ETH Zurich, along with University Hospital Zurich and the University of Zurich, have developed a bioresorbable airway stent. The custom made stents are 3D printed using digital light processing (DLP) and are based on CT scans of patients. The devices are made from a biocompatible resin that is flexible enough to replace the trachea. What’s more, the stent absorbs into the body post-surgery after seven weeks, replacing the need for a surgical procedure to remove the stent.
The material is a dual-polymer photoinks (oligomer) and so far has been tested in rabbits. The team also discloses that they can change and customize the bioresorbability of the stents. This would indicate that they may be able to significantly extend or reduce bioresorbability for different types of parts.

The macromonomers used are functionalized poly(D,L-lactide-co-ε-caprolactone)s [poly(DLLA-co-CL)s], which is analogous to the polycaprolactone (PCL) implants made with FDM and other technologies for stents and trachea implants. I’m a huge fan of PCL and marvel at its capabilities and uses in the body and 3D printing. The Zurich team now can make PCL tunable by mixing high and low molecular weight quantities according to the requirements.
“This resulted in 3D printed objects that combine the enhanced strength arising from high cross-linking density and high elasticity provided by long and flexible polymer chains. Printing at higher temperatures with our customized heating system and the addition of a low-MW polymer in the resin reduced viscosity and enabled the generation of objects with complex architecture at high resolution and surface quality. These inks allowed for the manufacturing of customized prototype bioresorbable airway stents with mechanical properties comparable to state-of-the-art silicone stents.”
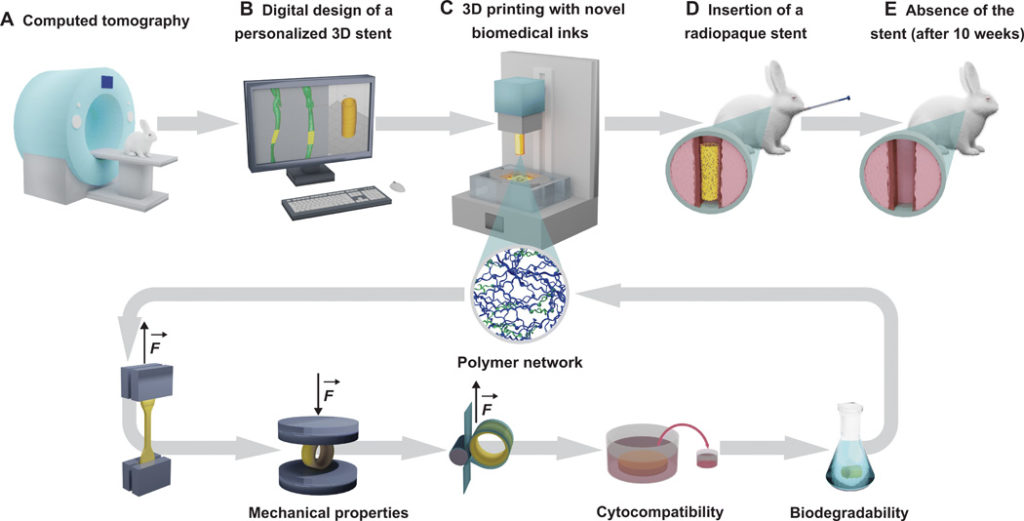
The team also reduced the photoinitiator needed, which is probably necessary for the implant to succeed. The resulting implant didn’t quite have the tensile strength of silicone but mechanical properties were adequate for it to perform its role. They also conducted cytocompatibility tests and, once these were adequate, performed a in vivo study in rabbits. In one of six study participants, the stent migrated “probably due to sneezing”. Live and learn, but the biocompatibility and bioresorbablity was adequate and no stent parts were recovered post mortem. Printed on an Asiga PICO2, this is a great stride forward in using DLP in the body.
We’ve previously looked at how 3D printed stents could be used for esophageal cancer, including a review paper of several esophageal cancer stents and self-expanding stents for esophageal cancer patients. We also reported on self-expanding shape memory nitinol stents, self-expanding stents for pediatric patients, FDM PLA and PCL stents for cardiovascular applications, PVA stents, stents for abdominal aortic aneurysms, teams at Northwestern trying to use Carbon’s technology to make stents, bioprinted vascular prosthetics made from a patient’s own cells and silicone lung stents.
In part, stents are a nice way to get approval from your local ethics committee and are much simpler than alternatives. Stents are also very testable and you can do a lot with them. Shape memory alloy, also called 4D printed or self-expanding, stents also propel this application forward because shape memory could make tiny foldable stents easy to implant via keyhole surgery, for example. They could then be programmed to fold out to be fully functional.
Medicated stents are also a burgeoning area. A stent could have cells printed on it to accelerate growth. The device might also disperse medicine in the body, promote the body’s natural response, or reduce reactions such as inflammation or rejection.
So, partially we’re seeing a real desire by researchers to experiment in the body with 3D printed polymers and, for this, stents are the most effective, demonstrable way to get started at the moment. But, we’re also seeing a very exciting application indeed that is much closer than many other bioprinting or other 3D printing technologies for in-the-body applications. The combination of 4D printing, printed medicine, and bioresorbability could also be a breakthrough area.
On the material and technology side, flexibility in DLP resins has been a bugbear for many companies. At the same time, using stereolithography (SLA) and DLP in the body has been problematic for many years due to cell viability and biocompatibility. Any biocompatibility advances in SLA and DLP can find immediate applications in dental, or other areas, in large volumes, as well.
If this holds up, think of Invisalign not using 3D printing to make the mold for their clear aligners but actually printing out the final aligner for use in your mouth. This is truly a very interesting study that could point the way to broader use of DLP and SLA in the treatment of airway obstructions or, indeed, many other similar issues. There could also be many different applications for these types of stents, as well. Even with regulators and companies hesitant to approve stents or go through the expensive process of moving toward medicated stents, I do believe that there is a lot of promise here. Bioresorbable, temporary structures to help the body heal could really advance medicine.
Subscribe to Our Email Newsletter
Stay up-to-date on all the latest news from the 3D printing industry and receive information and offers from third party vendors.
You May Also Like
IperionX Inks 10-Year Deal with Wisconsin Manufacturer for 80 Metric Tons of Titanium Per Year
IperionX, the Charlotte-based supplier of sustainable titanium powders used for additive manufacturing (AM) and metal injection molding (MIM), has signed a ten-year deal with United Stars, a group of industrial...
Gastronology Launches Industrial Production of 3D Printed Food for Dysphagia Patients
Food 3D printing has, in many ways, been an additive manufacturing (AM) segment looking for the right business case. While some applications are beautiful and others may or may not...
Lockheed Martin Leads $3M Investment in Q5D’s Electronics 3D Printing System
Q5D, an original equipment manufacturer (OEM) of robotic arm, hybrid additive manufacturing (AM) systems used for wire harness production, has closed a $3 million investment round. The investment arm of...
3D Printing News Briefs, April 6, 2024: Depowdering, Cybertruck Door Handles, & More
In today’s 3D Printing News Briefs, ioTech’s digital manufacturing CLAD technology is opening up opportunities for microelectronics and additive manufacturing. Hexagon and Raytheon Technologies commercially released the Simufact Additive Process...































