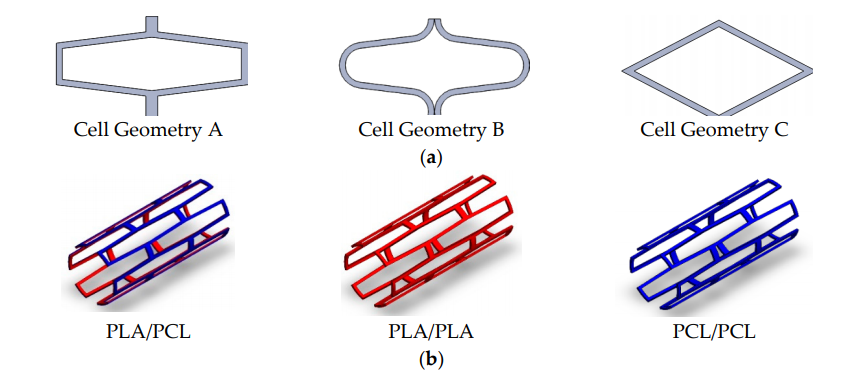
3D Printed PLA and PCL Composite Biodegradable Stents Show Promise
Biodegradable stents have shown great potential in reducing complications in patients, but they require further study, according to the authors of a paper entitled “3D-Printed PCL/PLA Composite Stents: Towards a New Solution to Cardiovascular Problems.” The researchers outline five main requirements that a biodegradable stent must meet:
- Their manufacturing process should be precise
- Degradation should have minimal toxicity
- The rate of degradation should match the recovery rate of vascular tissue
- They should induce rapid endothelialization to restore the functions of vascular tissue but should at the same time reduce the risk of restenosis
- Their mechanical behavior should comply with medical requirements, particularly the flexibility required to facilitate placement but also sufficient radial rigidity to support the vessel
Although the first three requirements have been thoroughly studied, according to the researchers, the last two have been overlooked. A possible way of addressing these issues would be to create composite stents using materials that have different mechanical, biological or medical properties, such as PLA or PCL. Fabricating stents with these materials using laser cutting, however – the traditional method of manufacturing stents – would not be possible. The researchers, therefore, decided to produce them using 3D printing.
 They 3D printed the stents using a tubular 3D printer. The stents were then seeded with cells and left for three days, and then tests were performed to assess the morphological features, cell proliferation, cell adhesion, degradation rate and radial behavior.
They 3D printed the stents using a tubular 3D printer. The stents were then seeded with cells and left for three days, and then tests were performed to assess the morphological features, cell proliferation, cell adhesion, degradation rate and radial behavior.
“The results prove the materials’ biological compatibility and encourage us to believe that PCL/PLA composite stents would comply with the fourth requirement, i.e., rapid endothelization without risk of restenosis,” the researchers state. “PCL’s better cell proliferation may be useful to increase the proliferation of endothelial vessel cells in the external wall of the stents, while an internal PLA wall may help to reduce the proliferation of cells that produce restenosis. However, further studies with other kinds of cells or substances need to be performed to confirm this. The results here show low cell proliferation because of the small amount of material that the stents have. Additional studies that use longer culture times may be beneficial to obtain better proliferation results.”
The researchers’ initial hypothesis was confirmed: the smaller the cell area of the stent, the better the cell proliferation rate. The cell shape of the stent, however, did not show any significant influence. Because of their different molecular weights, PCL showed better cell proliferation than PLA. PLA showed a much faster degradation rate, which limits its use for biodegradable stents. Radial behavior results show that composite PLA/PCL stents could be used to improve each material’s separate limitations, with PCL offering elasticity in the expansion stem and PLA providing rigidity in the recoil step.
Overall, 3D printing proved itself to be a promising method for producing stents. Both PCL and PLA showed themselves to be biocompatible, and the composite stents showed the most promise, with medium levels of degradation rates and mechanical modulus.
“Based on the results presented here, we believe that polymer composite stents manufactured with 3D-printing processes could be a highly effective solution to the current problems that stents made of polymers have,” the researchers conclude. However, FDA rules currently limit the use of 3D-printed stents in real clinical applications and, although PCL and PLA are FDA-approved materials, there are still open challenges to be met before approval for 3D-printed implantable medical devices can be obtained.”
Authors of the paper include Antonio J. Guerra, Paula Cano, Marc Rabionet, Teresa Puig and Joaquim Ciurana.
Discuss this and other 3D printing topics at 3DPrintBoard.com or share your thoughts below.
Subscribe to Our Email Newsletter
Stay up-to-date on all the latest news from the 3D printing industry and receive information and offers from third party vendors.
Print Services
Upload your 3D Models and get them printed quickly and efficiently.
You May Also Like
Printing Money Episode 38: Additive Manufacturing Deal Analysis with Rajeev Kulkarni
Welcome to Printing Money Episode 38. Rajeev Kulkarni returns for this episode, and we find it hard to believe it’s been nearly two years since his first appearance. In the interim,...
3D Printing News Briefs, May 14, 2026: Project Calls, Reseller, Reconstructive Surgery, & More
We’re starting off today’s 3D Printing News Briefs with two new Project Calls from America Makes. We’ll move on to some more business, with Axtra3D expanding its presence in North...
Fabric8Labs & University of Illinois Collaborate on 3D Printed Copper Cold Plates for Data Centers
Collaboration between emerging technology enterprises and research universities is one of the most consistently winning tactics for any nation building (or rebuilding) an industrial ecosystem. It’s an especially constructive approach...
3D Printing News Briefs: May 7, 2026: Metal Powder Bed Fusion, Surgical Plates, & More
In today’s 3D Printing News Briefs, we’ll start with a strategic collaboration to advance next-generation metal additive manufacturing (AM), before moving on to funding for surgical research. We’ll end with...