The FDA Releases Draft Guidance for Industry & Food & Drug Administration Staff Regarding 3D Printing
 When a magical new technology comes along, capable of stunning us at nearly every turn, it’s especially thrilling to be one of the rare few to be involved from the beginning. Users are operating on a new frontier—and a lawless one for the most part, to begin with. Eventually though, concerns arise, and so do rules. And indeed that is the case with 3D printing, as from issues regarding intellectual property to those of federal approval, laws and guidelines are slowly evolving. While we are a country that often takes pride in independence and eschews government involvement seeping into every nook and cranny of our lives, most do appreciate guidelines when it comes to the medical field.
When a magical new technology comes along, capable of stunning us at nearly every turn, it’s especially thrilling to be one of the rare few to be involved from the beginning. Users are operating on a new frontier—and a lawless one for the most part, to begin with. Eventually though, concerns arise, and so do rules. And indeed that is the case with 3D printing, as from issues regarding intellectual property to those of federal approval, laws and guidelines are slowly evolving. While we are a country that often takes pride in independence and eschews government involvement seeping into every nook and cranny of our lives, most do appreciate guidelines when it comes to the medical field.
And those have been coming along faster than most probably imagined, with more than 85 FDA applications approved for more 3D printed low level medical devices. Often, medical professionals are responsible for or involved in the creation of 3D printed models and devices, and have been looking toward the advent of specific guidelines so that more safety is ensured across the board. Now, the FDA has indeed moved closer to that with a new draft guidance for those pursuing 3D printing in regards to medicine. It’s basic, but it’s a start.
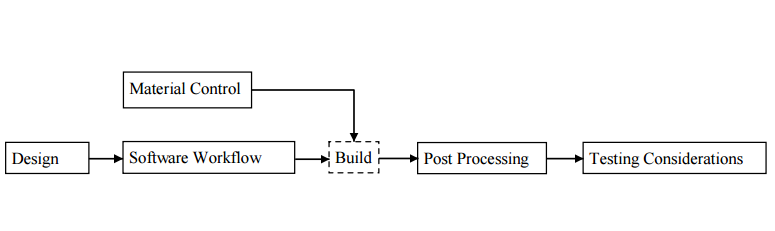
In ‘Technical Considerations for Additive Manufactured Devices: Draft Guidance for Industry and Food and Drug Administration Staff,’ the powers that be at the FDA indicate that as the popularity of 3D printing grows and seeps into the world of medicine, they’ve created this latest document to outline technical considerations regarding 3D printing, as well as offering ‘recommendations for testing and characterization for devices that include at least one AM fabrication step.’ The draft includes information regarding design and manufacturing considerations and device testing considerations. It is important to note that this guidance does not apply to the more complex processes going on in 3D printing—or bioprinting—such as those involving cells or human tissues.
The guidelines offer information regarding designs, whether they are standard or patient-specific, outlining concerns regarding alterations, and the effects they can have on the patients. The FDA points out that designs should result from strict parameters and recommends the obvious need for quality assurance at all points. The effects of imaging and interaction with various design models are to be carefully considered. Software and file conversions are discussed, along with the ins and outs of translating a digital design to a physical one:
“When a digital device design is finalized, additional preparatory processes are needed before the device can be additively manufactured. This is commonly accomplished using build preparation software. These processes can generally be divided into four steps: 1) build volume placement, 2) addition of support material, 3) slicing, and 4) creating build paths.”
The items put forth for guidance by the FDA certainly don’t seem to be overly aggressive by any means, and really point out what should be obvious to anyone making 3D printed devices—such as testing for performance characteristics similar to non-3D printed devices, but keeping in mind the difference in manufacturing.
“Since mechanical properties of the device may be impacted by orientation and location, it is important to ensure that production processes are properly developed, conducted, controlled, and monitored to ensure devices or components are not adversely affected by fabrication orientation,” says the FDA.
 While much of the guidelines are elementary, certainly more complex guidelines will come to pass in the fairly near future. Currently though, information regarding supports, residue, and cleaning processes are included, along with tips on slicing procedures when designing medical devices:
While much of the guidelines are elementary, certainly more complex guidelines will come to pass in the fairly near future. Currently though, information regarding supports, residue, and cleaning processes are included, along with tips on slicing procedures when designing medical devices:
“If the energy density is changed to reduce layer thickness and the additives are not adjusted properly, the layers may not cure or bond together completely,” says the FDA. “For systems where layers are created by melting the material, the layer thickness can similarly influence the energy needed to create a uniform melt pool to enable bonding to the layer below.”
“Your choice of layer thickness should be documented, and reflect a balance among the above-mentioned effects, accuracy, quality, and printing speed.”
Specifications regarding materials should be well-observed and well-documented, and “when any material specification is changed, the effect on the build process and the final device should be well understood and documented.” Safety in regards to the patient is of course of major concern as well:
“Given the iterative nature of AM, the starting material can be exposed to partial re-melting and solidification processes multiple times, which may result in unexpected or undesired material chemistries for some polymer systems. Therefore, if biocompatibility is not evaluated as described in the guidance of International Standard ISO-10993, use ‘Biological Evaluation of Medical Devices 952 Part 1: Evaluation and Testing,’” or if biocompatibility testing identifies a concern, additional material chemistry information may be needed…”
The FDA also covers issues regarding cleaning, sterilizing, biocompatibility, and labeling. All of these guidelines have come into being after feedback received from a 2014 public workshop related to 3D printing challenges. They are keenly aware that 3D printing offers unique challenges, and as more information—and issues—comes to light, their guidelines are sure to evolve further. What did you think of this information? Discuss in the FDA Draft Guidelines for 3D Printing Medical Models forum over at 3DPB.com.
[Source: RAPS]Subscribe to Our Email Newsletter
Stay up-to-date on all the latest news from the 3D printing industry and receive information and offers from third party vendors.
Print Services
Upload your 3D Models and get them printed quickly and efficiently.
You May Also Like
3D Printing Financials: 3D Systems Returns to Growth in Q1 2026
3D Systems (NYSE: DDD) reported one of its strongest quarters in recent years, showing signs that the company may finally be moving past the tough slowdown that has weighed on...
3D People Case Study Details Development of 3D Printed POV Camera Rig
A POV, or Point of View, camera rig, is a wearable support system that helps filmographers capture first-person footage, making the images more immersive. Some good examples of movies shot...
3D Printing News Briefs, May 2, 2026: Soft Robots, Agricultural Waste, & More
In this weekend’s 3D Printing News Briefs, we’ll start off with a multi-laser metal powder bed fusion 3D printer and post-processing news. We’ll end with research into soft robotics and...
Industrial Applications on Display at RAPID 2026: CERATIZIT & 3D Systems
Applications are where it’s at in the additive manufacturing (AM) industry. At the recent RAPID+TCT in Boston, I met with a few companies to learn about some of their very...