The FDA’s Principal Investigator for 3D Printed Medical Devices is Looking for Industry Feedback and No One is Giving It to Him
 Medical applications for 3D printing are a rapidly growing segment of the 3D printing industry, and the technology could radically alter the way that many medical issues are evaluated and treated. Applications as diverse as inexpensive, customized 3D printed prosthetic devices, patient-specific surgical implants, surgical pre-planning and even personalized medications all have the medical community changing the way that it thinks about healthcare. 3D printing technology could potentially lead us into an age of personalized medicine by allowing doctors, surgeons and pharmacists to tailor and fabricate medical devices and drugs for specific individuals, not create mass produced products for the average patient as they are now.
Medical applications for 3D printing are a rapidly growing segment of the 3D printing industry, and the technology could radically alter the way that many medical issues are evaluated and treated. Applications as diverse as inexpensive, customized 3D printed prosthetic devices, patient-specific surgical implants, surgical pre-planning and even personalized medications all have the medical community changing the way that it thinks about healthcare. 3D printing technology could potentially lead us into an age of personalized medicine by allowing doctors, surgeons and pharmacists to tailor and fabricate medical devices and drugs for specific individuals, not create mass produced products for the average patient as they are now.
In the United States, before any 3D printed medical device can be marketed and sold it must gain approval from the Food and Drug Administration (FDA) where it needs to be evaluated for safety. Currently the process of evaluation for new 3D printed devices can be tricky and extremely time consuming for manufacturers. The primary reason is because the technology is so new that there are no specific standards in place that companies are expected to adhere to before submitting products for review. Traditionally manufactured medical devices already have these standards in place so companies have a very clear idea of how they need to be made, what tests they must pass and what materials can and cannot be used. Currently the FDA is in the process of writing these regulations for 3D printed devices, and in May they released a draft of their Technical Considerations for Additive Manufactured Devices to the public for review.
“Most of the specifications right now are on the basic materials, bio formats, descriptive terms. We’re trying to bring knowledge of FDA best practices, knowledge of our regulatory requirements to the additive manufacturing standard so we can help foster industry growth, by letting them know what they need to do, letting them have clarity in things like testing methods,” explained James Coburn, the principal investigator for the FDA’s Center for Devices and Radiological Health.
Having these guidelines in place will be a huge development for 3D printed medical devices, and will offer significant guidance to the developers and manufacturers of these products. When the draft is finalized, it will lay out a clear roadmap for companies submitting products for review and to give them the best chance possible of it being approved. It will also be a major validation for the viability of 3D printing technology in general, and will most likely lead to an uptick in the development of additively manufactured medical products and services. The FDA has already evaluated and cleared 85 3D printed medical devices and 1 3D printed prescription drug, and they are clearly motivated to clear more. In addition to the released draft laying out the potential guidelines that the FDA is considering putting in place, it also focuses on specific areas where they believe the technology will make the most impact.
“With patient-specific devices, there are a lot of areas where people have been thinking about doing this, but it’s been cost prohibitive or technologically prohibitive, and 3D printing has opened up a lot of those doors,” continued Coburn.
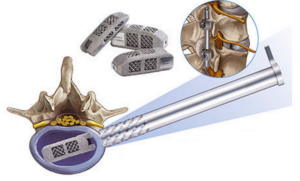
Among the devices that have already been cleared by the FDA are orthopedic implants that can be 3D printed to fit the patient’s body exactly and patient-specific surgical instruments that can be 3D printed to help doctors speed up the surgical process. These types of products were cleared using the federal agency’s current regulatory oversight over medical devices and drugs, which is not always the ideal method of evaluation. It is in both the medical community’s and the manufacturers’ best interest to have accurate and specific regulations in place both to ensure the products’ safety and to speed up the review process.
While all federal agency guidelines usually are released in draft form to the public simply for transparency’s sake, when it comes to 3D printed medical devices the need for specificity is vitally important. Because the technology is still considered emergent and evolving, the feedback and input from the industry that the FDA will be regulating is going to be invaluable to the final technical considerations. The FDA considered the draft their “initial thoughts” on the subject and have opened the document up for comment through August 8th, 2016. But according to The Hill so far they have only received 3 public comments since the draft was released in May.
I really can’t stress how important feedback from the 3D printing industry is to making sure that the FDA gets this right. Frankly, it is much easier to influence the rules before they are written than it is to change them after they have been written. The companies that manufacture 3D printed medical devices, implants and surgical tooling should be making the review of the draft a priority. After all, as Coburn notes “This only gets better with outside expertise”. You can learn more about how the FDA reviews 3D printed medical devices and follow the process of finalizing the draft of the eventual guidelines here. Discuss further in the FDA 3D Printing Regulations forum over at 3DPB.com.
Subscribe to Our Email Newsletter
Stay up-to-date on all the latest news from the 3D printing industry and receive information and offers from third party vendors.
Print Services
Upload your 3D Models and get them printed quickly and efficiently.
You May Also Like
Flashforge Bets on Meshy AI as Desktop 3D Printing Battle Intensifies
Competition in desktop 3D printing is brutal. Whereas before, firms competed through value engineering, Prusa clones now have an integrated hardware, sensor, and software setup that is making all the...
Ford Uses Binder Jet 3D Printing to Make Boat Propellers for Sharrow Marine
Ford’s Advanced Industrial Technology and Platforms (ATP) group has helped Sharrow Marine make a boat propeller in two weeks rather than 130 days. Thanks to the Michigan Central program, Ford...
Skuld to Work on DARPA’s Rubble to Rockets (R2R) Program
Skuld will work on the Defense Advanced Research Projects Agency’s (DARPA) Rubble to Rockets (R2R) Program, which turns scrap metal into missile components. Skuld will help with alloy design, characterization, and...
From “Magic” to Metal: How Intrepid Automation Wants to Make 3D Printing Matter at Scale
Ben Wynne still talks about 3D printing the way people do when they’ve felt that “wow” moment up close. Back in the early 2000s, he was working at HP’s advanced...