Northwestern University Uses 3D Printing to Create Customized Vascular Stents
 There are numerous examples of how 3D printing technology has proved advantageous to the medical field, from customized assistive devices to surgical preparation with patient-specific 3D printed models. This benefit is especially apparent in vascular treatments, whether it be through enhancing medical training or bioprinting vascular stents with human cells. Earlier this year, researchers from Harvard University managed to create 3D printed vascular tissues capable of living up to six weeks. Now, the prestigious Illinois-based Northwestern University is conducting their own similarly revolutionizing work by 3D printing customized vascular stents.
There are numerous examples of how 3D printing technology has proved advantageous to the medical field, from customized assistive devices to surgical preparation with patient-specific 3D printed models. This benefit is especially apparent in vascular treatments, whether it be through enhancing medical training or bioprinting vascular stents with human cells. Earlier this year, researchers from Harvard University managed to create 3D printed vascular tissues capable of living up to six weeks. Now, the prestigious Illinois-based Northwestern University is conducting their own similarly revolutionizing work by 3D printing customized vascular stents.
Led by Guillermo Ameer and Cheng Sun, two professors at Northwestern’s McCormick School of Engineering, the duo has developed a method to 3D print patient-specific vascular stents that are both flexible and biodegradable. Their process is based off of projection micro-stereolithography 3D printing, which enables them to fabricate stents from a specialized polymer formulated in Ameer’s laboratory. Their unique 3D printing technique, which Sun calls micro continuous liquid interface production (microCLIP), works similarly to the breakthrough CLIP technology developed by the 3D printing company Carbon.
The microCLIP process provided the Northwestern researchers with a handful of benefits, such as an extremely high resolution to print features as small as 7 microns. This is especially important for the production of these stents, which have fine mesh dimensions and can sometimes be smaller than 3 millimeters in diameter. The 3D printing technology is also capable of fabricating up to 100 stents at a time, making their process faster and more affordable than traditional methods. In fact, according to the engineering team, microCLIP is capable of printing a 4-centimeter vascular stent in just a matter of minutes.
“There are cases where a physician tries to stent a patient’s blood vessel, and the fit is not good,” Ameer said. “There might be geometric constraints in the patient’s vessel, such as a significant curvature that can disturb blood flow, causing traditional stents to fail. This is especially a problem for patients who have conditions that prevent the use of blood thinners, which are commonly given to patients who have stents. By printing a stent that has the exact geometric and biologic requirements of the patient’s blood vessel, we expect to minimize the probability of these complications.”
While most current stents are made with metal wire mesh, the research team utilized a citric-acid based polymer developed by Ameer, making their stent exceptionally flexible, biodegradable, and constructed with inherent antioxidant properties. These 3D printed stents can also be preloaded with drugs which are released at the site of the implant, improving the healing process in the walls of the blood vessel. The unique polymer material allows the stent to exercise its mechanical function during the vessel’s initial dilation and slowly dissolve as the re-opened blood vessel recovers.
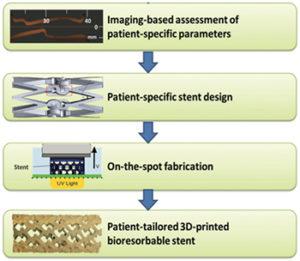 These patient-specific stents are critical to a successful implantation, as ill-fitting stents sometimes move around in the artery and can end up failing in their purpose. When this happens, physicians are required to re-open the blocked stent or bypass it with a vascular graft, a procedure that is both costly and risky. One disadvantage of the 3D printed polymer-based stent is that it is weaker than the traditionally used wire mesh, and may also take longer to fully expand upon deployment. To make up for this fact, Ameer and Sun increased the thickness of their struts to match the strength of a metal stent.
These patient-specific stents are critical to a successful implantation, as ill-fitting stents sometimes move around in the artery and can end up failing in their purpose. When this happens, physicians are required to re-open the blocked stent or bypass it with a vascular graft, a procedure that is both costly and risky. One disadvantage of the 3D printed polymer-based stent is that it is weaker than the traditionally used wire mesh, and may also take longer to fully expand upon deployment. To make up for this fact, Ameer and Sun increased the thickness of their struts to match the strength of a metal stent.
Their research, which was supported by the American Heart Association, was recently published online in the academic journal Advanced Materials Technologies. The research project also included Robert van Lith, a postdoctoral fellow in Ameer’s laboratory, and Evan Baker, a graduate student in Sun’s laboratory, as the co-first authors of the paper. As for the future implications of their research, Ameer plans to investigate how long it takes for the 3D printed stent to break down and absorb into the body. His team is also working to develop more innovative stent designs in order to improve their long-term performance. Discuss in the 3D Printed Stents forum at 3DPB.com.
Subscribe to Our Email Newsletter
Stay up-to-date on all the latest news from the 3D printing industry and receive information and offers from third party vendors.
Print Services
Upload your 3D Models and get them printed quickly and efficiently.
You May Also Like
AM Asia Watch: China’s HeyGears Lands $44M to Expand Beyond Dental 3D Printing
Chinese 3D printing company HeyGears raised more than 300 million Yuan (roughly $44 million) in a new Series C funding round as it looks to expand beyond its industrial and...
The University of Utrecht: “3D Printing Could Change Who Gets to Become a Manufacturing Power”
For decades, manufacturing has mostly been controlled by countries with huge factories, lower labor costs, and industrial systems that took years, sometimes decades, to build. But Utrecht University human geographers...
3D Printing News Briefs, May 28, 2026: Continuous Fiber Reinforcement, Bioprinted Trachea, & More
In today’s 3D Printing News Briefs, America Makes announced the winners of its JAQS-SQ Project Call. Axtra3D is partnering with Keystone Industries to expand its dental material ecosystem, while BigRep...
Asia AM Watch: China’s SHINING 3D Restarts IPO Review Process
SHINING 3D is moving forward again with its plans to go public in China, after restarting its Beijing Stock Exchange (BSE) initial public offering (IPO) review process and filing updated...


















































