Disease modeling is an important field to understand in terms of human health for the present and the future. A disease model is an animal or cells displaying all or some of the pathological processes that are observed in the actual human or animal disease. Studying disease models aids understanding of how the disease develops and testing potential treatment approaches. We will take a look at how disease models are constructed currently and how bioprinting will help build disease models in the future.
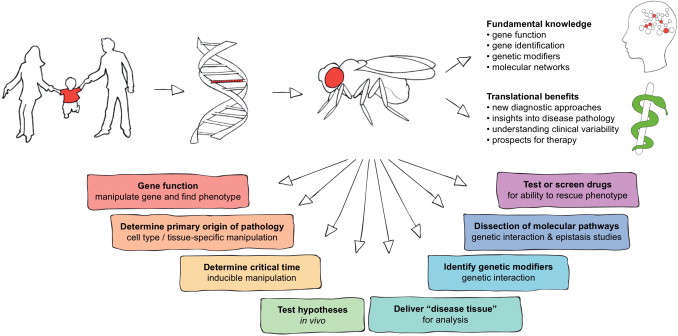
One must know of some preliminary knowledge before we continue to discuss about bioprinting and disease modeling. Disease modeling utilizes different hosts to study underlying pathology of a disease. We can not experiment on a human due to moral and ethical grounds, so instead we typically use a mouse, rat, a pea plant, fish, nematodes, as well as fruit flies for our host site. We then inject said host with a particular disease and it allows us to study how the disease operates within that host. With disease models, we are able to use the information gathered from experimentation to figure out how we can prevent or reduce the spreading of different diseases within the human race.When animal models are employed in the study of human disease, they are frequently selected because of their similarity to humans in terms of genetics, anatomy, and physiology. Animal models are preferable for experimental disease research because of their unlimited supply and ease of manipulation. We can also utilize in vitro cell assays and inject these with different diseases. This allows us to study cell interaction as well.
Rodent Model of Parkinson’s Disease
With the methodology roughly outlined, we will now explain how bioprinting is important for this field. Conventional two-dimensional (2D) in vitro assays and animal models have been unable to fully recapitulate the critical characteristics of human physiology. Physiology refers to how organisms, organ systems, organs, cells, and biomolecules carry out the chemical and physical functions that exist in a living system. Alternatively, three-dimensional (3D) tissue models are often developed in a low-throughput manner and lack crucial native-like architecture. The recent emergence of bioprinting technologies has enabled creating 3D tissue models that address the critical challenges of conventional in vitro assays through the development of custom bioinks and patient derived cells coupled with well-defined arrangements of biomaterials.
Bioprinting is usefull because we can derive materials from human cells to see better reactions without needing to mutate an animal host to become similar to the human genome. There are a variety of diseases that humans contrive but are not within different organisms and their respective genomes. This causes one to necessitate mutation within a host. Ideally one would stay away from mutation when studying disease as that may be a variable that causes imperfect interpretation of results and studies of pathology. Also it is important to consider that physiology of a human is very different from the physiology of a rat, mouse, a plant, and etc. With bioprinting we can actually create scaffolds that have physiological properties similar to human tissue. That allows for more accurate studies as well of interaction from a physiology and pathology framework.
There still lies some areas of concern in terms of bioprinting and disease modeling. It is important for a bioprinted material used in disease modeling to have the following properties:
- Ability to survive for longer time periods
- Allows for safe cell proliferation
- Allows for safe cell differentiation
In order to study a disease pathology and physiology one needs to be able to trust that the material we are using will be able to last and live long enough for useful data or information collected from experimentation. There does not seem to be a lot of research done currently on degradation times of bioprinted tissue scaffolds. This level of uncertainty causes disease modeling to shy away from bioprinting currently. It is important to have clinical viability, and uncertainty is not needed in clinical settings.
Cell proliferation refers to the process that result in an increase of the number of cells, and is defined by the balance between cell divisions and cell loss through cell death or differentiation. This can be further understood through mitosis. Mitosis refers to a type of cell division that results in two daughter cells each having the same number and kind of chromosomes as the parent nucleus, typical of ordinary tissue growth. It is difficult at this stage to induce such cell proliferation within bioprinted materials. There are various studies that show proliferation within bioprinted materials, but there is not enough work done to be able to safely control proliferation.
Cellular differentiation is the process where a cell changes from one cell type to another. Usually, the cell changes to a more specialized type. Differentiation occurs numerous times during the development of a multicellular organism as it changes from a simple zygote to a complex system of tissues and cell types. Cellular differentiation must be controllable. The ability to tune bioprinting properties as an approach to fabricate stem cell bearing scaffolds and to also harness the benefits of the cells multipotency is of considerable relevance to the field of biomaterials and bioengineering. Without a regimented method to control differentiation within a disease modeling context, various things may go awry within experimentation.
Proliferation and Differentiation
Overall, disease modelling is ripe for change. With bioprinting, the future is intriguing and will be more accurate in term of studying human pathology and physiology of diseases because of us having the ability to derive tissue from patient derived cells. There are still a number of items that need to be changed in order for bioprinting within the disease modelling context to be used more thoroughly, and researchers are attempting to fix those issues.
This article is part of a series that wishes to make bioprinting more accessible. It starts with bioprinting 101, Hydrogels, 3D Industrial Bioprinters, Alginate, Bioinks, Pluronics, Applications, Gelatin, Decellularized Extracellular Matrices, and Tissue Engineering & Regenerative Medicine.
Subscribe to Our Email Newsletter
Stay up-to-date on all the latest news from the 3D printing industry and receive information and offers from third party vendors.
You May Also Like
3D Printing Unpeeled: New Arkema Material for HP, Saddle and Macro MEMS
A new Arkema material for MJF is said to reduce costs per part by up to 25% and have an 85% reusability ratio. HP 3D HR PA 12 S has been...
3D Printing News Briefs, January 20, 2024: FDM, LPBF, Underwater 3D Printer, Racing, & More
We’re starting off with a process certification in today’s 3D Printing News Briefs, and then moving on to research about solute trapping, laser powder bed fusion, and then moving on...
3D Printing Webinar and Event Roundup: December 3, 2023
We’ve got plenty of events and webinars coming up for you this week! Quickparts is having a Manufacturing Roadshow, America Makes is holding a Member Town Hall, Stratafest makes two...
Formnext 2023 Day Three: Slam Dunk
I’m high—high on trade show. I’ve met numerous new faces and reconnected with old friends, creating an absolutely wonderful atmosphere. The excitement is palpable over several emerging developments. The high...