Imagine your own heart for a second. Then try to figure out how many cells it has. It’s not that simple. Some cells are very easy to spot while others are tightly attached to one another. Each cell is like a world of its own, and for centuries scientists have tried to count them and imitate the way they function in the lab. A few years back, a group of researchers even attempted to accurately pin down the total number of cells in the human body (it was over 37 trillion). But what can cells tell us about bioprinting organs? Actually, a lot. If the high amount of cell count in our bodies is so crucial to our survival, then cell density (the number of living cells per unit volume) is definitely important for scientists working to engineer tissues and biofabricate organs. In fact, the lack of cellular density is one of the main reasons many 3D printed human tissues are not yet safe for use in organ repair and replacement.
According to Mark Skylar-Scott and Sebastien Uzel, researchers from Harvard’s Wyss Institute for Biologically Inspired Engineering and John A. Paulson School of Engineering and Applied Sciences (SEAS), the ability to construct whole organs for therapeutic use remains a daunting challenge, requiring billions of cells to be rapidly organized into functional microarchitected units.
Engineering solid organs is the final frontier for 3D bioprinting. Researchers all over the world are hard at work to simulate the native tissue’s structure and functions. But up to now, there have been so many hurdles to creating organs for human transplant, that scientists even saw an opportunity in outer space and sent bioprinters to the International Space Station to try to 3D print tissue for harvest in microgravity, hoping that could be a solution to eventually creating organs viable to humans.
But there is still a lot of hope right here on Earth – especially after Skylar-Scott and Uzel developed a new technique that allows 3D printing to focus on creating the vessels necessary to support a living tissue construct. The method, called SWIFT (sacrificial writing into functional tissue), could help overcome that major hurdle by 3D printing organ-specific tissues with high cell density and function.

A branching network of channels of red, gelatin-based “ink” is 3D printed into a living cardiac tissue construct composed of millions of cells (yellow) using a thin nozzle
“This is an entirely new paradigm for tissue fabrication. Rather than trying to 3D-print an entire organ’s worth of cells, SWIFT focuses on only printing the vessels necessary to support a living tissue construct that contains large quantities of organ building blocks (OBBs), which may ultimately be used therapeutically to repair and replace human organs with lab-grown versions containing patients’ own cells,” suggested Skylar-Scott, a Research Associate at the Wyss Institute.
The co-first authors of breakthrough research into 3D printed organs propose a biomanufacturing method for assembling hundreds of thousands of OBBs – composed of patient-specific-induced pluripotent stem cell-derived organoids – into living matrices with high cellular density. Then, perfusable vascular channels are introduced via embedded 3D bioprinting. The OBB matrices exhibit the desired self-healing, viscoplastic behavior required for SWIFT.
The SWIFT process begins by forming hundreds of thousands of stem-cell-derived aggregates into a dense, living matrix of OBBs that contains about 200 million cells per milliliter. Next, a vascular network through which oxygen and other nutrients can be delivered to the cells is embedded within the matrix by writing and removing a sacrificial ink.
“Forming a dense matrix from these OBBs kills two birds with one stone: not only does it achieve a high cellular density akin to that of human organs, but the matrix’s viscosity also enables printing of a pervasive network of perfusable channels within it to mimic the blood vessels that support human organs,” said Uzel, a Research Associate at the Wyss Institute and SEAS.
The Wyss Institute reports that the cellular aggregates used in the SWIFT method are mixed with a tailored extracellular matrix solution to make a living matrix that is compacted via centrifugation. At cold temperatures, the dense matrix has the consistency of mayonnaise – soft enough to manipulate without damaging the cells but thick enough to hold its shape – making it the perfect medium for sacrificial 3D printing. In this technique, a thin nozzle moves through this matrix depositing a strand of gelatin “ink” that pushes cells out of the way without damaging them.
Once the cold matrix is heated to 37 °C, it stiffens to become more solid while the gelatin ink melts and can be washed out, leaving behind a network of channels embedded within the tissue construct that can be perfused with oxygenated media to nourish the cells.
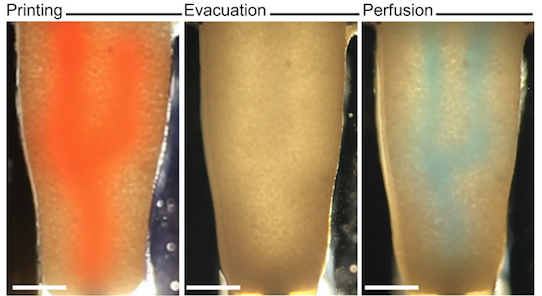
An image sequence showing the embedding, evacuation, and perfusion of branched vascular channels within a cardiac tissue matrix
“Our SWIFT biomanufacturing method is highly effective at creating organ-specific tissues at scale from OBBs ranging from aggregates of primary cells to stem-cell-derived organoids,” explained another author of the paper, Jennifer Lewis, a Core Faculty Member at the Wyss Institute. “By integrating recent advances from stem-cell researchers with the bioprinting methods developed by my lab, we believe SWIFT will greatly advance the field of organ engineering around the world.”
One of the best examples of the SWIFT biomanufacturing method is the creation of a perfusable cardiac tissue that fuses and beats synchronously over a seven-day period. The OBBs were mixed with the engineered extracellular matrix and human neonatal dermal fibroblasts to form a slurry that was subsequently compacted via centrifugation, yielding a cardiac tissue matrix. Once compacted, the cardiac matrix had an estimated cell density of 240 million cells per ml. Initially, the cardiac OBB construct beats asynchronously; however, after seven days in culture, the tissue construct beats spontaneously and synchronously.
Organ-specific tissues that were printed with embedded vascular channels using SWIFT and perfused in this manner remained viable, while tissues grown without these channels experienced cell death in their cores within 12 hours.
Wyss Institute Founding Director Donald Ingber claims that “the ability to support living human tissues with vascular channels is a huge step toward the goal of creating functional human organs outside of the body,” and that the research “ultimately has the potential to dramatically improve both organ engineering and the lifespans of patients whose own organs are failing,”
We can certainly expect to hear much more from the researchers at the Wyss Institute, since they are working with faculty members at Boston University and MIT to implant these tissues into animal models and explore their host integration as part of the 3D Organ Engineering Initiative. Perhaps most importantly, the very fact that researchers are helping in the evolution of 3D printing organs to solve a worldwide problem which up to now has had quite a few issues, mainly rejection of the organ transplanted, is noteworthy.
Organ transplantation is, by most experts’ definition, one of the most complex procedures in medicine, but scientists are going way beyond some of the most known bioprinting procedures to develop something different, to understand how to really create organs.
[Images: Wyss Institute at Harvard University]Subscribe to Our Email Newsletter
Stay up-to-date on all the latest news from the 3D printing industry and receive information and offers from third party vendors.
Print Services
Upload your 3D Models and get them printed quickly and efficiently.
You May Also Like
How Decibel Landed the Brands Everyone Wants
The first thing Adam Hecht will tell you is that 3D printing already has the technology. The harder part has been finding applications people actually want. That’s the gap Decibel...
HADDY’s Large-Format Robotic 3D Printing to Power Red Cat’s Drone Boat Production
In May 2025, Joris Peels, as is his custom, wrote a prescient article about Unmanned Surface Vehicles (USVs) and Unmanned Underwater Vehicles (UUVs), i.e., drone boats. Listing a multifaceted range...
RusselSmith Brings Additive to Ghana’s Maritime Sector
Nigerian firm RusselSmith is in talks with Ghana’s Maritime Authority (GMA) to bring 3D printing to Ghana’s maritime sector. The Commonwealth Enterprise and Investment Council (CWEIC) helped make the discussion...
3D Printing News Briefs, March 26, 2026: AMUK, IP Dispute, Asbestos, & More
We’re kicking off today’s 3D Printing News Briefs with an America Makes Project Call, and then moving on to additive manufacturing in the UK. Then we’ve got some legal news...
































