Pediatric Surgeon 3D Prints Aortic Valve Repair Testing Device
Nations around the world are taking steps to prevent heart disease, with both advice and legislation to promote healthy eating habits, annual check-ups, and even antismoking laws and regulations. But for some patients, it is not about a change in lifestyle, since heart ailments are part of their daily life. For children born with congenital heart disease (CHD), a birth defect in the heart that can be the cause of serious health complications later on in life, pediatric surgeons around the globe are trying to raise awareness about this most common birth defect. According to estimates, one in 1,000 babies is born with the condition every year. This means an annual 1.3 million CHD births. It usually entails an anomaly in the heart walls, valves or blood vessels, ranging in effect from simple defects to life-threatening conditions. In Argentina, pediatric cardiovascular surgeon Ignacio Berra has been searching for solutions to CHD his entire adult life and last year he designed and perfected an intraoperative test device for aortic valve repair using 3D printing.
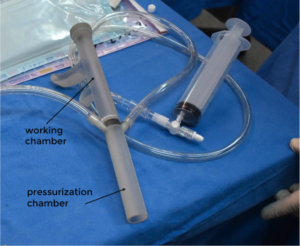
Since 2006, Berra has worked at Garrahan Hospital, in Buenos Aires, but in 2015, he won a scholarship to train together with other researchers at Boston Children’s Hospital and Harvard’s School of Medicine, where he began working on the device. The technique was presented at the Aortic Symposium held by the Association for Thoracic Surgery (AATS), and detailed how pediatric surgeons are trying to intraoperatively test and visualize the valve in its diastolic state (part of the cardiac cycle during which the heart refills with blood). The aortic valve pressurization device enables a regurgitant aortic valve to be inspected under typical diastolic conditions at the start of the repair to better understand mechanisms of aortic regurgitation (AR) as well as allowing surgeons to do a postoperatively test the efficacy of repair and the robustness of valve closure. The gadget was printed on a Stratasys Objet30 Prime 3D printer with a super clear biocompatible resin to allow for visual inspection and is described by Berra and fellow researchers as a two-piece cylindrical tube with a length of 26 cm and a working channel with inlet and outlet valves, as well as a pressurization chamber, which has two side ports. On one side they use it to test the pressure, thanks to a sensor, while the other side port can be used to introduce measurement tools; for example, an endoscope to allow visualization of the valve and aortic root.
“One of the key factors when developing this type of innovation is reproducibility. Especially in developing countries, such as Argentina, where we would benefit from generating this type of device, cheaply, for daily use in operations. Our hospitals are solely about care, so they are missing the in-house research facilities that others have in Europe and North America. This means that in order to keep working and improving the device (as well as generating other projects), I had to create my own 3D printing company, called LEW, with a fully equipped operating room for pre-clinical trials, a Stratasys Objet30 Prime, Stratasys own resins, software and industrial engineers and designers who help us develop the ideas,” explained Berra to 3DPrint.com
According to the specialist, in children, surgical repair of the valve is usually preferred over valve replacement. However, aortic valve repair is technically challenging and is currently in transition from surgical improvisation to a reproducible operation and an option for many patients with aortic valve disease. A major challenge for surgeons during aortic valve repair is to intraoperatively assess valve dysfunction before repair and to predict valve competence after repair. Up until now, there has been no similar test in use for the aortic valve, although the concept has been proposed. So the device is ideal for with children with CHD.
“The model was originally designed in the computer and then 3D printed in three parts and used at Boston Children’s Hospital to test the pressure valve. This means that, by pressurizing and using an endoscope, surgeons can see the closed aortic valve during the intraoperative period while the aorta is transacted, and it only takes one minute. To perform the preclinical trials I used pig hearts, but the method has advanced more in the US, since they are soon going to start a clinical study to formally test its validity with the FDA. In Argentina, we are only using it to test a model of the aorta prior to surgery, but we expect to begin using it in children soon,” Berra went on.
The technique validates, during the surgery, that the operation was successful and ensures that the child will be fit to continue living a normal life. Berra suggested that “without the aortic valve pressurization device it is difficult for a surgeon to intraoperatively predict valve competence after repair solely on the basis of what they see, that is, to know for sure that the valve will not yield, because if it breaks after the surgery, the turbulent blood flow can lead to endocarditis, a serious infection of the valves.” According to Berra and the other researchers behind the device, this new capability could increase the success rate of aortic valve repair, leading to higher repair rates and also encouraging less experienced surgeons to attempt it.
“Although surgeons are heavily trained, surgeries are not all that simple, each case is different, especially in children with CHD. When we have to work with patients that have such complicated pathologies, there is a need to generate new solutions with what we have on hand, and unfortunately in Argentina, availability is not in our dictionary. Doctors fight against an ancient bureaucratic system to translate an idea into a reality with the potential to save lives,” he revealed.
Berra’s ambition for research and innovation is a family trait. His father is a veterinary and researcher who instilled a desire for knowledge. Both him and his dad are part of LEW, as well as his sister, an immunologist, and brother, an engineer in charge of designing all the devices before they go through the printer. They are also working on perfecting a 3D printed artificial heart which they plan to make commercially available in the country, and which will cost much less than what an imported version costs, usually around $70,000. With this and many more projects in the works, the company could soon become one of the few generating novel ideas in the region.
[Images: Ignacio Berra and 3DPrint.com]Subscribe to Our Email Newsletter
Stay up-to-date on all the latest news from the 3D printing industry and receive information and offers from third party vendors.
Print Services
Upload your 3D Models and get them printed quickly and efficiently.
You May Also Like
Creality Marks 12 Years with KliTek and AI-Powered Ecosystem Expansion
For 12 years, Creality has advanced accessible 3D printing technologies, enabling global users to turn ideas into tangible creations. What began as a desktop 3D printer manufacturer has evolved into...
Asia AM Watch: Creality Launches $177M Hong Kong IPO as HKEX Trading Debut Nears
Creality has formally launched its Hong Kong initial public offering (IPO), marking one of the biggest public market moves by a 3D printing company in 2026 and offering a new...
How AtomForm’s 12-Nozzle System Cuts Multi-Color FDM Transition Waste by Up to 90%
Pull up the print stats on multi-color FDM jobs. The number that stings isn’t time; it’s material efficiency. On six-color models, single-nozzle systems consume significant filament during transition flushing, expelling...
3D Printed Weapons Keep Showing Up in Crime
In the past few weeks, activity around 3D printed weapons in the U.S. has increased across several fronts. States including California, Colorado, New York, and Washington are moving forward with...