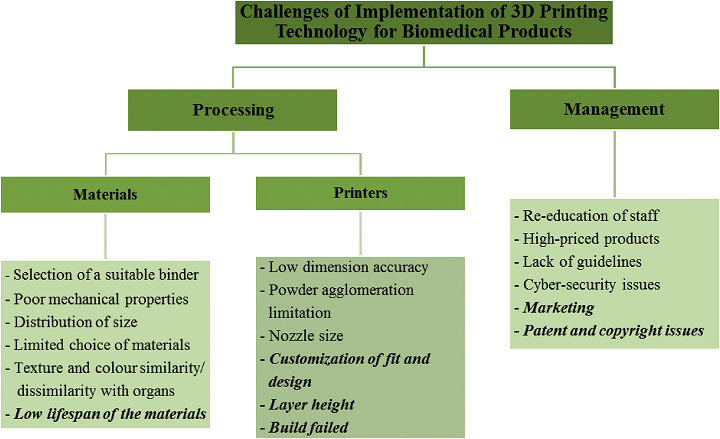
“Furthermore, there also are six challenges in the management of manufacturing biomedical products using 3D printing technology, and these include staff re-education, product pricing, limited guidelines, cyber-security issues, marketing, and patents and copyright,” the researchers stated.
For example, multiple cost factors affect the application of 3D printing to biomedical products, including utility, technological maintenance, and materials, as well as “hardware, software, and system integration [83].” Total adoption of 3D printing in the hearing aids industry required teaching the staff how to use the new printers and software, in addition to skilled technicians to work on the sculpting, molding, and curing.
“Meanwhile, Gao et al. [85], found that the challenge of producing a 3D product is the lack of guidelines for a fundamental design. The materials and machines used vary according to the type of biomedical products that need to be produced. Therefore, a designer is required to carry out a trial and error process to obtain the desired products,” the team wrote. “This claim was also supported by Pavlovich et al. [86], who stated that the coordinated standards and regulatory pathways for biomedical products are still lacking.”
Finally, as 3D printing typically requires a connection to the internet for helpful features like remote diagnosis troubleshooting, this also opens up opportunities for cyber attacks that can remotely compromise the systems. Because these attackers can alter STL files, CAD models, and even the actual 3D printer, “the confidentiality, integrity, availability of data and even fabricated physical components in these systems are at risk.”
The researchers interviewed three people, representing top management at Companies X, Y, and Z, in order to “investigate the challenges of utilizing 3D printing on biomedical products in Malaysia.”
“The qualitative case study method was chosen in this study, as it enables a strong description to address the research questions [89],” they explained.
Respondent 1 (R1) is from Company X, which was founded in 1990 to develop new use cases for 3D printing with high potential, and is now “the largest group of software developers in the industry and is one of the largest facilities involved in 3D printing technology in the world.” Respondent 2 (R2) is with Company Y, which got its start in the 2000s and has gained plenty of experience with 3D bioprinters, creating customized scientific setups, and 3D printing living tissue. Respondent 3 (R3) comes from Company Z, which is Malaysia’s top CAD, CAM, and CAE solutions provider and offers training, certification courses, and consultancy and engineering services as well.
After the interviews, the team identified some of the top challenges, such as selecting good binders. R1 said that a difficult challenge for designers and engineers is choosing “a suitable binder for the 3D printing process because each product or design has its own unique binder.” R2 agreed, stating that companies need to “select suitable binders because not all types of binders are biocompatible.”
“Binder selection is based on the targeted application and capabilities of the printer and printer head. For biodegradable parts, the binder must also be biodegradable, non-toxic, easy to handle, and readily available,” the team explained. “In the context of renewable materials, the binder should also be based on natural or renewable resources. Hence, due to these requirements, common binders for 3D-printing are not acceptable [90].”
Another challenge is mechanical strength.
“For specific 3D printed materials, the mechanical performance of the final print is very important,” R3 said. “There are challenges to produce 3D printed biomedical products with good mechanical strength and suitable to the human body. The final printed part mainly depends on the inter-diffusion and re-entanglement between the deposition rasters of the fused polymer.”
Powder size distribution and shape is another material challenge for 3D printing biomedical products, because, as R3 pointed out, this can “affect the optical and thermal properties of particles.”
“Lack of pore interconnectivity network caused by the lower bounds of porosity affects the mechanical properties of 3D printed biomedical products.”
In fact, finding any material with good properties for the human body is a challenge, as they need to be similar to, and suitable for, tissues and organs.
“So, actually we have to examine numerous properties about the material. First, it must be biocompatible to make sure this material can be used in the patient’s body,” R1 said.
“Sometimes the challenge is to choose (the) right material properties. Because sometimes the customer says he wants (a) flexible material (that) does not break. However, this is difficult for us to (achieve). We have to make it clear, how to make it flexible, but not broken or torn. So, (to obtain the) ideal material properties according to customer requirements in materials selection is the challenge for us.”
Sometimes, customers request biomedical products that are identical to the real human organ, for surgical training or planning purposes; multi-color 3D printing makes this possible, though the texture can be harder to match. But a material’s lifespan is another challenge to consider, as its properties could be negatively affected if used after its expiration date, thus harming the patient.
“All the material(s) have (expiry) date(s). The lifetime for the resin is very short. Therefore, the expired material is very (challenging) for us,” R3 said. “When we purchase a syringe of composite, three important aspects are the storage condition, batch number and the expiration date. Most of the direct materials have a limited shelf life.”
Moving on to challenges with 3D printers, R1 noted that “the accuracy of 3D printed biomedical products depends on the design,” and R2 agreed, noting that materials were also important for ensuring accuracy:
“Accuracy also depends on materials. For instance, standard SLA resin will produce more dimensionally accurate parts than flexible resin. Standard materials are recommended for parts where high accuracy is critical.”
In order to achieve the proper level of accuracy, materials and machines need to be chosen carefully. This leads us to another challenge – the size and diameter of the 3D printer nozzle, which can affect the width of each line in 3D printed biomedical products.
“If you use (a) 3D printer for doing large quantities of 3D printed biomedical products, you will want to make sure your extruder is laying down the right amount,” stated R3. “Depending on the 3D printer, several nozzles can be interchanged reasonably easy.”

Loading a custom-made 3D printer, which gives more control over tissues created by the FIBERS project. (Image: North Carolina State University and the University of North Carolina at Chapel Hill)
When choosing the size of the nozzle, users must balance print speed and how much filament is extruded to make the right decision.
Repeatable protocols and technologies are required to bioprint thick tissues, along with customizing the design and fit and choosing the right analytical and computational modeling techniques for achieving optimal microarchitecture for biomedical products. Additionally, another challenge is layer height.
“Printing parameters like layer height play a crucial role in fabricating biocompatible scaffolds with required mechanical strength and pore size,” R2 stated. “Layer height is ordinary. The faster it prints, the less the quality of the product. If we want to produce a delicate model and want to be 30 millimetres (mm), then we have to set it up for slow production. Because, a higher layer means lower quality.”
Build failure was the last 3D printer challenge discussed for biomedical product applications. The most common causes are models that are too thin, not adding supports for overhangs, materials not horizontal on the build plate, power loss, extrusion errors, or filament jams.
“The best way to prevent over extrusion is to ensure that the layer height is less than the nozzle diameter and the speed of the cooling fan is increased. Additionally, to avoid this issue, the engineers should check the nozzle for clogs and increase the hot-end temperature [104],” the researchers noted.
Finally, the three respondents and the researchers moved on to challenges in management for 3D printed biomedical products. It’s helpful to offer training programs for employees who are new to the technology.
“We have training in Belgium for new workers, so new employees can get the new information about 3D printing technology. Even here, we have dedicated trainers. The trainers are (workers) who (have) been working a long time. So, those trainers will be mentors for (newbies). Therefore, usually we will set the programme or training for them and do it internally,” R1 said. “If it is about business or sales, we will have access to another company to come here to do some training or talk. Usually because of many years of experiences (in) 3D printing, we have internal trainers that can give training or (talks).”
Sometimes, it’s even necessary to re-educate staff members when adopting 3D printing, as they will need special skills so companies can follow product specifications and industry regulations.

Skills training is part of the approach Workshops for Warriors takes to fill current and future manufacturing roles. (Image: Stratasys)
Another challenge is the expensive materials and machines that are necessary to 3D print biomedical products; a low price tag equals less functionality, smaller build size, and lower print quality.
“To start the project, (USD)116 thousand to 1 million is required. But now, the bio-printer that we bring is affordable, below (USD)232 thousand,” R2 said. “So, we have a goal (that) in Malaysia all universities (should) have a bio-printer.”
Management also needs to consider the challenge of complicated standards and procedures, since medical products need to be safe to use in the human body. Another major challenge is cybersecurity.
R3 said, “When we run the production using 3D printing technology, a malicious input could come from an integrated connection layer in the form of a malicious real-time controlling command that can change the production design.”
Marketing is an emerging challenge, especially in Malaysia, where 3D printing is still in its infancy in terms of adoption, especially for biomedical applications.
“When we joined some events and conferences, some people were clueless about 3D printing because they (have) never heard of the technology,” R2 said. “And it is possible that most people still (do) not know about the existence of 3D printing technology for manufacturing biomedical products in Malaysia.”
The researchers also learned from the three respondents that another new challenge for 3D printing biomedical products is patent and copyright issues, which protect the original inventions and ideas.
“Usually, if I make (a) software and then you also see the process that I used to make (the) software and you copy it, it is really hard to prove that (it) is my creation and (another person copied) my invention,” R1 explained. “(It is so), especially for software, because in (the) whole process of 3D printing, the software is very important for us. This is because we will start using CT or MRI, then convert the data into a 3D model and use the software for creating new things. So, software is our focus for IPA. So, your question on how important the software is, well the answer is Yes. It is very important to us.”
R2 said that Malaysia is “approximately three to five years behind in utilizing 3D printing technology,” especially since many innovations have been patented in other countries. All three respondents agreed that patent and copyright issues are definitely a challenge, and suggested that incentives should be offered, or a subsidiary created, by the Malaysian government to help companies with this.
“This study could be a guideline for new manufacturers, human resources and the management sector. For new companies intending to adopt this technology, the qualitative sharing experience from this study will provide an early insight into what the company will encounter,” the researchers concluded. “It is anticipated that the findings of this study will assist Malaysians to obtain concise information about the utilization of 3D printing technology in the manufacturing industry.”
Discuss this and other 3D printing topics at 3DPrintBoard.com or share your thoughts below.
Subscribe to Our Email Newsletter
Stay up-to-date on all the latest news from the 3D printing industry and receive information and offers from third party vendors.
You May Also Like
3D Printing News Briefs, April 13, 2024: Robotics, Orthotics, & Hypersonics
In 3D Printing News Briefs today, we’re focusing first on robotics, as Carnegie Mellon University’s new Robotics Innovation Center will house several community outreach programs, and Ugogo3D is now working...
Rail Giant Alstom Saves $15M with 3D Printing Automation Software 3D Spark
3D Spark has entered into a three-year deal with the rail giant Alstom. Alstom, a transport behemoth with annual revenues of $16 billion, specializes in the manufacture of trains, trams,...
Meltio Expands Global Reach with New Partnerships in the Americas and Europe
Spanish 3D printing manufacturer Meltio has expanded its sales network across the globe. With the addition of three new partners in the United States, Brazil, Argentina, and Italy, Meltio aims...
3D Printing Webinar and Event Roundup: April 7, 2024
Webinars and events in the 3D printing industry are picking back up this week! Sea-Air-Space is coming to Maryland, and SAE International is sponsoring a 3D Systems webinar about 3D...