3D Technology Allows MEDICREA to Create New FDA-Approved Spinal Implants Giving Patients Dramatic Relief
 The excitement surrounding the 3D printing industry, resulting in countless mind-boggling innovations, is an energy and a source of endless wonder that we have the privilege of immersing ourselves in every day here at 3DPrint.com. And while it’s an ongoing learning experience regarding so much, from tempting new hardware in the marketplace to innovations like friendly 3D printed robots, and even complex governmental strategies overseas for homes and infrastructure, it’s obvious that the most important innovations in 3D are those that are able to bring people a better way of life, allowing them to enjoy quality time with their families and mobility once again.
The excitement surrounding the 3D printing industry, resulting in countless mind-boggling innovations, is an energy and a source of endless wonder that we have the privilege of immersing ourselves in every day here at 3DPrint.com. And while it’s an ongoing learning experience regarding so much, from tempting new hardware in the marketplace to innovations like friendly 3D printed robots, and even complex governmental strategies overseas for homes and infrastructure, it’s obvious that the most important innovations in 3D are those that are able to bring people a better way of life, allowing them to enjoy quality time with their families and mobility once again.
One c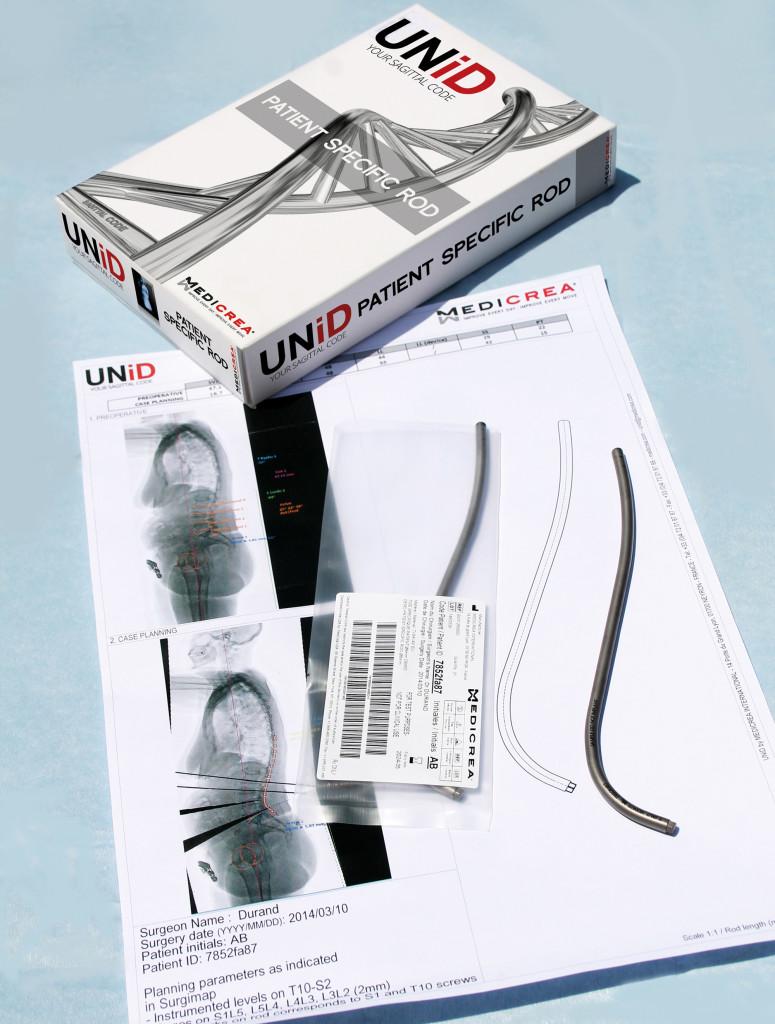 an see traditional processes beginning to change in ways we simply never expected thanks to 3D printing technology—and so much for the better. But this can’t be better exemplified anywhere other than the medical arena. It’s with daily gratitude that we see one miraculous new invention after another appearing, as doctors and researchers creating and using 3D technologies are not just changing the world, they are making people feel better. And in some cases, allowing patients to live.
an see traditional processes beginning to change in ways we simply never expected thanks to 3D printing technology—and so much for the better. But this can’t be better exemplified anywhere other than the medical arena. It’s with daily gratitude that we see one miraculous new invention after another appearing, as doctors and researchers creating and using 3D technologies are not just changing the world, they are making people feel better. And in some cases, allowing patients to live.
For those enduring chronic pain, companies like MEDICREA, constantly bringing new innovations to the spinal implant industry, are making a substantial difference for patients who are actually able to regain their daily lives.
The Lyon, France headquartered company (with subsidiaries in the US, Germany, and UK also) has just announced FDA approval of the very first patient-specific cervical rod, called UNiD. Designed for spinal surgeries, they state that this was ‘secured’ by their PASS OCT posterior cervical stabilization system, which is complementary to the device. Not only has the UNiD rod been approved, history has already been made with the implantation of one in New York on February 17th—and certainly, many many more to follow.
“The patient-specific cervical rod option is a natural extension of the UNiD portfolio. By providing patient-specific realignment implant options for the entire spine, we are again confirming MEDICREA’s leadership position in personalized, made-to-measure treatments,” said Denys Sournac, President and CEO. “This continued expertise brings pioneering technologies and services to the spine that are uniquely dedicated to improving clinical results for each patient.”
 MEDICREA immediately went to work expanding the UNiD technology upon the FDA clearance and they have clarified that their services for spinal alignment will cover:
MEDICREA immediately went to work expanding the UNiD technology upon the FDA clearance and they have clarified that their services for spinal alignment will cover:
- Cervical
- Thoracic
- Lumbar
“The service model for the UNiD cervical rod follows the same effective collaboration between MEDICREA’s UNiD lab and spinal surgeons worldwide, which has surpassed the creation of custom UNiD thoracolumbar rods for more than 650 implantations to date,” states the company in their most recent press release.
What makes this rod and the resulting surgery so unique is that the company is able to use 3D printing technology to achieve what we hear so much about today—patient-specific care. And in this case, it’s patient-specific surgery too, offering a real gamechanger for the surgeons themselves. The rod’s shape is matched with the patient’s own spinal alignment, and allows for a customized plan for the procedure.
The level of customization available is key because this new product is able to directly affect the position of the head and neck—and according to the team at MEDICREA, this dramatically changes how the patient feels. That’s exactly what individuals suffering from constant pain want to hear as they consider a major surgery, and as pioneers in the custom spinal implant industry, the MEDICREA team was able to use new 3D technology to allow for greater relief in patients who truly are suffering.
Not only do patients feel better, but MEDICREA is supplying surgeons with better tools, offering more streamlined solutions that are less invasive, faster, and easier—and all of this means less time in the operating room and less time for patients under anesthesia.
“I am so happy to have the UNiD cervical rod available. This pre-contoured patient-specific implant has eliminated the difficulty of manually bending transition rods, saving OR time and optimizing the execution of my operative plan,” said Dr. Themistocles Protopsaltis, noted as the first surgeon to use MEDICREA’s UNiD patient-specific services on the cervical spine. “My professional collaboration with MEDICREA’s UNiD lab translated easily into a seamless intra-operative experience and the best possible care I can offer my patient—a personalized treatment.”
“It’s quite a challenging patient population as there are patients who are debilitated, can’t stand up very well, can’t walk very well, can’t do their activities in daily living, Getting them better alignment with some special tools that our partners at MEDICREA have been able to give us to help us optimize our corrections, we can get these patients back to living the live that they really want to live.”
 For surgeons still using more traditional implants without the assistance of 3D technology, they don’t have the luxury of precise, fine-tuned rods and must instead ‘approximate’ the rod shape, manually. This can be very challenging, and doubly so when two rods are being used. This requires the utmost precision and cannot be performed with the naked eye.
For surgeons still using more traditional implants without the assistance of 3D technology, they don’t have the luxury of precise, fine-tuned rods and must instead ‘approximate’ the rod shape, manually. This can be very challenging, and doubly so when two rods are being used. This requires the utmost precision and cannot be performed with the naked eye.
“The UNiD Cervical Rod technology removes this existing surgical barrier by creating two strictly identical single or dual-diameter rods manufactured specifically for the patient and delivered directly to the operating room,” states the Medicrea team in their press release.
The rods are going to be available in two alloys—Titanium TA6V ELI/Cobalt Chromium—and in a single-diameter (3.5mm) or dual-diameter option (3.5mm transitioning to either 5.5mm or 6.0mm). They are secured using MEDICREA’s versatile PolyAxial Spine System (PASS) posterior fixation platform, PASS OCT Posterior Cervical Stabilization System, which allows 11mm of gentle reduction capability across all anchorages and medial or lateral rod placement.
Most will agree—even without a medical degree—this is certainly a novel technology, aided by the futuristic world of 3D. We will continue to follow as many more surgeons around the globe realize the benefits in using the UNiD rods, allowing them to better plan out their surgeries, execute their operative plans without error, reduce the opportunity for rod failure, and be in and out of the operating room faster. Discuss in the 3D Printed Medicrea Implants forum over at 3DPB.com.
Subscribe to Our Email Newsletter
Stay up-to-date on all the latest news from the 3D printing industry and receive information and offers from third party vendors.
Print Services
Upload your 3D Models and get them printed quickly and efficiently.
You May Also Like
Astrobotic Tests Rocket Engine Made with Elementum 3D Materials
Astrobotic has completed a series of hot-fire tests for its Chakram rotating detonation rocket engine, with additive manufacturing (AM) playing an important role in how the engine was built. The...
ARC & ORNL Form Partnership to Accelerate AI-Enabled Manufacturing for Defense
Last year, Autonomous Resource Corporation (ARC) became the surprising owner of Desktop Metal’s (DM’s) assets following the bankruptcy of the one-time additive manufacturing (AM) unicorn, an acquisition that cost ARC...
HP Stock Jumps on 3D Printing Buzz
HP (NYSE: HPQ) had its best day in over a year this week, with shares jumping more than 7% on Tuesday. Interestingly, the move was quickly tied to 3D printing,...
HP’s MJF 1200 Targets Entry-Level AM — And Could Shift the Competitive Landscape
HP has launched an entry-level MJF solution, the HP Multi Jet Fusion 1200 3D Printer Solution, which will be available in 2027. What is the 1200 exactly? What does it...











































