 One year ago, we heard about the work of researchers with Rice University, who used an OpenSLS 3D printer to develop 3D printed lattices for bone tissue engineering. The researchers began their work in late 2015, and have been hard at work studying 3D printed bone tissue scaffolding. Rice bioengineer Antonios Mikos is leading the research team, and they’ve enhanced their original 3D printed scaffold, in an effort to make it more sophisticated, so they can better study how Ewing’s sarcoma (ES, a pediatric bone cancer) cells respond to stimuli, such as shear stress (the force that tumors experience when viscous fluids flow through the bone). These scientists have concluded that a scaffold structure, whether it’s natural or not, does in fact have an effect on how “cells express signaling proteins that help cancer grow.”
One year ago, we heard about the work of researchers with Rice University, who used an OpenSLS 3D printer to develop 3D printed lattices for bone tissue engineering. The researchers began their work in late 2015, and have been hard at work studying 3D printed bone tissue scaffolding. Rice bioengineer Antonios Mikos is leading the research team, and they’ve enhanced their original 3D printed scaffold, in an effort to make it more sophisticated, so they can better study how Ewing’s sarcoma (ES, a pediatric bone cancer) cells respond to stimuli, such as shear stress (the force that tumors experience when viscous fluids flow through the bone). These scientists have concluded that a scaffold structure, whether it’s natural or not, does in fact have an effect on how “cells express signaling proteins that help cancer grow.”
This means that if 3D printed materials can be designed to better determine how tumors generate, then it may become easier to test treatments for bone cancer. Scaffold porosity, which is the percent of empty space created by pores in a structure, along with the size and shape of pores, can change the permeability of nutrients and media, impact cell attachment, and even facilitate cell migration. According to the Rice University scientists, 3D printing lets them get closer than ever before to impersonating the architecture of actual bone. This research was recently published in a paper, titled “Effects of shear stress gradients on Ewing sarcoma cells using 3D printed scaffolds and flow perfusion” in the American Chemical Society journal ACS Biomaterials Science and Engineering.
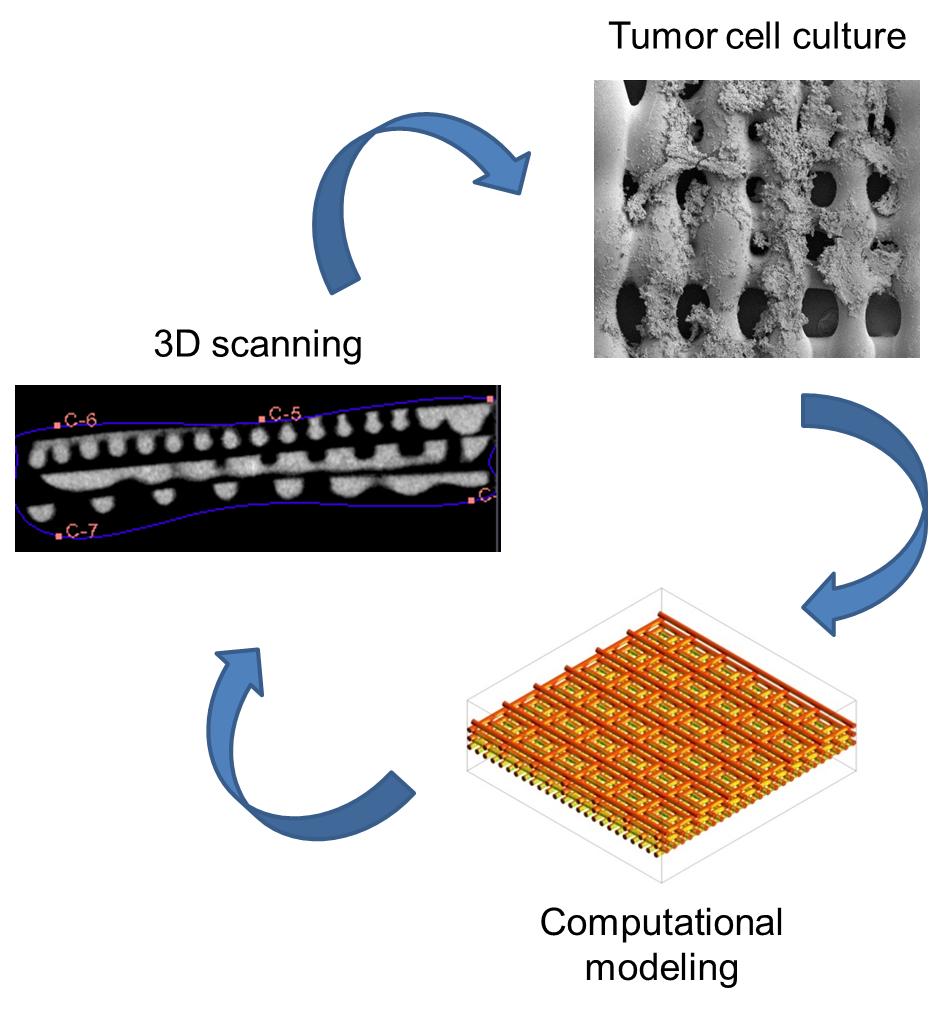
Rice researchers combined experiments with computational modeling to find that the structure of a scaffold, natural or not, has a very real effect on how cells express signaling proteins that help cancer grow. [Courtesy of the Mikos Research Group]
According to the research paper’s abstract, “As cancer biologists elucidate the relevance of 3D in vitro tumor models within the drug discovery pipeline, it has become more compelling to model the tumor microenvironment and its impact on tumor cells. In particular, permeability gradients within solid tumors are inherently complex and difficult to accurately model in vitro. However, 3D printing can be used to design scaffolds with complex architecture, and flow perfusion can simulate mechanical stimulation within the tumor microenvironment. By modeling these gradients in vitro with 3D printed scaffolds and flow perfusion, we can identify potential diffusional limitations of drug delivery within a tumor.”
According to Mikos, what’s really special is the scaffold itself: the 3D printed, bone-like polymer has different sizes of pores to “constrain fluids” that flow through, which, depending on the orientation of the scaffold, will apply different degrees of shear stress to the actual tumor cells.
Mikos said, “We aim to develop tumor models that can capture the complexity of tumors in vitro and can be used for drug testing, thus providing a platform for drug development while reducing the associated cost.”
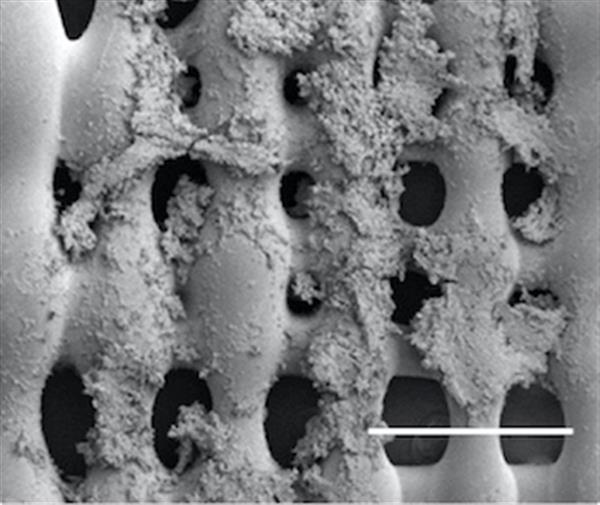
Sarcoma (bone cancer) cells proliferate on the surface of a 3-D printed scaffold created at Rice University. Experiments at Rice showed that the size of pores in the scaffold, which mimics the extracellular matrix in bone, and the pores’ orientation make a difference in how cells proliferate in the presence of a flowing fluid, like blood. [Courtesy of the Mikos Research Group]
The scaffolding was 3D printed in flat sections, with pores measuring 0.2 mm, 0.6 mm, and 1.0 mm. Each 3D scaffold, seeded with tumor cells, was built by stacking three layers on top of each other. In order to make the simulations more realistic than cells growing in a regular petri dish, the scaffolds were put into a flow perfusion reactor, which mimics a biological environment by the push and pull of both tissues and fluid. The researchers discovered that the cells placed in conditions with zero fluid flow proliferated worse than the cells under flow.
The layers of scaffolding with the smallest pores showed much more proliferation once the viscous fluid began flowing; the smallest pores restrict permeability, so it seems to me, and I am not a scientist, like the cells would just bunch up together instead of going through the pores. The scientists also discovered that the cells under fluid flow had an increase in production of an insulin-like growth factor protetin, or IGF-1, which is a “ligand on the surface of sarcoma cells and part of the signaling pathway that plays a critical role in resistance to chemotherapy.” The orientation of the three different pore sizes was also a factor in how much IGF-1 was produced by the cells.
The conclusion of the paper’s abstract reads, “Our results highlight how 3D printed scaffolds, in combination with flow perfusion in vitro, can effectively model aspects of solid tumor heterogeneity for future drug testing and customized patient therapies.”

2015 Image: Rice graduate student Marco Santoro led the study to test the spread of bone cancer cells in realistic environments. [Photo: Jeff Fitlow]
The other co-authors of the paper are:
- Rice University: graduate students Brandon Smith and Eric Molina
- University of Oklahoma: graduate student Cortes Williams and Professor Vassilios Sikavitsas
- University of Maryland: graduate student Charlotte Piard and Fischell Family Distinguished Professor John Fisher
- University of California, San Diego: postdoctoral researcher Jesse Placone
- University of Texas MD Anderson Cancer Center: research assistant Brian Menegaz, senior research scientist Salah-Eddine Lamhamedi-Cherradi, and Associate Professor Joseph Ludwig
The Howard Hughes Medical Institute, the National Science Foundation, the National Institutes of Health, and the Armed Forces Institute of Regenerative Medicine all supported this research. Discuss in the Rice University forum at 3DPB.com.
[Source: Rice University]Subscribe to Our Email Newsletter
Stay up-to-date on all the latest news from the 3D printing industry and receive information and offers from third party vendors.
Print Services
Upload your 3D Models and get them printed quickly and efficiently.
You May Also Like
HP Continues to Lower Barriers to Adoption with Compact MJF 1200 & Other RAPID + TCT Announcements
This week at RAPID+TCT in Boston, HP Additive Manufacturing Solutions is celebrating ten years in the AM market. The company launched its Multi Jet Fusion 3D printing technology in Barcelona...
3D Printing News Briefs, April 11, 2026: Energy Targets, DoW Contracts, Nike Air Max, & More
We’re starting with 3D printing for energy applications in this weekend’s 3D Printing News Briefs, and then moving on to military and defense 3D printing. Finally, Nike Sportswear is focusing...
BigRep Launches ONE.5X 3D Printer, Announces New Massive Dimension Partnership at RAPID + TCT 2026
As the whole world is starting to realize, the Hormuz supply chain fallout is only just beginning to filter into the global economy, and the rising cost of plastics should...
Portsmouth Naval Shipyard Installs 3D Printed Part on In-Service Submarine in Key Milestone for US Defense Sector
I’ve been tracking the US Navy’s additive manufacturing (AM) buildup as it relates to submarines for a while now, and even amidst the AM efforts that all the branches have embarked...































