Wake Forest: In Situ 3D Printing of Tailored Skin Grafts, a Possible Step Forward in Tissue Engineering
In recent years bioprinting has proved to be an important tool for tissue engineering applications and it holds great promise towards the fabrication of skin tissue. A team from Wake Forest Institute for Regenerative Medicine (WFIRM) has brought progress in this direction by developing a proof of concept validation of a mobile skin bioprinting system that provides rapid on-site management of full-thickness wounds. The prototype is intended to be used for wound healing and the coverage of large wound areas directly on the patient. The experiments were conducted on mice and pigskin, so this experiment is years away from being trialed in humans. For now, it is great news if you’re trying to repair a ham. Nonetheless, this study represents a significant advance in potential personalized wound treatment approaches as it is similar and goes further than other approaches in bioprinting skin directly onto patients.
It also represents a very strategic Intellectual Property move by Wake Forest to claim, potentially patent and exploit a logical path to large scale wound treatment. Bioprinting gradients, correct vascularization, actually implementing patient specific bioprinting, the combination of scanning and printing, and in situ printing of skin are all pivotal areas. This is especially relevant since Wake Forest has in the past been quick to press release and slow to publish when compared to others working in the field.

A member of WFIRM team operates with the bioprinter
This is salient since this idea could represent a substantial improvement in health care. Large, nonhealing or chronic wounds as the ones derived from burn injuries, diabetes or venous and pressure ulcers represent a burden for patients and mean large costs on health services. Only in the United States, over 7 million patients suffer each year from this issue, representing an expenditure of $25 billion each year. There is no need to explain then the importance of the improvement in regenerative medicine for our society.
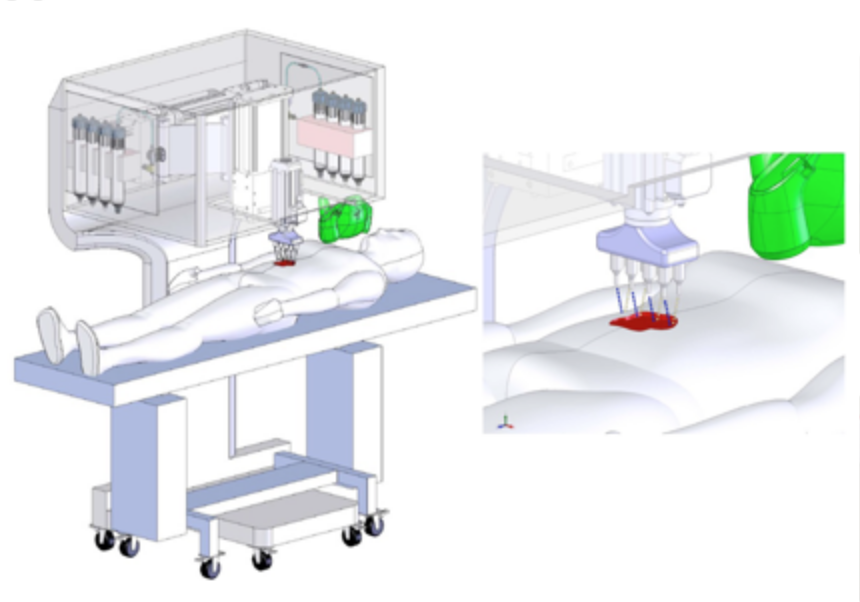
The bioprinting system developed by the scientist at WFIRM offers a step forward to the improvement of this issue. It is a bedside machine that can tailor skin tissue directly on the patient.
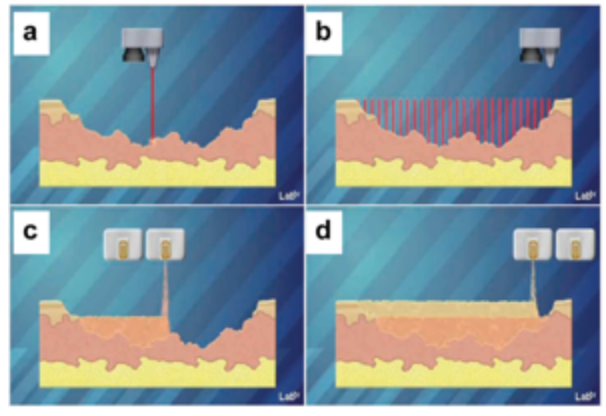
The main components of the skin bioprinter system consist of a hand-held 3D scanner and a printing head with an XYZ movement system containing eight 260 µm diameter nozzles, each driven by an independent dispensing motor. The ZScanner Z700 scanner offers the ability to capture the entire wound in a continuous scan and it creates a wound map for printing.
Directly from the abstract:
“Once the scan is complete, the wound data is compiled to form a model of the wound. The scanned wound area is then processed using Geomagic Studio software orient the model to standard coordinates so the import into Artcam is ideal. Scanned data is in the form of an STL file that has been outputted from Geomagic and inputted to Artcam 3D software to obtain the full volume and the nozzle path needed to print the fill volume.”
Once the data is taken, it can be modified to better fit the structure of the wound to treat:
“The user can define a series of bitmap images where each non-black pixel corresponds to a cell drop, and the color of the pixel corresponds to the cell type to print. This method allows the user to create complex structures using any cell type in any configuration. Finally, a list of available cell types loaded in the printer is provided to the user by the print-head configuration.”
Once ready, it is used to generate the fill volume and the path points for nozzle head to print. The delivery system is similar to the ones used in traditional inkjet printing. It has several cartridges, each contains different biomaterials and every single nozzle is connected to a separate cartridge. It 3D prints directly on the wound a double layered skin substitute consisting dermal fibroblasts and epidermal keratinocytes cells that exactly match the patient’s wound.
“The wound depth is then split into layers on the Z axis to determine which layers correspond to the dermis and epidermis. Each layer is overlaid with a series of XY lines that cover the entire wound area. These lines are used in conjunction with the cartridge-based delivery system to determine a path for the printer. The skin cell delivery system is controlled by a custom software employing a three-tiered architecture design based on the Microsoft.NET Framework 2.0 and written in C++.”
Skin bioprinting concept via WFIRM
What makes this technology special is that while being mobile, the skin bioprinter has the ability of 3D print customize skin tissue directly on a patient by scanning and measuring the patient’s injury. Through the compilation and manipulation of data, it makes possible to manage extensive wound surface and deposit with precision customize biomaterial that simulates skin.
In contrast with the existing technologies used to heal wounds such as using a manually seeded matrix or cell spraying, this has possible advantages. The bioprinted skin may prove to be the most adequate due to its ability to place specific cell types into needed position and creating a complex skin structure that improves wound healing with better aesthetic properties. From an intellectual property standpoint, Wake Forest has really made a very timely disclosure towards what is the most straightforward path to personalized in situ 3D printing of skin tissue.
At the same time, this technique has shown a positive outcome when applied on mice and pigskin wounds. The future of its success lies in the ability of this approach in conducting a successful clinical trial in humans with prosperous results, which we all hope for.
Subscribe to Our Email Newsletter
Stay up-to-date on all the latest news from the 3D printing industry and receive information and offers from third party vendors.
Print Services
Upload your 3D Models and get them printed quickly and efficiently.
You May Also Like
Flashforge Bets on Meshy AI as Desktop 3D Printing Battle Intensifies
Competition in desktop 3D printing is brutal. Whereas before, firms competed through value engineering, Prusa clones now have an integrated hardware, sensor, and software setup that is making all the...
Ford Uses Binder Jet 3D Printing to Make Boat Propellers for Sharrow Marine
Ford’s Advanced Industrial Technology and Platforms (ATP) group has helped Sharrow Marine make a boat propeller in two weeks rather than 130 days. Thanks to the Michigan Central program, Ford...
Skuld to Work on DARPA’s Rubble to Rockets (R2R) Program
Skuld will work on the Defense Advanced Research Projects Agency’s (DARPA) Rubble to Rockets (R2R) Program, which turns scrap metal into missile components. Skuld will help with alloy design, characterization, and...
From “Magic” to Metal: How Intrepid Automation Wants to Make 3D Printing Matter at Scale
Ben Wynne still talks about 3D printing the way people do when they’ve felt that “wow” moment up close. Back in the early 2000s, he was working at HP’s advanced...