Annual SOT Meeting in NOLA: Presentations Offer Validity for Using Organovo’s exVive 3D Printed Human Liver Tissue in Research
 This is an exciting week for many in New Orleans. While one could say that at virtually any time—and residents would probably chuckle heartily at the comment—if you are interested in the world of bioprinting, this should make sense to you from a scientific standpoint, as indeed there were some eye-opening presentations today being given at the Society of Toxicology’s (SOT) 55th Annual Meeting and ToxExpo, ongoing from March 13-17 in NOLA.
This is an exciting week for many in New Orleans. While one could say that at virtually any time—and residents would probably chuckle heartily at the comment—if you are interested in the world of bioprinting, this should make sense to you from a scientific standpoint, as indeed there were some eye-opening presentations today being given at the Society of Toxicology’s (SOT) 55th Annual Meeting and ToxExpo, ongoing from March 13-17 in NOLA.
We gave you a slight preview last week regarding the event and what would be offered via Organovo and their presenters, with the main focus on their first major product release, exVive 3D Human Liver Tissue, which is the subject of several presentations we can now fill you in on.
 Each discussion is greatly detailed, and meant to offer a variety of ways to explain how broadly applicable the exVive 3D Human Liver Models are in terms of processes like assessing drug safety and being able to detect liver disease and injuries. Another main theme—and bonus of this technology—is that Organovo scientists see animal testing as being much closer to elimination due to (thankfully) its inferiority in comparison to the precision that models by Organovo can offer.
Each discussion is greatly detailed, and meant to offer a variety of ways to explain how broadly applicable the exVive 3D Human Liver Models are in terms of processes like assessing drug safety and being able to detect liver disease and injuries. Another main theme—and bonus of this technology—is that Organovo scientists see animal testing as being much closer to elimination due to (thankfully) its inferiority in comparison to the precision that models by Organovo can offer.
All of the following data, presented today at SOT, gives evidence which greatly supports using exVive3D Human Liver Model.
Title: Functional Evaluation of Bioprinted Human Liver Organoid as a Liver Injury Model
Presenter: Kazuhiro Tetsuka, Ph.D., Astellas Pharma Inc.
Poster: 2001: Poster Board – P405
This presentation discussed the implications of drug-induced liver injuries and how they can result in drugs having to be withdrawn from the market. Here, the speaker focused on acetaminophen and how both its long- and short-term effects can be characterized in a 3D-bioprinted model of the human liver, and how it can be used to both predict and evaluate true toxicity of chemicals. With bioprinting, the cell types are able to be aligned and controlled spatially.
Using a Novogen Bioprinter, human liver organoids (HLOs) were made from:
- Human hepatocytes
- Stellate cells
- Endothelial cells of the umbilical vein
“To measure the long-term effects of APAP, 3D-HLOs underwent treatment with APAP at 0, 0.3, 1, 3, 10, or 30 mM for 1, 7, 14, or 28 days. To measure the short-term effects of APAP, 3D-HLOs underwent treatment with APAP at 30 mM for 6 h, followed by either termination of culture or further culture without APAP for 1, 3, or 18 h as a washout stage,” states the abstract for the presentation.
Examination of all details did show that 3D HLOs in such a culture are biologically viable, as well as offering the conclusion that the 3D HLO is valid for the evaluation of ‘hepatotoxicity caused by long-term compound treatment, as well as liver recovery following the removal of chemicals.’
Title: Modeling Drug Induced Hepatic Fibrosis In Vitro Using Three-Dimensional Liver Tissue Constructs
Presenter: Leah M. Norona, Doctoral Candidate, The University of North Carolina at Chapel Hill and The Hamner Institutes
Poster: 1996: Poster Board – P348
This presentation discussed what a challenge human toxicity risk assessment is in terms of looking at compound-induced hepatotoxicity that leads to fibrosis. It’s difficult to characterize the intercellular interactions that happen as the liver is progressively injured, and although animal models can be used to assist here somewhat, the researchers see that human tissue-like model systems must be used to see sufficiently the central events which lead up to fibrogenesis. Using bioprinted tissue, the researchers were able to repeat modeling of the liver injury.
“Significant time dependent elevations of LDH were observed for both TAA (by Day 3) and MTX (by Day 11) and were accompanied by the acquisition of a fibrogenic phenotype as supported by preliminary cytokine, gene expression data, and histologic evidence of collagen deposition,” states the abstract for the presentation.
“In comparison, treatment with TGF-β1, a known pro-fibrogenic cytokine, yielded moderate to severe fibrotic change in the tissue with little evidence of hepatocellular damage. These preliminary data provide strong proof-of-concept that 3D bioprinted liver tissues can recapitulate drug-, chemical- and TGF-β1-induced fibrogenesis on a cellular, molecular, and histological basis.”
It’s thought that with these new strategies, researchers will be able to able to perform better chemical risk assessment as they have an improved way to see the key attributes initially causing and allowing liver injury to progress.
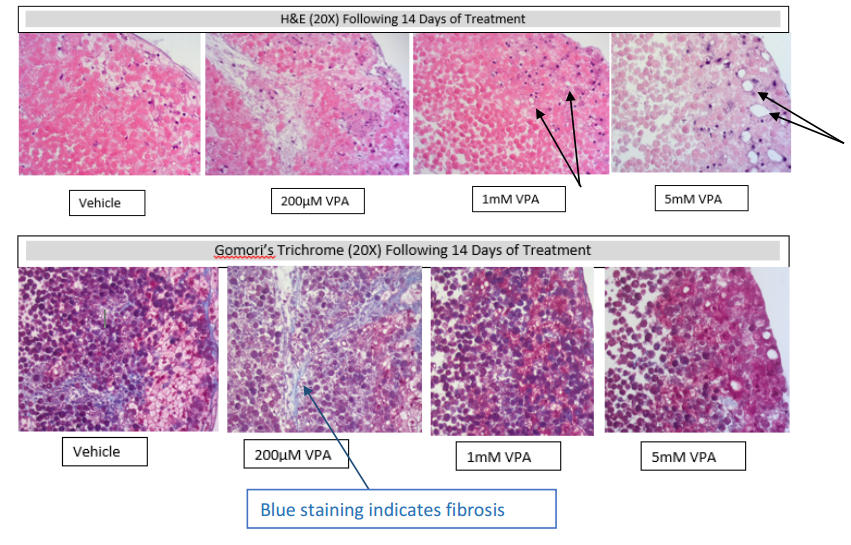
 Title: Utilization of exVive3D Human Liver Tissues for the Evaluation of Valproic Acid Induced Liver Injury
Title: Utilization of exVive3D Human Liver Tissues for the Evaluation of Valproic Acid Induced Liver Injury
Presenter: Candace Grundy, Organovo
Poster: 2003: Poster Board – P407
This presentation discussed that often 2D cell cultures are not able to display the true and complex ‘microenvironment’ of liver tissue, often resulting in inadequate animal testing trials. Because 3D bioprinted human liver tissues offer a much better reflection of the tissue construction, researchers are able to assess drug-induced liver injury (DILI) much better, as well as outcomes from a biochemical and histologic standpoint.
For the study, they looked at the DILI response to a compound called Valproic Acid (VPA). This is known to cause steatosis. The researchers treated the 3D-bioprinted human liver tissues for 14 days with VPA.
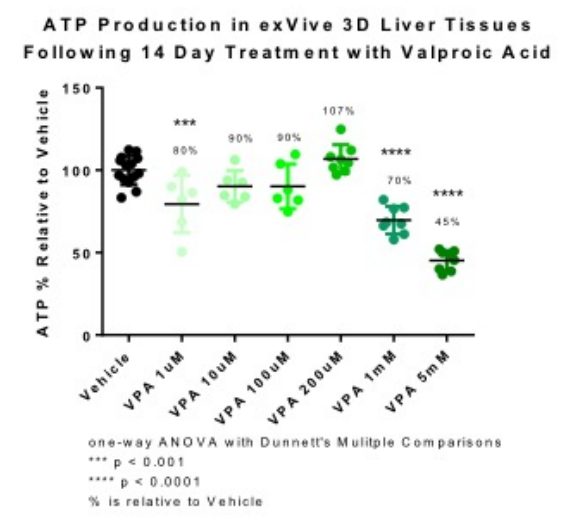
“Tissue ATP levels were decreased 70% and 45% relative to vehicle following 14 day treatment with 1mM and 5mM VPA, respectively. Following the observations seen with ATP levels, the higher doses of 200µm, 1mM and 5mM were selected to further evaluate the mechanism of observed tissue damage,” stated the abstract. “GSH levels were measured at 24hr and 72hr to determine the acute oxidative stress response.”
As they performed histological evaluation of the tissues, researchers were able to see that indeed there was ‘dose-dependent damage’ that they considered to be consistent with steatosis. As a result, they were able to conclude that the exVive 3D human liver tissues can be used to examine DILI mechanisms ‘in vitro,’ proven by the results seen in ‘biochemical and histologic endpoints following treatment with VPA.’
Title: Inflammatory Response of Kupffer Cells in 3D Bioprinted Human Liver Tissues
Presenter: Rhiannon N. Hardwick, Ph.D., Organovo
Poster: 1959: Poster Board – P311
This presentation discussed how hepatocellular damage is made worse by inflammation ‘mediated by Kuppffer cells.’ These cells are often used, according to the researchers, in cultures with hepatocytes to examine how inflammation occurs due to stimuli.
“In the current study, LPS-mediated activation of KC at 24 and 72hrs was investigated in 3D bioprinted human liver tissues. Induction of pro- and anti-inflammatory cytokines was measured via electrochemiluminescence in tissues comprising primary hepatocytes, stellate cells, and endothelial cells (Hep:KC-), and compared to tissues containing KC,” states the abstract.
As the experiment progressed, variables and current data were able to measure ‘a robust donor-specific KC response to inflammatory stimuli, thus enabling investigation of immune-mediated drug induced liver injury in a 3D human liver tissue.
Title: Monocrotaline Toxicity in 3D Bioprinted Human Liver Tissue
Presenter: Umesh M. Hanumegowda, MVSc Ph.D. DABT, Bristol-Myers Squibb
Poster: 3562: Poster Board – P254
This presentation discussed an alkaloid, pyrrolizidine, that not only causes liver injury in animals, but also causes a ‘hepatic venoocclusive disorder’ in humans. Liver injury occurs after a series of ‘cellular insults,’ which cause fibrotic changes.
For this study, the researchers examined the effects of MCT in 3D-bioprinted human liver tissue comprising of primary hepatocytes, hepatic stellate cells, and endothelial cells. As they treated the bioprinted tissues with MCT at different concentrations, they noticed increases in soluble LDH, as well as decreases in albumin production as early as three days in. Testing led to a number of observations regarding early tissue damage and fibrotic events.
“These observations capture the spectrum of changes induced by MCT ranging from reduced hepatocellular function and vascular remodeling, which may involve endothelial cell migration, organization, proliferation, apoptosis, and endothelial-tomesenchymal transformation to early fibrotic events,” states the abstract.
You can find out more about Organovo and their use of 3D human tissue in research and therapeutic applications here. What do you think of the topics presented? Discuss in the Organovo 3D Human Liver Tissue forum over at 3DPB.com.
Subscribe to Our Email Newsletter
Stay up-to-date on all the latest news from the 3D printing industry and receive information and offers from third party vendors.
You May Also Like
3D Printing Financials: Fathom Struggles in Financial Quicksand During Critical Transition
Facing a year of key transitions and financial pressures, Fathom (Nasdaq: FTHM) has filed its annual report for 2023 with the U.S. Securities and Exchange Commission (SEC). The document outlines...
Latest Earnings Overview for Australian 3D Printing Firms Titomic and AML3D
Australian 3D printing manufacturing firms Titomic (ASX: TTT) and AML3D (ASX: AL3) reported their financial results for the period from July to December 2023, marking the first half of their...
3D Printing Webinar and Event Roundup: April 7, 2024
Webinars and events in the 3D printing industry are picking back up this week! Sea-Air-Space is coming to Maryland, and SAE International is sponsoring a 3D Systems webinar about 3D...
3D Printing Financials: Unpacking Farsoon and BLT’s 2023 Performance
In the Chinese 3D printing industry, two companies, Farsoon (SHA: 688433) and Bright Laser Technologies, or BLT (SHA: 688333), have recently unveiled their full-year earnings for 2023. Farsoon reported increases...