Florida Atlantic University Researcher Reviews Different Esophageal Stents, Including the 3D Printed Variety
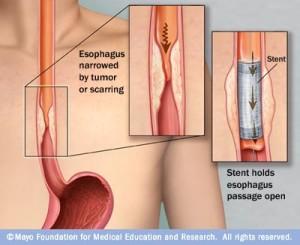 Ranking sixth among all types of cancer in mortality, esophageal cancer kills about 16,000 people in the US annually, with about 500,000 new cases diagnosed around the world last year (according to the American Cancer Society). More than half of these patients are unfortunately diagnosed when the cancer is advanced enough that a tumor blocks the esophagus, which is typically when doctors insert a palliative, self-expanding esophageal stent to allow them to eat and drink. According to a paper, titled “A Review of Self-Expanding Esophageal Stents for the Palliation Therapy of Inoperable Esophageal Malignancies,” that was published by Florida Atlantic University researcher Dr. Yunqing Kang, researchers have been looking into other therapeutic applications for these stents.
Ranking sixth among all types of cancer in mortality, esophageal cancer kills about 16,000 people in the US annually, with about 500,000 new cases diagnosed around the world last year (according to the American Cancer Society). More than half of these patients are unfortunately diagnosed when the cancer is advanced enough that a tumor blocks the esophagus, which is typically when doctors insert a palliative, self-expanding esophageal stent to allow them to eat and drink. According to a paper, titled “A Review of Self-Expanding Esophageal Stents for the Palliation Therapy of Inoperable Esophageal Malignancies,” that was published by Florida Atlantic University researcher Dr. Yunqing Kang, researchers have been looking into other therapeutic applications for these stents.
The abstract reads, “Esophageal stents have evolved in stages over the years. Current clinically used stents commonly include stainless steel or nitinol self-expandable metallic stent (SEMS) and self-expandable plastic stent (SEPS). There are many choices of different types of stents and sizes, with fierce competition among manufacturers. However, current stent technology, whether uncovered, partially covered, fully covered SEMS or SEPS, has their own advantages to solve the dysphagia, stricture, and fistula problems, but they also cause some clinical complications. The ideal stent remains elusive. New 3D printing technique may bring new promising potential to manufacturing personalized esophageal stents. Drug-eluting stents could be the new avenue to do more than just pry open a stricture or cover a defect in the esophageal lumen, a possibility of proving local anticancer therapy simultaneously. Additionally, the lack of esophageal cancer animal models also hinders the progress of stent development. This paper reviews these topics for a comprehensive understanding of this field. In a conclusion, the ultimate goal of the future esophageal stent would have multifunction to treat the underlying conditions and restore esophageal function to near normal.”
 Dr. Kang was actually the recipient of a $141,743 grant three years ago to develop a biodegradable, 3D printed polymer stent that could avoid the complications of metal stents, while at the same time serve as a drug delivery system. He has obviously continued his work, and in this study, he reviewed the applications of a variety of stents, including self-expanding, biodegradable, 3D printed, and drug-eluting.
Dr. Kang was actually the recipient of a $141,743 grant three years ago to develop a biodegradable, 3D printed polymer stent that could avoid the complications of metal stents, while at the same time serve as a drug delivery system. He has obviously continued his work, and in this study, he reviewed the applications of a variety of stents, including self-expanding, biodegradable, 3D printed, and drug-eluting.
Self-expanding metal stents, or SEMS, are the most widely used kind for malignant esophageal cancer, and come in covered, uncovered, and partially uncovered. However, use of SEMS has led to complications like recurrence of tissue growth, bleeding, fistulas, and stent migration. Because they don’t have the painful, rigid metal ends of SEMS, self-expanding plastic stents (SEPS) don’t cause these issues, and can also be easily removed. In addition, they can improve quality of life and dysphagia, and reduce how many dilatation sessions patients with benign structures require. But, while studies show that SEPS have a much higher rate of migration than SEMS, they are often used to treat perforations, fistulas, and esophageal leaks.
“Although SEPS causes high rate of migration, it appears to be safer than metallic stent as the plastic material may not cause significant tissue trauma,” Dr. Kang wrote.
Biodegradable (BD) stents were developed to reduce the number of complications from migration and growth of hyperplastic and tumor growth tissue, as well as to avoid removal.
“Two types of biodegradable polymer stents are available currently,” Dr. Kang wrote. “One is the ELLA-BD stent (ELLA-CS, Hradec Kralove, Czech Republic), which is composed of polydioxanone, a surgical suture material (Figure 2(b)) [35], and the other one is the poly-L-lactic acid (PLLA)-BDstent (Marui Textile Machinery, Osaka, Japan), which consists of knitted PLLA monofilaments [23].”
Biodegradable stents don’t need to be removed once they’re implanted, which means less time on the operating table for patients, and less chance of recurrent dysphagia. But, studies show they’re still likely to migrate, which causes the patient pain.
“From these results, it can be seen that although biodegradable stents may provide a valuable alternative to SEPS and SEMS, and also may eliminate the need for repeat esophageal dilations, biodegradable stents still presented some complications of migration and tissue regrowth. Also, biodegradation may lead to the collapse of stents after placement due to the collapsed degradation of the stent, quickly losing the mechanical strength,” Dr. Kang explained. “From these studies, it can be seen that the degradation properties of a BD stent determine its mechanical integrity. Studies showed that both ELLA-BD stent and PLLA-BD stents, the two currently available BD stents, can be degraded by hydrolysis, which is accelerated at low ambient pH. The stents began to degrade after 4 to 5 weeks and dissolved during a period of 2 to 3 months.”
Dr. Kang concluded that longer term studies are still needed to investigate “the relative efficacy or safety of esophageal biodegradable stents.”
“Therefore, the question of which type of stent should be recommended for the effective treatment of complex and refractory benign strictures, also malignant tumor remains unclear.”
3D printing is also used to create a variety of stents, such as airway stents, vascular stents, and biodegradable stents as well. The technology offers advantages over conventional fabrication of polymer stents, and is our best shot for personalization.

Photographs show the different types of 3D printed stents with different structures and material ratios (a). The stent was compressed and then recovered to the original shape (b).
“In our study, we used a 3D printing technology to produce a flexible polymer esophageal stent (Figure 3) [49]. We found that our 3D printing technique can print an esophageal stent with different size and shape,” Dr. Kang explained. “This is the first study using 3D printing technique to produce a polymer esophageal stent. Although the function of the 3D-printed flexible polymer stent has not been proved in vivo, the in vitro study showed that the 3D-printed esophageal stent has promising potential to treat malignant esophageal diseases. It can self-expand, and 3D printing technique can design and print different sizes and shapes of the stent easily.”
Again, the researcher noted that more study was required.
Dr. Kang also looked at drug-eluting stents (DESs), which are good for vascular applications but not clinically available yet to help treat esophageal cancer. Studies continues on this, and researchers have tried loading anticancer drugs, like 5-fluorouracil and paclitaxel, and whether sustained release of a stent with a bilayer film structure was possible.
Finally, many types of models, like 3D cell spheroid models and animal xenograft/orthotopic ones, are used to evaluate the functionality and efficacy of a new drug delivery or treatment in cancer studies. But it’s not as easy for esophageal cancers with strictures, or benign esophageal diseases, due to the diameter and length of most FDA-approved stents not being applicable in these models. Dr. Kang stated that we need larger-sized animal models, like pigs or dogs, to study esophageal cancer and stents.“This kind of localized sustained delivery system in combination with the stent appeared to be a promising strategy to treat malignant esophagus cancer,” Dr. Kang wrote.
“The challenge is not only because of the complex surgery on a large animal, but also the potential difficulty of inoculating tumor cells or tissue in the orthotopic esophagus for tumor formation,” Dr. Kang explained. “In the future, new animal models or alternative animal modelling technologies still need to be developed and established for esophageal cancer stenting.”
I don’t enjoy thinking about testing medical theories out on animals, but I admit I see the point of needing larger animals for a necessary evil such as this.
Currently, popular palliation therapies for patients with inoperable esophageal malignancies suffering from dysphagia who use esophageal stents includes SEMS, SEPS, and biodegradable stents. But, as Dr. Kang’s research shows, novel stents that can get around some of the existing complications are desperately needed.
“In addition to improving the functionality of the drug-loaded stent with markedly reduced adverse effects, new ideal stents will allow to be tailored to individual needs at much lower cost,” Dr. Kang concluded. “Additionally, there is an unmet need to develop a large animal esophageal cancer model in vivo and establish a functional esophageal cancer model in vitro to test stents and study esophageal cancers.”
Discuss this research, and other 3D printing topics, at 3DPrintBoard.com or share your thoughts below.
Subscribe to Our Email Newsletter
Stay up-to-date on all the latest news from the 3D printing industry and receive information and offers from third party vendors.
Print Services
Upload your 3D Models and get them printed quickly and efficiently.
You May Also Like
Johns Hopkins University Researchers Develop HyFAM Technology
Two scientists from Johns Hopkins University, Nathan C. Brown and Jochen Mueller, have developed a hybrid manufacturing technology they call HyFam, or Hybrid Formative Additive Manufacturing. Their work on this technology...
3D Printing G-Code Gets an Upgrade: T-Code
Good old G-Code still manages many 3D printers, great and small. Just like the STL, it’s a standard that enables collaboration while also holding the additive manufacturing (AM) industry back....
AM Rewind: The Biggest News and Trends of 2024
After a sluggish 2023, driven by persistent inflation and geopolitical tensions, 2024 has seen some recovery. Economic growth climbed from about 2.8 percent in 2023 to a modest 3.2 percent...
Metal Wire 3D Printer OEM ValCUN Announces Plans for 2025 Expansion
ValCUN, a Belgian original equipment manufacturer (OEM) of wire-based metal additive manufacturing (AM) hardware, has announced that the company has entered the next phase of its growth trajectory, making key...




































