New Research Paper Takes In-Depth Look at Challenges and Benefits of 3D Printed Pharmaceuticals
According to a new research paper, “In spite of all regulatory hurdles associated with 3D printing medicine, the FDA approved the first 3D printed pill, Spritam® (levetiracetam) in August 2015. In this case the product is considered as approving new mass production for equivalent product.”
The notion of 3D printed pharmaceuticals caught the attention of Preethy Ani Jose, with the Oxford College of Pharmacy, and Peter Christopher GV, from pharmaceutical company Strides Shasun Limited in Bangalore, and the two worked together to write the paper, titled “3D Printing of Pharmaceuticals – A Potential Technology in Developing Personalized Medicine,” which was recently published in the Asian Journal of Pharmaceutical Research and Development.
The abstract reads, “The 3D PRINTING technology has caught the attention of medical devices industry and pharmaceutical industry due to its applications on various platform in health care industry. Even though this technology exists for a long time it is of public interest highly now due to the approval of 3-D printed tablet and other medical devices and also with the advent of USFDA’s guidance on technical considerations specific to devices using additive manufacturing which encompasses 3-dimensional (3D) printing has triggered many thoughts about this technology which needs to be considered for successful delivery of intended product. This paper presents regulatory agencies expectations, limitations, problems in establishing such setups for production of drug products, advantages, disadvantages, applications, methods and associated risks involved in manufacturing. It also provides the comprehensive review of the current status of research and development on this platform.”
The efficient transportation of a pharmacologically active compound in the body for the purposes of safe therapeutics is called drug delivery. By controlling the release profile of pharmaceutical products, their safety and efficiency can be improved, but there is a slightly increased risk of unwanted side effects when it comes to customized medicine and dosing; however, this probability is higher in the geriatric and pediatric populations, as bulk pill manufacturing is more concentrated on the average population.
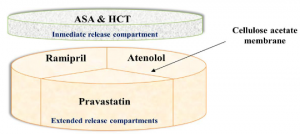
Because of this, many researchers have turned to 3D printing to create new, custom methods of drug delivery, as it can hold dosage formulations of either single blend or multi layer tablets that have sustained release properties, thus lowering how many pills a patient has to take each day, and often they must be consumed.
“3D printing technology has high potential in individualized dosage form concept called the polypill concept,” the introduction to the paper reads. “This brings about the possibility of all the drugs required for the therapy into a single dosage form unit.”
However, there are challenges to getting 3D printed pharmaceuticals on the market, including proper testing of Design and Manufacturing Process Considerations, validating the 3D printing process and software to determine the level of accuracy, and documentation in order to confirm that any products conform to existing guidelines.
The paper reads, “Important issues concerning 3D printed medicines like tort liability and intellectual rights need to be addressed to protect manufacturers and end users.”
The paper also discussed the various 3D printing methods that are being used to fabricate pharmaceuticals. If scientists can move past the challenges, and figure out how to use 3D printing to their advantage, then hospitals, pharmacies, and patients around the world can benefit from custom, personalized medication.
Discuss this and other 3D printing topics at 3DPrintBoard.com or share your thoughts below.
Subscribe to Our Email Newsletter
Stay up-to-date on all the latest news from the 3D printing industry and receive information and offers from third party vendors.
Print Services
Upload your 3D Models and get them printed quickly and efficiently.
You May Also Like
3D Printing News Briefs, April 30, 2026: Support-Free Titanium, Drug Delivery, & More
In today’s 3D Printing News Briefs, we’ll start with Makelab’s new website, and move on to commercialization of support-free metal 3D printing in South Korea. We’ll end with drug delivery...
Post-Printing at RAPID 2026: PostProcess Technologies & Mass Finishing
The whole 3D printing workflow was on display in Boston at RAPID+TCT last week, from design software and 3D printing hardware all the way to post-processing and finishing solutions. For...
RAPID 2026 in Pictures: The Coolest & Craziest Things on the Show Floor
Last week, North America’s premier 3D printing trade show, RAPID+TCT 2026, came to Boston. I spent two days trekking the show floor, trying to see as much as I could,...
RAPID Roundup 2026: New Machines and Market Moves
RAPID + TCT 2026 wrapped up yesterday, but the show floor proved there’s a lot happening across the additive manufacturing (AM) space, especially when it comes to new hardware and...