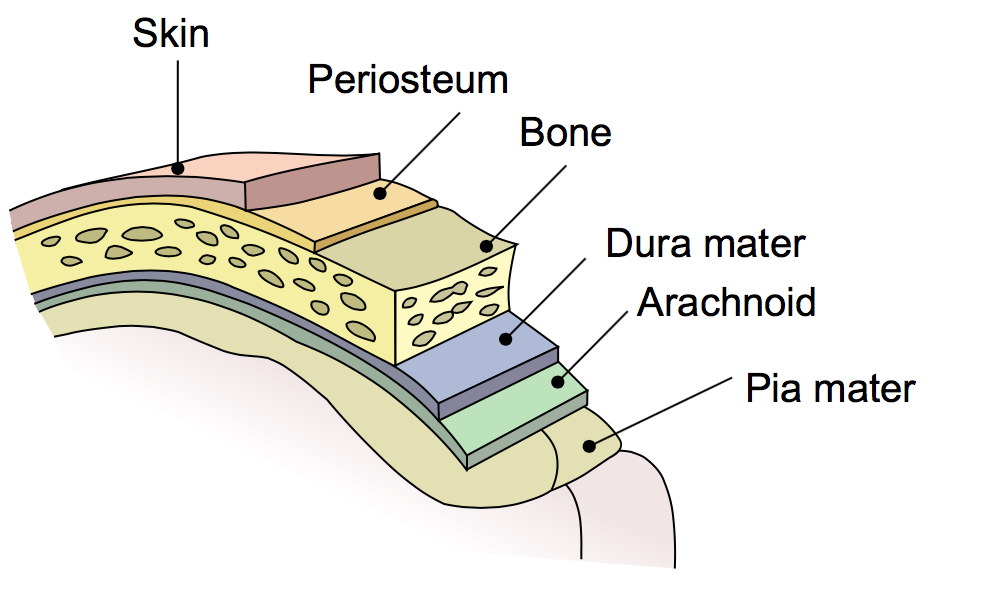
 Whatever value you place on the human mind, the human brain is a wondrous thing. As it sits in the skull, it is protected only by a thin layer of tissue called the Dura Mater. This tissue is divided into two layers, a superficial layer which is the membrane of the inside of the skull, and a deep layer known as the meningeal layer that protects the brain itself. When brain surgery is performed, after the skull is opened, the surgeons must cut through this protective matter in order to access the brain. Once the surgery is complete, something must be done to replace the protective membrane or the patient is put at grave risk.
Whatever value you place on the human mind, the human brain is a wondrous thing. As it sits in the skull, it is protected only by a thin layer of tissue called the Dura Mater. This tissue is divided into two layers, a superficial layer which is the membrane of the inside of the skull, and a deep layer known as the meningeal layer that protects the brain itself. When brain surgery is performed, after the skull is opened, the surgeons must cut through this protective matter in order to access the brain. Once the surgery is complete, something must be done to replace the protective membrane or the patient is put at grave risk.
 With a thickness of only 0.2 millimeters, the dura mater may appear to the eye to be no more than a wet paper towel but it has proved extremely difficult to replace. For this reason, it caused a great stir in the medical community when, earlier this year, Maipu Regenerative Medical Technology, also known as MedPrin, introduced the first generation of artificial dura mater products that had been 3D printed. Chief Technology Officer and Professor at Tsinghua University, Xu Tao described product as an unqualified success:
With a thickness of only 0.2 millimeters, the dura mater may appear to the eye to be no more than a wet paper towel but it has proved extremely difficult to replace. For this reason, it caused a great stir in the medical community when, earlier this year, Maipu Regenerative Medical Technology, also known as MedPrin, introduced the first generation of artificial dura mater products that had been 3D printed. Chief Technology Officer and Professor at Tsinghua University, Xu Tao described product as an unqualified success:
“In March of 2011, the product received CE certification in the European Union. Since then, it has been exported to dozens of counties in Europe and to the United States and is being used in hospitals such as the world-renowned Cambridge University. To date, it has been used in over 10,000 patients without a single report of adverse reactions.”
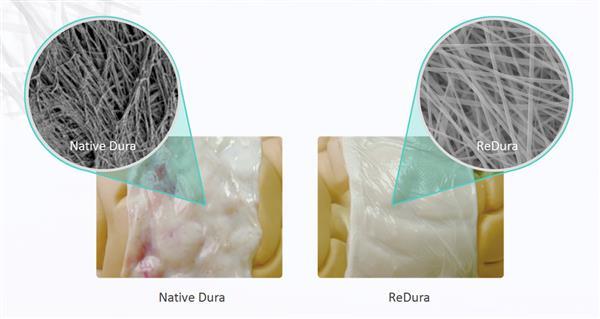
 This 3D printed membrane, named ReDura replaces older techniques for recovering the brain that often left the patients with problematic side effects, not the least of which was its tendency to set off metal detectors as it was embedded with metal filaments. This new product is entirely 3D printed and very closely resembles the patient’s native dura material.
This 3D printed membrane, named ReDura replaces older techniques for recovering the brain that often left the patients with problematic side effects, not the least of which was its tendency to set off metal detectors as it was embedded with metal filaments. This new product is entirely 3D printed and very closely resembles the patient’s native dura material.
This isn’t the kind of thing you just play around with or download from Thingiverse. In fact, to accurately trace its development, we have to go back to 2001 when Xu Tao was a PhD student in the United States studying under Thomas Boland, widely acknowledged to be the father of 3D printed organs. Two years later, Tao successfully printed live cells and two years after that was granted a patent for his amazing invention. In 2009, he teamed up with a faculty member at Clemson University, Yuan Yuyu, and together they founded the first bioprinting company in China. Now, they have continued to build on the promise that Tao showed as a student with this latest revolutionary product.
One of the primary benefits of the ReDura 3D printed membrane is that it is eventually absorbed into the body. As YuYu explained:
“To put it simply, ReDura provides the structure in which cells and tissues can grow. After a period of approximately two months, the meningeal tissue has grown and the ReDura begins to slowly dissolve into two toxin-free components: water and carbon dioxide. After the tissue is fully dissolved and absorbed, there is no foreign material left in place but instead it has been replaced by naturally regenerated dura tissue.”
 The primary difference between the 3D bioprinting method used to create ReDura and what we could now refer to as ‘traditional’ 3D printing is in the printheads. In bioprinting, two print heads are in use; one is filled with human cells known as ‘bio-ink’ and the other with a printable water based gel – a form of biological paper. It is the paper that supports the growth of the cells, numbering in the millions. Tao explained the process:
The primary difference between the 3D bioprinting method used to create ReDura and what we could now refer to as ‘traditional’ 3D printing is in the printheads. In bioprinting, two print heads are in use; one is filled with human cells known as ‘bio-ink’ and the other with a printable water based gel – a form of biological paper. It is the paper that supports the growth of the cells, numbering in the millions. Tao explained the process:
“When the membranes are printed, the machine sprays out a fiber-like ink onto the biological paper. This simulates the collagen structures found in the body. It is formed into hundreds or even thousands of layers of molecular membrane, and the resulting product can be adapted for use in an individualized manner on a case by case basis. The key to successfully bioprinting ReDura is the use of the patient’s own autologous cells in order to prevent the immune system from reacting against what it perceives as a foreign body. In time, this will allow on-demand organ printing to take place.”
 Not ones to rest on their laurels, the team announced that they anticipate a second generation of the ReDura material will likely receive its certification in the next month and be available for distribution and use all over the world. Let us know your thoughts on this innovative approach to a complicated procedure in the 3D Printed Dura Mater forum thread on 3DPB.com.
Not ones to rest on their laurels, the team announced that they anticipate a second generation of the ReDura material will likely receive its certification in the next month and be available for distribution and use all over the world. Let us know your thoughts on this innovative approach to a complicated procedure in the 3D Printed Dura Mater forum thread on 3DPB.com.
Subscribe to Our Email Newsletter
Stay up-to-date on all the latest news from the 3D printing industry and receive information and offers from third party vendors.
You May Also Like
IperionX Inks 10-Year Deal with Wisconsin Manufacturer for 80 Metric Tons of Titanium Per Year
IperionX, the Charlotte-based supplier of sustainable titanium powders used for additive manufacturing (AM) and metal injection molding (MIM), has signed a ten-year deal with United Stars, a group of industrial...
Gastronology Launches Industrial Production of 3D Printed Food for Dysphagia Patients
Food 3D printing has, in many ways, been an additive manufacturing (AM) segment looking for the right business case. While some applications are beautiful and others may or may not...
Lockheed Martin Leads $3M Investment in Q5D’s Electronics 3D Printing System
Q5D, an original equipment manufacturer (OEM) of robotic arm, hybrid additive manufacturing (AM) systems used for wire harness production, has closed a $3 million investment round. The investment arm of...
3D Printing News Briefs, April 6, 2024: Depowdering, Cybertruck Door Handles, & More
In today’s 3D Printing News Briefs, ioTech’s digital manufacturing CLAD technology is opening up opportunities for microelectronics and additive manufacturing. Hexagon and Raytheon Technologies commercially released the Simufact Additive Process...































