(A) Low-magnification view of scaffold struts showing macro pore sizes of around 500 μm together with (B) an inserted high-magnification SEM image of the surface morphology of the MesoCS scaffold struts before immersion. The microstructure of (C) PL and (D) DL scaffolds after soaking in SBF for 7 days.
We often hear about 3D printed scaffolds, which provide structure to printed cells while they grow and develop, being used in the medical field, as they can help with tissue regrowth, have an effect on how cells express signaling proteins that help cancer grow, and improve regeneration. A collaborative team of researchers from the National Taiwan University Hospital, the China Medical University Hospital, and Asia University have been studying 3D printed bone scaffolds lately.
There’s a major need around the world for bone repairs, as the population continues to age and suffer from bone defects and disorders. But one of the worst is destroyed bone in the orofacial region, which could lead to loss of function and a reduced quality of life without surgical intervention to reconstruct the hard tissues using grafts and implants. Many different types of material, like calcium phosphate and calcium sulfate, are used in synthetic bone grafts, but have issues such as a lack of osteoconductivity and manipulation difficulties.
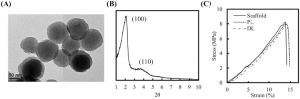
(A) TEM and (B) small-angle XRD MesoCS patterns. (C) The compressive stress–strain curve of the MesoCS scaffold, PL scaffold, and DL scaffold.
The multi-university research team recently published a paper, titled “Effects of bone morphogenic protein-2 loaded on the 3D-printed MesoCS scaffolds,” that takes a look at the effects of various loading methods on novel grafting material bone morphogenetic protein-2 (BMP-2), which was loaded with a mesoporous calcium silicate (MesoCS) scaffold created with FDM 3D printing on a 3D bioprinter from GeSiM. The team created new bone substitutes that have both osteoconductive and osteoinductive potential.
The team wrote in the paper, “In this study, there are two methods of loading BMP-2: (1) the pre-loading (PL) method by mixing MesoCS and BMP-2 as a raw material for a 3D-printer, and (2) the direct-loading (DL) method by soaking the 3D-printed MesoCS scaffold in a BMP-2 solution. The characteristics of MesoCS scaffold were examined by transmission electron microscopy (TEM), X-ray diffraction (XRD) and scanning electron microscopy (SEM). Their physical properties, biocompatibility, and osteogenic-related ability were also evaluated.”
3D printed scaffolds provide an environment quite similar to in vivo conditions for cell infiltration, and can be made with various biomaterials that help repair bone defects. Materials based in CS have excellent potential for regenerating bone when used as bioactive, biodegradable grafting materials. However, traditional CS powder is limited in drug delivery applications by its limited nanopore structure and large particle sizes, so the researchers developed a mesoporous CS nanoparticle with a diameter between 2 and 50 nm.
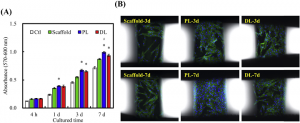
(A) Adhesion and proliferation of WJMSCs cultured on various scaffolds. “*” statically significant difference (p < 0.05) from Scaffold. “#” statically significant difference (p < 0.05) from DL group. (B) The fluorescence images of nuclei (blue) and cytoskeleton (green) in WJMSCs cultured on the MesoCS scaffold control, PL, and DL groups for 3 and 7 days.
“The 3D MesoCS/PCL scaffolds showed excellent biocompatibility and physical properties. After soaking in simulated body fluid, the bone-like apatite layer of the PL and DL groups could be formed. In addition, the DL group released fifty percent more than the PL group at the end of the first day and PL showed a sustained release profile after 2 weeks,” the researchers wrote in the paper.
“The 3D MesoCS/PCL porous scaffolds were successfully fabricated via a 3D printing system and were tested in vitro and were found to show good cellular activity for cell behavior although the PL method was not favorable for clinical application in relation with the preservation of BMP-2. With regards to different growth factor loading methods, this study demonstrated that PL of BMP-2 into MesoCS prior to printing will result in a more sustained drug release pattern as compared to traditional methods of scaffolds directly immersed with BMP-2.”
The study shows that by pre-loading BMP-2 into MesoCS before 3D printing, with regards to differing methods of loading growth factors, there will be a more sustained drug release pattern than traditionally manufactured scaffolds immersed directly with BMP-2.
Co-authors of the paper are Kuo-Hao Huang, Yen-Hong Lin, Ming-You Shie, and Chun-Pin Lin.
Discuss this story and other 3D printing topics at 3DPrintBoard.com or share your thoughts in the Facebook comments below.
Subscribe to Our Email Newsletter
Stay up-to-date on all the latest news from the 3D printing industry and receive information and offers from third party vendors.
You May Also Like
World’s Largest Polymer 3D Printer Unveiled by UMaine: Houses, Tools, Boats to Come
The University of Maine has once again broken its own record by unveiling the largest polymer 3D printer in the world. Surpassing its 2019 achievement, the new Factory of the...
Changing the Landscape: 1Print Co-Founder Adam Friedman on His Unique Approach to 3D Printed Construction
Additive construction (AC) is much more versatile than it seems, at first: as natural as it is to focus on the exciting prospect of automated home construction, there’s far more...
Gorilla Sports GE’s First 3D Printed Titanium Cast
How do you help a gorilla with a broken arm? Sounds like the start of a bad joke a zookeeper might tell, but it’s an actual dilemma recently faced by...
3D Printing Webinar and Event Roundup: April 21, 2024
It’s another busy week of webinars and events, starting with Hannover Messe in Germany and continuing with Metalcasting Congress, Chinaplas, TechBlick’s Innovation Festival, and more. Stratasys continues its advanced training...