K2M Group Expands 3D Printed Spinal Solutions Portfolio Following FDA Clearance for CASCADIA Titanium Systems
 For most, magic is in the air from the moment you unpack that 3D printer, unveiling what is usually an elegant looking machine that acts as the bearer of many things good—allowing us to act on concepts and designs like never before, whether engaged in making modern jewelry, household goods, car parts, or so many other areas as the list becomes nearly infinite. From the desktop at home or workshop, there’s no doubt that creative joy is being taken to a new level, and a self-sustainable, affordable one at that.
For most, magic is in the air from the moment you unpack that 3D printer, unveiling what is usually an elegant looking machine that acts as the bearer of many things good—allowing us to act on concepts and designs like never before, whether engaged in making modern jewelry, household goods, car parts, or so many other areas as the list becomes nearly infinite. From the desktop at home or workshop, there’s no doubt that creative joy is being taken to a new level, and a self-sustainable, affordable one at that.
So just imagine being a researcher, taking that one giant step further, in the medical field. Today, scientists and companies engaged in R&D all over the world have made nearly unfathomable strides, offering that 3D printing magic to others, and often helping to relieve suffering from and even reversing some conditions. While bioprinting is a very hot topic and the race to the finish line for 3D printing human organs is on like no other, the world of 3D printed models and implants is very real—and the benefits being offered to patients today are unprecedented—and much welcomed.
Due to 3D printed implants, many patients now have a new lease on life they never expected. And titanium is very much on the radar today for allowing this due to its strength in combination with 3D printing. Thanks to K2M Group Holdings, Inc. and their recent FDA 510(k) clearances, the material will be helping many more in the form of CASCADIA Cervical and the CASCADIA AN Lordotic Oblique Interbody System featuring Lamellar 3D Titanium Technology, responsible for using advanced 3D printing to make innovative new medical devices.
 K2M is a global medical device company that designs, develops, makes, and markets proprietary complex spine and minimally invasive spine technologies and techniques. The use of titanium powder permits them to make 3D printed implants which are ‘grown’ with an intense laser beam that results in the creation of a porous structure that allows for the re-growth of bone, and more specifically, bony integration throughout an implant.
K2M is a global medical device company that designs, develops, makes, and markets proprietary complex spine and minimally invasive spine technologies and techniques. The use of titanium powder permits them to make 3D printed implants which are ‘grown’ with an intense laser beam that results in the creation of a porous structure that allows for the re-growth of bone, and more specifically, bony integration throughout an implant.
“I’m excited to have the Lamellar 3D Titanium Technology available for the cervical spine as it presents a balance of roughness and porosity that allows the potential for bone to grow into the implant,” said Tom Morrison, MD, a neurosurgeon at Polaris Spine & Neurosurgery Center in Atlanta, Georgia, who completed the first surgical case using CASCADIA Cervical Interbody System. “Additionally, the biomechanical stiffness of the CASCADIA implants are similar to PEEK and less than a more-traditional solid titanium design.”
As is often the case with 3D printing, models and implants that were once not able to be made traditionally can now be affordably manufactured with one of the greatest benefits of all: customization. The CASCADIA Cervical Interbody System is an innovation designed to relieve suffering for patients with degenerative spinal disorders. The system operates as a fusion device allowing for greater support and stabilization, and ultimately, comfort. While it’s one thing to read about such conditions, to suffer from one is a terrible thing, and receiving a solution for that pain is a gift beyond measure.
K2M’s CASCADIA AN Lordotic Oblique Interbody System is another such innovation bringing relief and treatment to those who suffer from degenerative disc disease (DDD) and degenerative scoliosis, and is cleared for use with both autologous and allogenic bone graft tissue in such patients. Surgeons are provided with a comprehensive lineup of anatomically designed interbodies for oblique placement through a transforaminal-lumbar approach.
“3D printing is a strategic priority for K2M, and we are excited to expand our portfolio to include the CASCADIA Cervical and CASCADIA AN Lordotic Oblique Interbody Systems featuring our innovative Lamellar 3D Titanium Technology,” stated K2M President and CEO Eric Major.
“K2M was the first leading spine company to introduce a 3D-printed titanium interbody device and is now the only company to offer a complementary allograft solution in the United States. Through continued development efforts and subsequent product launches, K2M offers the most comprehensive portfolio of FDA-cleared 3D-printed spinal devices on the market, cementing K2M as the leader in the 3D printing of spinal devices.”
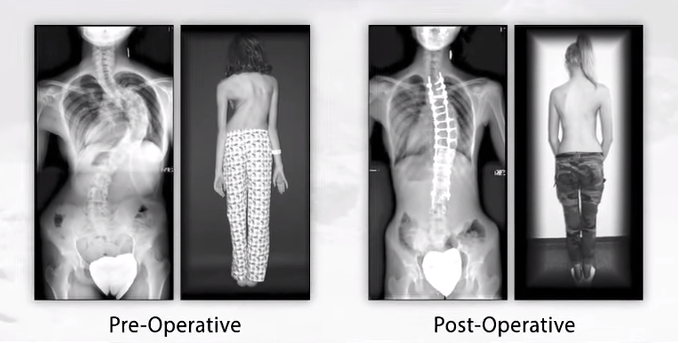 While the two regulatory clearances represented substantial progress, the global medical device company also received additional clearance to commercialize other offerings for both the CASCADIA TL and CASCADIA Cervical Interbody Systems. The whole portfolio is rounded out by the CASCADIA Lateral and AN Interbody Systems also—both of which in the past year were also given 510(k) clearance from the FDA.
While the two regulatory clearances represented substantial progress, the global medical device company also received additional clearance to commercialize other offerings for both the CASCADIA TL and CASCADIA Cervical Interbody Systems. The whole portfolio is rounded out by the CASCADIA Lateral and AN Interbody Systems also—both of which in the past year were also given 510(k) clearance from the FDA.
On a mission to become the leading company in the world for complex spinal solutions, it would seem that K2M is well on their way, along with improving the lives for many who are suffering from spinal pathologies. See K2M to find out more about their technologies and techniques used by spine surgeons to solve difficult spinal pathologies. Discuss this latest technology to receive FDA clearance over in the K2M 3D Printed Titanium Implants forum at 3DPB.com.
[Source: OrthoSpineNews]
Subscribe to Our Email Newsletter
Stay up-to-date on all the latest news from the 3D printing industry and receive information and offers from third party vendors.
Print Services
Upload your 3D Models and get them printed quickly and efficiently.
You May Also Like
Reinventing Reindustrialization: Why NAVWAR Project Manager Spencer Koroly Invented a Made-in-America 3D Printer
It has become virtually impossible to regularly follow additive manufacturing (AM) industry news and not stumble across the term “defense industrial base” (DIB), a concept encompassing all the many diverse...
Inside The Barnes Global Advisors’ Vision for a Stronger AM Ecosystem
As additive manufacturing (AM) continues to revolutionize the industrial landscape, Pittsburgh-based consultancy The Barnes Global Advisors (TBGA) is helping shape what that future looks like. As the largest independent AM...
Ruggedized: How USMC Innovation Officer Matt Pine Navigates 3D Printing in the Military
Disclaimer: Matt Pine’s views are not the views of the Department of Defense nor the U.S. Marine Corps Throughout this decade thus far, the military’s adoption of additive manufacturing (AM)...
U.S. Congress Calls Out 3D Printing in Proposal for Commercial Reserve Manufacturing Network
Last week, the U.S. House of Representatives’ Appropriations Committee moved the FY 2026 defense bill forward to the House floor. Included in the legislation is a $131 million proposal for...


































