
3D printed models of hearts (Photo credit: Alphr)
3D printing has taken off at lightning speed, with innovations emerging around the world continually—and virtually unregulated. While there may be some serious discussions and expectations regarding ownership and common sense regarding designs, most of the legal angles are still in the embryonic stages. And that brings us to tissue engineering. Jamil Ammar tackles a provocative subject that has the potential to become much more complex over the years, in ‘Defective Computer-Aided Design Software Liability in 3D Bioprinted Human Organ Equivalents.’
The creative aspect of 3D printing is one important part of potential intellectual property rights, but in relation to legalities, there are serious liabilities that could be connected to defects in bioprinting. Ammar leads us through the process of bioprinting, from CAD software design to CAD designs to scanning of organs, and the eventual bioprinting of such complex tissue. While there are still so many challenges to overcome before actual organs are created and implanted in humans, worrying about the legalities may seem like jumping the gun; but Ammar does bring up important issues regarding the ‘what ifs’ surrounding software or a design that could be defective.
“In the medical 3D bioprinting field, three theories are, in principle, relevant to the protection of the patient against injuries that are attributable to defective CAD software: (i) medical malpractice (a subset of negligence law), (ii) breach of warranty under the Uniform Commercial Code (UCC), and (iii) strict liability,” states Ammar. “None of these theories, however, adequately address the range of injuries that could potentially arise due to use of defective CAD software.”
In this study, Ammar that there are concerns and confusion regarding the definitions and roles of:
- Software developers
- Fabricators
- Manufacturers
- Products v. services dichotomy
The products v. services topic relates to whether computer software can be the subject of a product liability case. While courts may have not been ready to extend liability to software, Ammar reminds us that manufacturers can still be considered liable. He goes on to define OED’s as Organ Equivalent Devices which is a nifty term that does indeed remind us of the stakes.
“Product liability is a critical policy issue in the field of 3D bioprinting. It is necessary to reconsider the premise that software developers, especially in a healthcare setting, are not intrinsically subject to strict liability rules in relation to the software they provide,” states Ammar.
He also points out that the ‘extensive immunity’ afforded to the 3D printing set is not appropriate, considering the content and the potential for harm to medical patients.
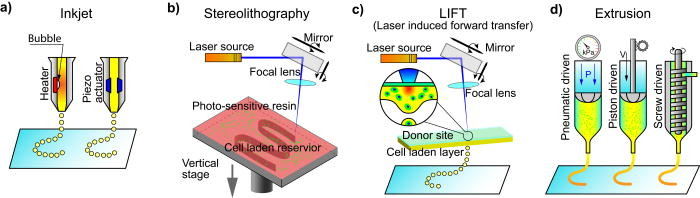
A typical bioprinting process (from ‘Extrusion bioprinting of soft materials: An emerging technique for biological model fabrication‘)
“Liability regimes currently consist of a collection of different legal systems that do not properly fit the needs of OED manufactures due to the fact that OED bioprinting combines both products and services,” states Ammar. “Healthcare professionals, medical device manufacturers, and medical software developers have, traditionally, been clearly separated; this is no longer the case, particularly when OED design and bioprinting are carried out by the same entity.”
Liability could potentially fall on medical professionals using CAD files, and developers of the files who write CAD programs but do not use personally use them. While bioprinting may not be ‘addressed’ by the FDA, obviously items that could be placed into the human body must be deemed completely safe by someone. Approval may be hard to give when bioprinted tissue has barely been categorized. Transplants are overseen by the Health Resources Services Administration (HRSA), but because OEDs are not yet specifically classified, regulations cannot be solidified.
There is also further gray area between the term developer and manufacturer:
“Given the undisputed impact of the CAD software, should the producer of the CAD print file (CAD user) be considered as the manufacturer or semi-manufacturer of the OED? The FDA defines a manufacturer as ‘any person who designs, manufactures, fabricates, assembles, or processes a finished device,” states Ammar. “The term ‘manufacturer’ includes, but is not limited to, those who perform the functions of ‘contract sterilization, installation, relabeling, remanufacturing, repacking, or specification development, and initial distributors of foreign entities performing these functions.”
Ammar asks how CAD users should then be viewed; after all, they are involved in the bioprinting process. Detecting and defining defects is another huge issue. Who is responsible for said defect follows that question, along with the reminder that hospitals are historically not in the business of distributing ‘products,’ so who is liable when things go terribly wrong for the patient with a bioprinted implant? Ammar points out that the hospital medical personnel are most likely going to be responsible for performing the bioprinting. While 3D printing is a disruptive technology, legal disruptions may not be what technological revolutionaries had in mind.
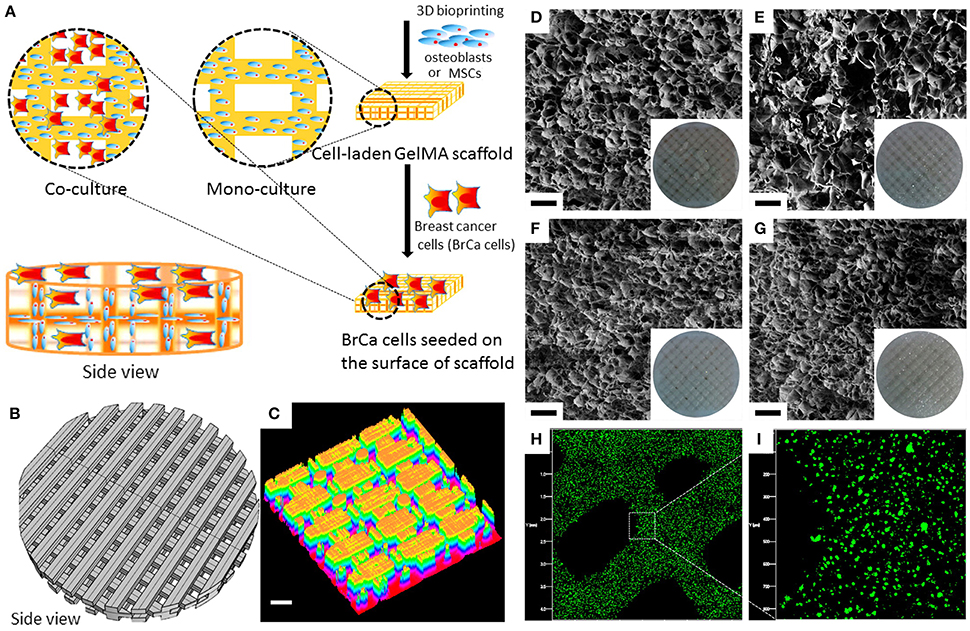
Bioprinting CAD files in breast cancer studies (Photo credit: ‘Engineering Breast Cancer Microenvironments and 3D Bioprinting’)
“Effective 3D OED bioprinting offers the promise of bridging the current shortage of donor organs, thus enhancing patient quality of care. Creating a streamlined approach to assessing the requirements of effective, reliable, and high-quality CAD software is an important first step,” says Ammar, who proposes a new approach for handling defective CAD software—by allowing consumers to launch tort action against CAD developers.
“The strict liability regime offers several advantages relative to negligence and/or malpractice regimes, which can be utilized to enhance patient safety. The proposed approach avoids the nearly impossible task of proving a breach in standard of care, allowing stakeholders to benefit from clearer and lower evidentiary standards,” concludes Ammar. “Equally significant, the cost factor of our proposal does not, in principle, lead to an overreliance on technology, which would risk defective outcomes or a reduction in the creation of would-be optimal solutions.”
Legal concerns related to 3D printing have not only advanced, but they have expanded immensely in the past few years. While copyrights are an enormous concern—and have caused trouble major headaches for some users—legal implications are coming to the forefront more often. On the flip side, 3D printing is often used to help in legal situations, with 3D printed models even used in murder trials. Learn more about emerging legal details and liability in bioprinting here. What do you think of this news? Let us know your thoughts! Join the discussion of this and other 3D printing topics at 3DPrintBoard.com.
Hydrogels used in bioprinting. (Photo: Reliawire / BioPrinting 101)
Subscribe to Our Email Newsletter
Stay up-to-date on all the latest news from the 3D printing industry and receive information and offers from third party vendors.
You May Also Like
New Report: Semiconductor Industry to See $1.4B in 3D Printing Revenues by 2032
“The semiconductor sector has become the most strategically significant area of global industry.” Truer words are hard to come by when it comes to the modern world, and they are...
Will Photonic-Crystal Lasers Revolutionize 3D Printing?
Powder bed fusion (PBF) for metals and polymers predominantly utilizes lasers as the primary heat source. Some directed energy deposition (DED) technologies also employ lasers, while various vat polymerization methods...
3D Printing Unpeeled: Orbex Investment, IndoMIM and HP, Ultrasonic Waves
INDO-MIM has bought three HP Metal Jet S100 printers, operating two in India and one in Texas. This is a win for HP because the company has deep experience in...
3D Printing Webinar and Event Roundup: April 21, 2024
It’s another busy week of webinars and events, starting with Hannover Messe in Germany and continuing with Metalcasting Congress, Chinaplas, TechBlick’s Innovation Festival, and more. Stratasys continues its advanced training...































