Finland: Studying the Impact of Medical Imaging Inaccuracies on End-Use Implants
Finnish researchers report on inaccuracies in the AM process chain, releasing their findings in ‘Cumulative Inaccuracies in Implementation of Additive Manufacturing Through Medical Imaging, 3D Thresholding, and 3D Modeling: A Case Study for an End-Use Implant.’
3D printing emerged in the 80s as a prototyping tool, but over the years designers and engineers have begun to rely on the technology for functional and critical parts in applications like automotive/racing and aerospace; however, the medical realm has been enormously—and positively—impacted with greater access to 3D printed models, devices, and implants.
“AM has been widely implemented in the medical industry, and it has gained clearance for more than 80 AM medical devices such as maxillofacial implants and a knee joint replacement system from the United States Food and Drug Administration (FDA),” state the authors in their study.
“Substantial AM growth is enabled by its economical lot-size-one that allows for unprecedented geometrical freedom in one build cycle using minimal set-up because it is a general-purpose digital technology which is not constrained by patient-specific tooling. Further, it enables a unique supply chain with increased communication and responsiveness that complements the state of urgency in the medical field.”
Customized implants are often made from titanium today, especially for those targeting the head or neck area. Still, there is greater need for porous titanium, able to promote osseointegration with the correct mechanical properties in place. There is also greater need for error-free imaging, leading to better patient-specific treatment as CT scans are converted to 3D images and consequently, 3D prints. While fan-beam multi-slice CTs (MSCT) are used, and may offer better resolution in terms of low contrast, the cone-beam CT (CBCT) system offers the following benefits:
- Acceptable image quality
- Good scanning time
- Better affordability
- Reduced radiation dose
As 2D images are converted to 3D, however, the use of thresholding techniques is required, whether users employ a volume-based, contour-based, or point-cloud-based approach. Volume-based is most commonly used, via a marching cubes algorithm.
“Each step of the process can contribute geometric error to the resulting implant,” stated the researchers.
Over time, researchers have studied the chain of inaccuracies in CT systems and resulting processes, resulting in questions regarding details like voltage, thresholding values, software, varying AM techniques, varying hardware, and more, leaving the authors here to note that all of these concerns ‘have prompted additional verification studies that account for the whole process chain.’
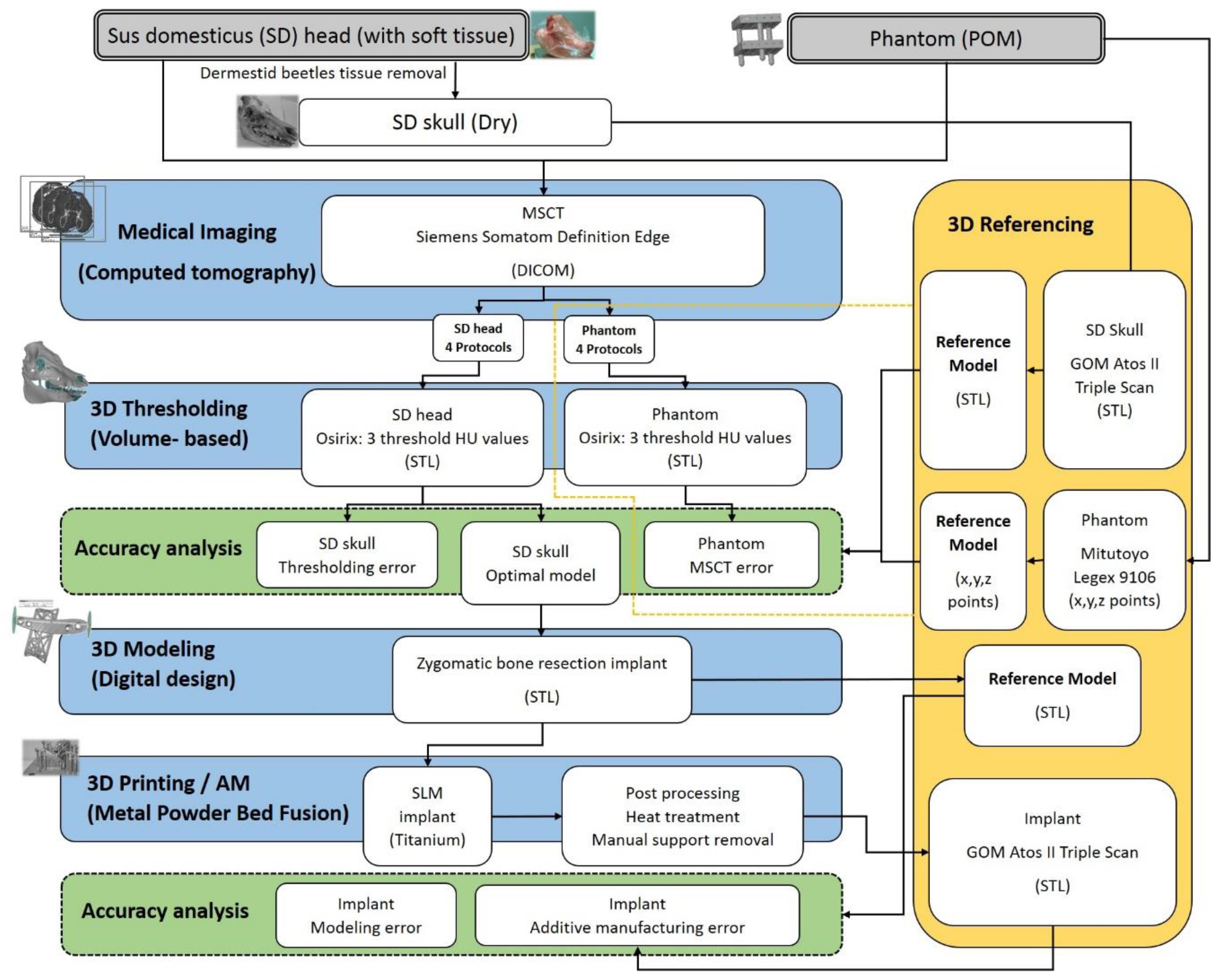
In following a case study involving pre-operative planning for an implant, the authors investigate possible inaccuracies and how they impact an end-use implant.
Using a nine-month old dry skull from a pig, the researchers scanned the specimen and made reference points. They also designed a QA phantom for confirming measurements, made of Polyoxymethylene (POM), a material used for several QA phantoms in other studies.
“Assuming a case where a patient had to undergo a resection of a zygomatic bone tumor, an implant was digitally modeled to replace the deformity using 3Data Expert (11.0.0.32, Deskartes Oy, Espoo, Finland) software. The implant was designed using the right zygomatic bone projections of the SD head that were obtained from the optimal CT imaging and thresholding parameters,” stated the researchers.
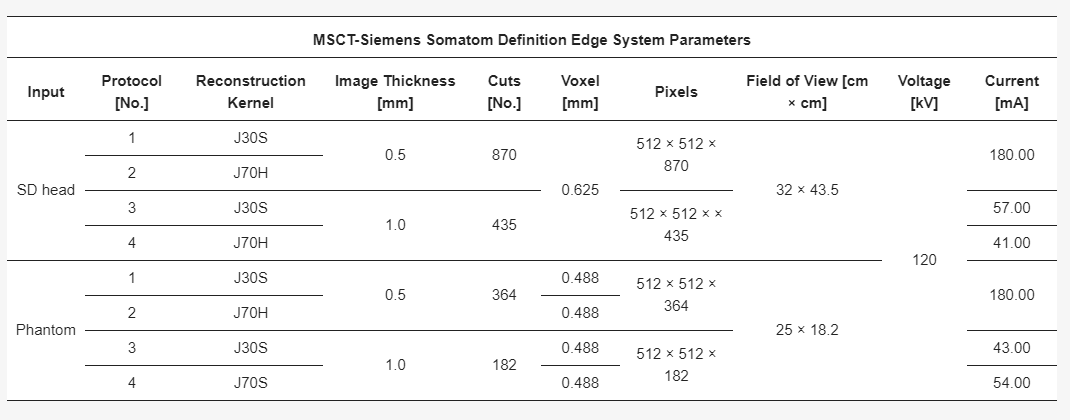
Parameters of multi-slice computed tomography (MSCT) imaging for the sus domesticus (SD) head and the Phantom.

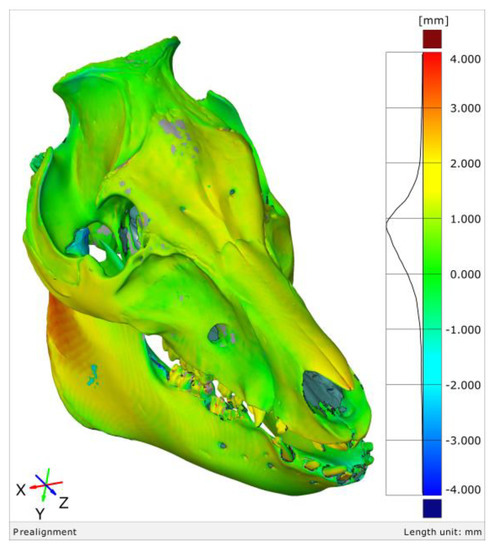
The mesh sensitivity analysis of the SD skull surface with respect to the segmented SD head surface.
“The worst-case CT imaging error emerged in the z-axis of the MSCT system and generated the second least error value compared to the other processes,” stated the researchers.
Results showed that the surface did contain a suitable mesh size, at ‘steady state values’ from the third to the sixth iterations. The thresholding error for this case showed negatively skewed asymmetric deviations to the left. The authors noted that this error was ‘one of the most significant errors compared to the other processes,’ leading them to recommend that medical professionals should be aware that the error ‘enables correction of a systematic effect for the implant.’
Such an error also propagates to the implant as it was created as a negative of the segmented sample head. Manual thresholding alternatives may be required too, and while that may take extra time, the authors suggest that such effort may yield more accurate .stl models.
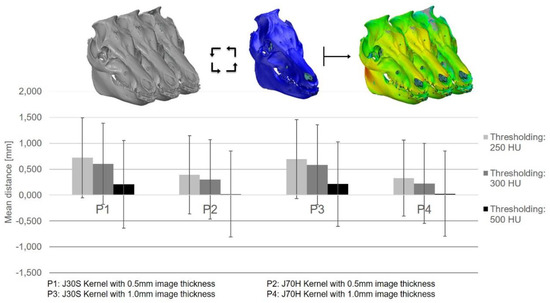
The thresholding error of the segmented SD head as a function of the CT Protocols and Hounsfield units with respect to the optically scanned dry skull (blue).
“Practitioners that require a high accuracy for the implants, e.g., the orbital floor implants, should respect the recommended safety margin. The prescribed correction measures can be taken if the intended processes and equipment are identical to this study or after implementing the applied methodology with the relevant processes and equipment as a quality control and assurance protocol,” concluded the researchers.
“Moreover, it should be considered that it may not always be absolutely imperative to design the implant to fit perfectly at the initial phase. The perforation of the lingual mucosa is one of the complications related to the patient-specific implant. If the lingual foil of the implant scaffold is too high, it is prone to perforate the lingual mucosa during the postoperative healing. For example, the extraction of teeth in the osteotomy line will lead to a resorption and further, remodeling of the marginal alveolar crest, which has to be considered during the planning of the implant.”
What do you think of this news? Let us know your thoughts! Join the discussion of this and other 3D printing topics at 3DPrintBoard.com
[Source / Images: ‘Cumulative Inaccuracies in Implementation of Additive Manufacturing Through Medical Imaging, 3D Thresholding, and 3D Modeling: A Case Study for an End-Use Implant’]Subscribe to Our Email Newsletter
Stay up-to-date on all the latest news from the 3D printing industry and receive information and offers from third party vendors.
Print Services
Upload your 3D Models and get them printed quickly and efficiently.
You May Also Like
Creality Marks 12 Years with KliTek and AI-Powered Ecosystem Expansion
For 12 years, Creality has advanced accessible 3D printing technologies, enabling global users to turn ideas into tangible creations. What began as a desktop 3D printer manufacturer has evolved into...
Asia AM Watch: Creality Launches $177M Hong Kong IPO as HKEX Trading Debut Nears
Creality has formally launched its Hong Kong initial public offering (IPO), marking one of the biggest public market moves by a 3D printing company in 2026 and offering a new...
How AtomForm’s 12-Nozzle System Cuts Multi-Color FDM Transition Waste by Up to 90%
Pull up the print stats on multi-color FDM jobs. The number that stings isn’t time; it’s material efficiency. On six-color models, single-nozzle systems consume significant filament during transition flushing, expelling...
3D Printed Weapons Keep Showing Up in Crime
In the past few weeks, activity around 3D printed weapons in the U.S. has increased across several fronts. States including California, Colorado, New York, and Washington are moving forward with...