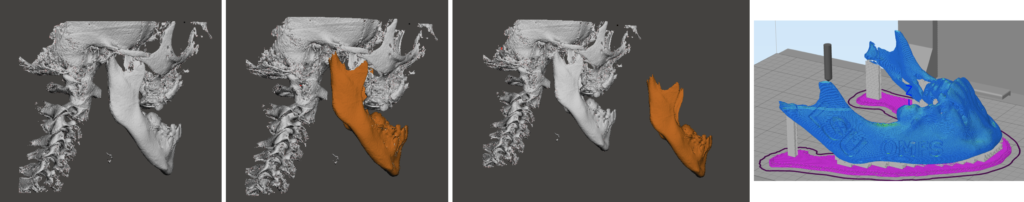
Researchers at Louisiana State University (LSU) developed a new technology to 3D print polyvinyl alcohol (PVA) medical devices. From 3D bone grafts to stents, the development allows for the production of a wide variety of biosynthetic tissues and drug delivery devices, which are very geared towards geometries. Christen Boyer, a bioprinting engineer and recent Postdoctoral Fellow at LSU Health Sciences Center in Shreveport, along with vascular cell biologist, tissue engineer, and professor at LSU Health Sciences Center, Steven Alexander, have been working on a patented 3D printed process that uses PVA to form multi-use medical devices and tissues. Their objective is to help the medical community’s pressing need to have on-demand tissue replacements for patients with diseased or damaged tissues.
They asserted that tissue replacements today suffer from a lack of appropriate donors, tissue typing, availability, homogeneity, and variety, leading to the failure in many types of transplants. And that while current 3D printing for tissues seeks to solve that predicament, it is a highly complex process that requires a specialized laboratory and printing facility. Their solution just received a patent by the United States Patent and Trademark Office and is a novel twist that uses PVA, a tunable material, that is also commonly used in the medical industry.
“By the time Christen came to the lab (LSU Health Sciences Center Laboratory), we began thinking we could use PVA and stabilize it, so we created an approach that uses gas-based 3D chemistries to stabilize it. And this is really kind of a breakthrough because anything can be made, we could 3D print this bioplastic in any shape and so far, it seems to be extremely biocompatible,” described Alexander during an interview with 3DPrint.com.
According to Boyer and Alexander, the method generates biologically compatible 3D printing scaffolds that support cell engraftment because of the high level of protein binding, which is a result of the stabilization process. Additionally, these matrices can be stored frozen until needed, and even frozen 3D milled for patient-specific anatomies.
Alexander went on to say that “this chemistry has actually been described before by other research groups, but what has been primarily done with those is that they have used them to create films and nothing three-dimensional. Instead, we use 3D printing to create a large variety of different shapes that can be made to patient-specific contours and needs and then we use a gas-linked cross-linking process which stabilizes it so that it can be infiltrated with cells, matrix proteins, and more.”
Working along with Hrishikesh Samant, a transplant surgeon at LSU Health, Boyer and Alexander came up with a novel crosslinked PVA (XL-PVA) 3D printed stent infused with collagen, human placental mesenchymal stem cells (PMSCs), and cholangiocytes. The customized living biliary stents have clinical applications in the setting of malignant and benign bile duct obstructions. Overall, this approach may allow physicians to create personalized bio-integrating stents for use in biliary procedures and lays a foundation for new patient-specific stent fabrication techniques.
Their article “3D Printing for Bio-Synthetic Biliary Stents” suggests that a biodegradable stent that dissolves allowing its remnants to be expelled into the duodenum would eliminate the need for a secondary device removal procedure, which would increase the risk for recurrent biliary occlusion or stenosis.
Another study the duo engaged in involved 3D printing antimicrobial and radiopaque constructs. They suggest that PVA 3D printing with iodine represents an important new synthetic platform for generating a wide variety of antimicrobial and high-visibility devices, such as 3D meshes and stents. Boyer considers that PVA loaded with iodine (PVA-I) or other agents could be geared towards bandages custom-made to specific anatomies or even burn victims.
In their study “Three-Dimensional Printing Antimicrobial and Radiopaque Constructs,” the team successfully 3D printed PVA devices, cross-linked the PVA, and then iodized the prints through gaseous sublimation. They claim that this approach provides a highly accessible, inexpensive, and “tunable” architecturally diverse synthetic platform for producing antimicrobial and radiopaque devices and meshes.

“The cross-linking process is quite beautiful under electron microscopy and a key element to the patent’s multi-functionality,” says Boyer (Credit: Boyer and Alexander)
“A lot of times the implanted materials, for instance during a hip replacement, constitute a nidus of infection (that is a focus of infection), that is they become difficult places for the immune system to reach and a hiding place for the bacteria. Instead, if surgeons used a material that had iodine infused in its structure, it would have inherently anti-microbial properties. This is another exciting use of the iodized PVA or PVA-I that we are patenting,” indicated Alexander.
According to the experts, the iodination of implants, such as vascular stents, may create materials that have the ability to reduce or eliminate colonization with bacteria and to reduce the risk of localized infection associated with implantation of foreign materials. Thanks to 3D printing they get to fabricate custom implants with tailored concentrations of iodinated material for imaging and antimicrobial applications.
“We are currently testing them (PVA prints with ceramics and placental stem cells) in cranial implants in rabbits at the LSU Health Science Center, that is, we are using them as non-load bearing bone replacements for rabbits as part of the preliminary studies,” said Alexander. Moreover, we have began a study with the New Iberia Research Center, at the University of Louisiana at Lafayette, where the researchers will be using the 3D printed PVA stem cell scaffolds in cranial implants in non-human primates, as well as looking at non-load bearing bones, which is just the first step as a bone replacement to see how well the material integrates. Our pre clinical tests suggest that it can be used for cancer reconstruction in the next few years. The PVA is superior to the other materials that are out there right now, such as cadaver bone which has a lot of problems with sterility, this is a nicer alternative and it can be made according to computer generated imaging.”
Boyer explains that having 3D printers makes a “huge difference since they allow us to design shapes with porosities that we couldn’t have done with traditional methods: 3D printing opens the door for unique geometries.”
The two managed to transform the lab at LSU into a tissue engineering lab that now has six 3D printers, but the researchers rely heavily on traditional FDM and SLA printers. Also, they depend on Boyer’s programming for creating the G-code and the STL files from the patient data.
As for the future of their collaboration, they plan to keep working together, even though Boyer has recently begun working with bioprinting market leader CELLINK. This year they will extend their work to develop synthetic meniscus using some of the materials they have been working with for the past years. Boyer says they have also begun pursuing licensing with other companies. One example of this is with experienced companies working with synthetic bones to infuse them with growth factors that will make for replacement material. They will also work to create a muscle and epithelial infused replacements for regions where the esophagus of a patient could be damaged.
 During the two and a half year initial collaboration, Boyer and Alexander helped raise over $170,000 in funding for these projects. All the stem cell work is supported by the LSU Center for Tissue Engineering and Regenerative Medicine (CTERM), and their funding comes from the Osteo Science Foundation and the State of Louisiana.
During the two and a half year initial collaboration, Boyer and Alexander helped raise over $170,000 in funding for these projects. All the stem cell work is supported by the LSU Center for Tissue Engineering and Regenerative Medicine (CTERM), and their funding comes from the Osteo Science Foundation and the State of Louisiana.Subscribe to Our Email Newsletter
Stay up-to-date on all the latest news from the 3D printing industry and receive information and offers from third party vendors.
Print Services
Upload your 3D Models and get them printed quickly and efficiently.
You May Also Like
3D Printed Bone Grafts From Georgetown Researchers Could Replace Traditional Implants
Researchers at Georgetown University are developing a new type of 3D printed bone graft designed to work more like real human bone. Instead of relying on metal implants or donor...
TV’s Scarpetta Suggests We Can 3D Print Full Human Organs. Reality Is… Not Yet
Having spent a good part of my journalistic career covering crime stories, it’s hard for me to stay away from any crime or medical forensic drama. So when Scarpetta premiered...
At RAPID + TCT 2026, Executive Keynotes Break Down What’s Next for AM
While AI is expected to be a major focus at RAPID + TCT 2026, the event is also putting the spotlight on something just as important: leadership. This year’s Executive...
3D Printing Moves Deeper Into Production as Parts Near $110B by 2034
A new report takes a closer look at how much 3D printing is actually being used in real production. The numbers point to a market that is already growing at...