Triastek Succeeds in First In-Human Study of its 3D Printed Medication
Triastek, a Chinese developer of pharmaceuticals produced with additive manufacturing (AM), announced that the company has completed the first in human (FIH) study of T21, a medication for ulcerative colitis (UC). As with all of its products, Triastek prints T21 with its proprietary Melt Extrusion Deposition (MED) platform.
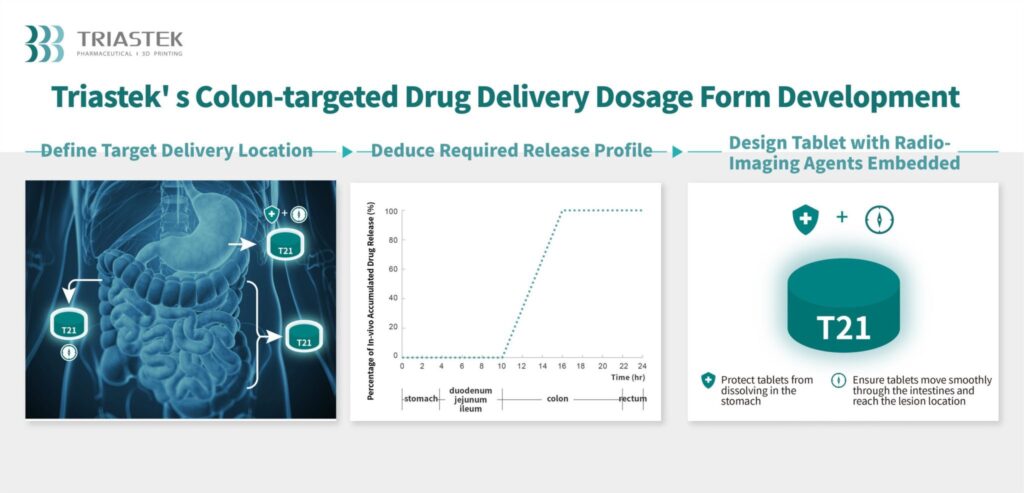
Thus far, Triastek has placed a particular emphasis on designing drugs with geometries that can only be achieved via AM, primarily in order to control where a given medication is released once ingested, and the rate of release of the medication’s active ingredients in the patient’s system. Regarding T21, these features could lead to a breakthrough for individuals suffering from UC, as it is difficult for the oral medications currently on the market to target the colon, specifically.
Based on imaging results from the FIH study, Triastek has shown that upon consumption, T21 tablets are delivered and released directly to the colon. In addition to a breakthrough for UC patients, this could also be a significant accelerator for Triastek’s efforts to gain approval for a New Drug Application (NDA) for T21.
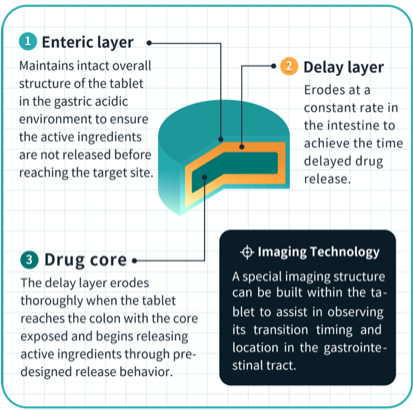
In a press release about Triastek’s completion of its FIH study for T21, Professor Xiaoling Li, the co-founder and chief scientific officer of Triastek, explained, “The [FIH] study data with T21 verifies the precise colon delivery capability of the MED process, and the platform is poised to become the novel drug delivery system of choice for colon targeted new product with either local efficacy or systemic absorption. We hope to continue showcasing how Triastek’s 3D printing processes can bring technical solutions to pharmaceutical companies for efficient product development of optimized drug delivery, ultimately leading to the ability to provide patients with more clinically valuable medicines.”
As I was told by Triastek’s CEO, Dr. Senping Cheng, “…[T21 can mitigate] potential side effects from systemic exposure [that could result] if the medicine was released elsewhere in the body.” Again, this means that using AM for pharmaceuticals could not only lead to all the potential supply chain advantages that have led companies in virtually every industry to start exploring AM: for pharmaceuticals specifically, AM could lead to better quality products right now.
Of course, this is easier said than done, with the regulatory process for medications taking place on a timescale that makes the aerospace sector look like a breeze. On the other hand, the supply shortages in the pharmaceutical industry have been more intractable than virtually any other industry in the last few years, so this could become a catalyst for an accelerated push to use new manufacturing techniques including AM. This is not to suggest that pharmaceutical regulations should be loosened, but rather that corporations and public agencies should work together to prioritize the use of AM and other automation technologies in the cases where positive results have been proven.
Along these lines, it is interesting that in 2022, global pharma giant Eli Lilly announced that it would collaborate with Triastek to research potential new applications for the MED platform. This is relevant here because in April, 2023, the FDA rejected Eli Lilly’s NDA for a new UC drug, mirikuzmab. Notably, the reason for the rejection was related to concerns the FDA had with Eli Lilly’s manufacturing operations in Branchburg, NJ, involving inadequate quality control.
Images courtesy of Triastek
Subscribe to Our Email Newsletter
Stay up-to-date on all the latest news from the 3D printing industry and receive information and offers from third party vendors.
Print Services
Upload your 3D Models and get them printed quickly and efficiently.
You May Also Like
Aibuild Says New FETS Simulation Tool Is 10,000x Faster for AM
Aibuild has launched FETS for Additive Manufacturing, a Finite Element Thermomechanical Simulation tool that lets you simulate stress, distortion, thermal effects, and thermomechanical effects. The solution has been optimized for...
ORNL Continues Research Into Making Large Format Metal Parts by Combining AM and HIP
This can’t be stressed enough: the US needs to secure its supply chains for energy and power generation components. And, while the US needs to prioritize that objective across the...
ARC & ORNL Form Partnership to Accelerate AI-Enabled Manufacturing for Defense
Last year, Autonomous Resource Corporation (ARC) became the surprising owner of Desktop Metal’s (DM’s) assets following the bankruptcy of the one-time additive manufacturing (AM) unicorn, an acquisition that cost ARC...
DOE Backs DMG Mori AI Tool to Speed Qualification of 3D Printed Parts
Since 2015, the U.S. Department of Energy (DOE) has awarded projects that leverage the high-performance computing (HPC) capabilities of the U.S. national laboratories to optimize manufacturing processes. The High-Performance Computing...

















































