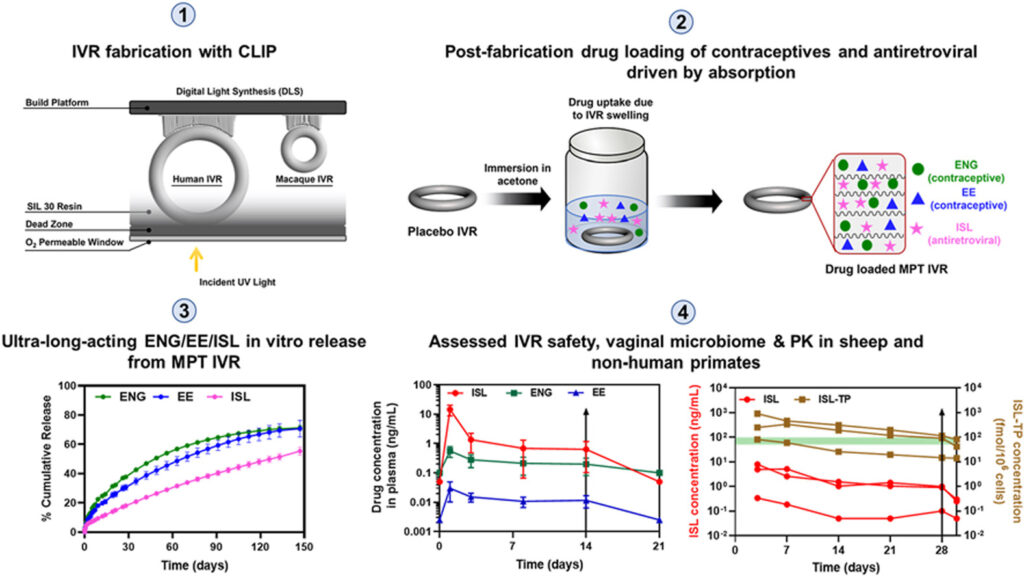
A collaborative effort by researchers from the U.S. Centers for Disease Control (CDC) and University of North Carolina (UNC) Eshelman School of Pharmacy, including Isabella C. Young and Priya Srinivasan, has led to the creation of a novel 3D printed contraception device. This intravaginal ring, printed with Carbon’s vat photopolymerization 3D printing technology and silicone resin, has been designed specifically for women with HIV who often face limited contraception options. In the device, Etonogestrel (ENG), ethinyl estradiol (EE), and islatravir (ISL) were integrated into the silicone poly(urethane) IVR in a controlled single-step drug loading process driven by absorption.
With a focus on empowering women to have control over their contraception, the researchers sought a design that would combine HIV prevention and contraception in one easy-to-use solution. They chose 3D printing as it could potentially offer better release kinetics, thus improving efficacy. Unlike traditional production methods, which use heat and pressure that can interfere with the drugs’ effectiveness in the ring, the team opted for 3D printing, selecting Carbon’s vat photopolymerization technology over material extrusion to avoid heat. This allowed them to use Carbon’s SIL30 material to craft a soft implant comfortable for use within the body. The innovation reflects not only a significant step forward in medical technology but also an earnest response to safer outcomes, especially concerning HIV prevention and women’s health.
Concerns with Photopolymers
I greatly commend the innovative effort to develop 3D printed drug loading devices, and I look forward to seeing more inventions that can be worn in and around the body. This path showcases a promising future for medical technology. However, I must express serious concerns regarding the long-term use of vat polymerization materials within the body.
The material known as SIL30 has approval for skin contact but not for prolonged internal use, such as spending half a year inside the body. Leaching of substances from vat polymerization implants into the body has been reported, including photoinitiators. The material itself contains compounds like 2,2′,2”-nitrilotriethanol, which is linked to reproductive issues and potential harm to unborn children. Additionally, SIL30 can cause skin allergies and contains Diethylene Glycol Monomethyl Ether Methacrylate (2-(2-methoxyethoxy)ethyl methacrylate), a substance that can be acutely toxic and irritate the skin.
While proper curing should eliminate toxicity and related issues, we must be exceedingly cautious with these materials, especially in cases of long-term exposure. We need to recognize that biocompatibility for skin contact is a different consideration than an implant designed to remain in the body for months. As we embrace this new frontier, it is paramount that we take extreme care to safeguard the future development and rollout of this technology over the coming decades.
In pursuing safer alternatives for internal use within the body, we might consider options like the low-temperature material extrusion of polycaprolactone, or perhaps await the creation of a new generation of resins. It’s vital to understand that just because something is labeled as biocompatible or safe for skin contact does not mean it’s suitable for all uses, at all times. We must keep this in mind both in experimental settings and as we plan to bring such materials to market.
The procedures to achieve fully cured parts are equally crucial in this context. While I don’t wish to single anyone out, I must express my skepticism towards the long-term use of any vat polymerization parts within the body. Our industry’s prosperity depends on ensuring the long-term safety of our products, and this must be our guiding principle as we innovate and grow.
Subscribe to Our Email Newsletter
Stay up-to-date on all the latest news from the 3D printing industry and receive information and offers from third party vendors.
Print Services
Upload your 3D Models and get them printed quickly and efficiently.
You May Also Like
Heating Up: 3D Systems’ Scott Green Discusses 3D Printing’s Potential in the Data Center Industry
The relentless rise of NVIDIA, the steadily increasing pledges of major private and public investments in national infrastructure projects around the world, and the general cultural obsession with AI have...
Formlabs Teams Up with DMG MORI in Japan
In late June, Nick Graham, Chief Revenue Officer at Formlabs, announced on LinkedIn that the company had partnered with DMG MORI, one of the world’s leading machine tool companies, to...
EOS in India: AM’s Rising Star
EOS is doubling down on India. With a growing base of aerospace startups, new government policies, and a massive engineering workforce, India is quickly becoming one of the most important...
3D Printing News Briefs, June 25, 2025: R&D Materials, 3D Printed Veneers, & More
In today’s 3D Printing News Briefs, 3DXTECH has launched a program that gives customers early access to experimental materials, and the first Lithoz CeraFab Multi 2M30 in the Czech Republic...