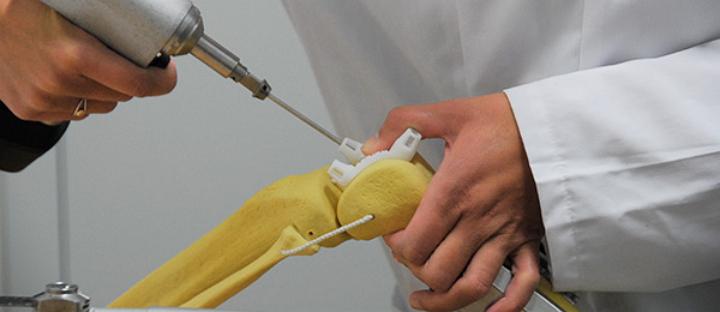
Materialise NV has signed an agreement with Lima Corporate for the use of surgical guides for partial knee implants, and now Lima will offer the Materialise-powered surgical knee guide system in the EEA region and Switzerland. The deal means Lima will now collaborate with Materialise in the design and production of surgical guides which allow surgeons to plan surgeries in 3D and use 3D printed, patient-specific guides.
Back in 1945, Carlo Leopoldo Lualdi set up Lima and began production of surgical instruments, and by 1978 the company began a cooperative effort with the Orthopaedic Surgery Institute in Udine, Italy. Now Lima Corporate is a global medical device company providing reconstructive orthopedic solutions to surgeons, and they design, develop, manufacture and market joint replacement and repair solutions that include large joint implants for hip and knee and systems.
 Lima will use the Materialise Knee Guide System – 3D surgical planning software and patient-specific surgical guides for knee surgery – which allows teams to plan knee surgeries based on a patient’s unique anatomical and structural requirements. The system is used to make pre-operative plans and fabricate patient-specific guides which are then 3D printed for use during operative procedures.
Lima will use the Materialise Knee Guide System – 3D surgical planning software and patient-specific surgical guides for knee surgery – which allows teams to plan knee surgeries based on a patient’s unique anatomical and structural requirements. The system is used to make pre-operative plans and fabricate patient-specific guides which are then 3D printed for use during operative procedures.
Materialise say the Knee Guide System is an end-to-end solution, and the first total knee surgery to be planned using the Knee Guide System was done during September of 2014. The technology proceeds from pre-planning of knee replacement surgeries all the way through 3D printing necessary surgical guides fabricated from data take from 2D X-ray images.
“Materialise first pioneered medical image based guide technology, including solutions for the knee, as part of our company-wide mission to realize a better and healthier world through meaningful applications of 3D printing,” says Jeroen Dille, the Director of the Materialise Clinical Unit. “Through this collaboration with Lima, orthopedic surgeons will be able to benefit from the advantages that 3D printing can offer in the planning and execution of partial knee arthroplasty.”
Luigi Ferrari, the CEO of Lima Corporate, says the system is an ideal addition to the stated mission of his company.
“Lima is committed to supporting healthcare professionals in their daily efforts to improve the lives of their patients and to enabling better outcomes for patients and healthcare systems. The aim of our collaboration with Materialise is to allow the surgeons we work with to continue to pre-operatively plan and use patient-specific guides in pursuit of more predictable surgical outcomes,” Ferrari said of the agreement.
 Materialise now has 25 years of experience in 3D planning and printing for medical applications, and that experience includes the development of dedicated 3D visualization and planning software. The company also offers engineering and design services and produces 3D printed, patient-specific guides and implants.
Materialise now has 25 years of experience in 3D planning and printing for medical applications, and that experience includes the development of dedicated 3D visualization and planning software. The company also offers engineering and design services and produces 3D printed, patient-specific guides and implants.
Dille says that, based on a pre-operative plan, patient-specific guides are designed and 3D printed for use during surgery and the agreement means Lima Corporate will offer the solution to its surgeons for use with a unicondylar knee system Lima recently acquired from Zimmer.
Lima Corporate also offers the largest number of EBM machines for additive manufacturing dedicated to the medical device sector in the world.
What do you think of this agreement between Materialise and Italian orthopedic specialists Lima Corporate? Let us know in the Materialise and Lima Corporate forum thread on 3DPB.com.
Subscribe to Our Email Newsletter
Stay up-to-date on all the latest news from the 3D printing industry and receive information and offers from third party vendors.
You May Also Like
3D Printing News Briefs, April 13, 2024: Robotics, Orthotics, & Hypersonics
In 3D Printing News Briefs today, we’re focusing first on robotics, as Carnegie Mellon University’s new Robotics Innovation Center will house several community outreach programs, and Ugogo3D is now working...
Rail Giant Alstom Saves $15M with 3D Printing Automation Software 3D Spark
3D Spark has entered into a three-year deal with the rail giant Alstom. Alstom, a transport behemoth with annual revenues of $16 billion, specializes in the manufacture of trains, trams,...
Meltio Expands Global Reach with New Partnerships in the Americas and Europe
Spanish 3D printing manufacturer Meltio has expanded its sales network across the globe. With the addition of three new partners in the United States, Brazil, Argentina, and Italy, Meltio aims...
3D Printing Webinar and Event Roundup: April 7, 2024
Webinars and events in the 3D printing industry are picking back up this week! Sea-Air-Space is coming to Maryland, and SAE International is sponsoring a 3D Systems webinar about 3D...