 3D printing is big, but bioprinting has quickly developed into something substantially more; in fact, it’s become an enormous–and fascinating–realm of its own, which should give us all cause for excitement and optimism. And leading the way is Organovo, with a team that can be proud to know they are well on their way to effecting enormous change in medical research.
3D printing is big, but bioprinting has quickly developed into something substantially more; in fact, it’s become an enormous–and fascinating–realm of its own, which should give us all cause for excitement and optimism. And leading the way is Organovo, with a team that can be proud to know they are well on their way to effecting enormous change in medical research.
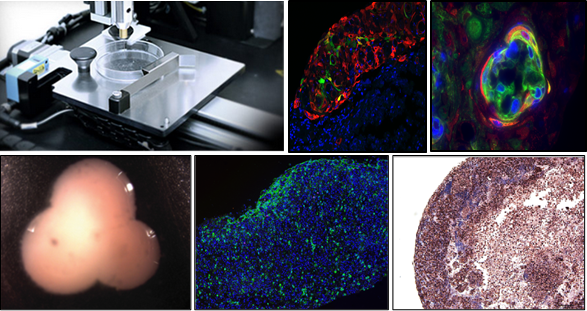
 Based in San Diego, the 3D biology company is quickly being recognized as one of the top innovators in the scientific and health fields, as they create functional human tissue with their new technology. While breakthroughs lately in bioprinted kidney tissue have had them in the news (this exVive3D product is scheduled for release in the third quarter), earning impressive acclaim and attention, their first product, exVive 3D Human Liver Tissue, is the subject of five presentations at the Society of Toxicology’s (SOT) 55th Annual Meeting and ToxExpo, from March 13–17 in New Orleans.
Based in San Diego, the 3D biology company is quickly being recognized as one of the top innovators in the scientific and health fields, as they create functional human tissue with their new technology. While breakthroughs lately in bioprinted kidney tissue have had them in the news (this exVive3D product is scheduled for release in the third quarter), earning impressive acclaim and attention, their first product, exVive 3D Human Liver Tissue, is the subject of five presentations at the Society of Toxicology’s (SOT) 55th Annual Meeting and ToxExpo, from March 13–17 in New Orleans.
The detailed presentations will each work to educate and explain how the exVive 3D Human Liver Models allow for ‘broad applicability’ in allowing scientists and medical professionals to assess drug safety , as well as being able to detect a range of liver injuries, to include steatosis, or ‘fatty liver’ and fibrosis, which is the initial states of liver scarring which can lead to cirrhosis.
“Organovo’s exVive3D Human Liver Model provides an accurate, predictive and reproducible model of human liver biology for preclinical toxicity testing,” said Paul Gallant, general manager, Organovo. “At the SOT Annual Meeting, we and our pharmaceutical customers will be highlighting recent results that show our 3D bioprinted human liver tissue effectively models in vivo tissue composition and physiology.”
One major theme running through their presentations also is that with these new models, the need for testing on animals may be closer to elimination—and especially as the results are inferior to what Organovo’s models can offer, ultimately.
“Drug-induced liver injury remains a major cause of late-stage clinical failures and market withdrawal, often due to poor translation from preclinical animal studies to clinical outcomes,” said Dr. Sharon Presnell, chief technology officer and executive vice president of research & development, Organovo.
“Organovo’s exVive3D human liver model replicates complex cell-cell interactions and key elements of native tissue architecture to enable the detection of multiple clinically-relevant modes of tissue injury, including necrosis, immune-mediated tissue damage, steatosis, and fibrosis. When a preclinical or clinical-stage asset presents a challenging safety or efficacy signal, exVive3D provides the unique resolving power of a controlled human tissue microenvironment to investigate mechanism and develop solutions.”
 Presented data will give evidence which supports using exVive3D Human Liver Model in:
Presented data will give evidence which supports using exVive3D Human Liver Model in:
- Investigating in vitro mechanisms of drug-induced liver injury with biochemical and histologic endpoints
- Evaluating liver toxicity caused by long term compound treatment, as well as liver recovery after drug removal
- Capturing the spectrum of drug-induced changes at the tissue level, including reduced liver function and vascular remodeling
- Demonstrating drug, chemical, and TGF-β1 induced liver fibrosis at the cellular, molecular, and histologic level
- Interrogating and identifying over time the key cellular and molecular events underlying fibrogenesis
- Characterizing the short and long-term effects of acetaminophen
- Examining immune-mediated, drug-induced liver injury
Organovo will offer an exhibitor-hosted session on March 15, at 9:00 a.m. Central Time (CT), in Room 212 to include oral presentations on recent advances using bioprinted 3D human liver tissues to assess drug-induced liver toxicity. It will be presented by Sharon Collins Presnell, Ph.D., of Organovo, and Umesh M. Hanumegowda, MVSc Ph.D. DABT, of Bristol-Myers Squibb.
Following that is the lineup of presentations for the day, to include:
Poster Presentations: March 15, 9:30 a.m. – 12:45 p.m. CT, at CC Exhibit Hall
Title: Functional Evaluation of Bioprinted Human Liver Organoid as a Liver Injury Model
Presenter: Kazuhiro Tetsuka, Ph.D., Astellas Pharma Inc.
Poster: 2001: Poster Board – P405
Title: Modeling Drug Induced Hepatic Fibrosis In Vitro Using Three-Dimensional Liver Tissue Constructs
Presenter: Leah M. Norona, Doctoral Candidate, The University of North Carolina at Chapel Hill and The Hamner Institutes
Poster: 1996: Poster Board – P348
Title: Utilization of exVive3D Human Liver Tissues for the Evaluation of Valproic Acid Induced Liver Injury
Presenter: Candace Grundy, Organovo
Poster: 2003: Poster Board – P407
Title: Inflammatory Response of Kupffer Cells in 3D Bioprinted Human Liver Tissues
Presenter: Rhiannon N. Hardwick, Ph.D., Organovo
Poster: 1959: Poster Board – P311
Late-breaking Poster Presentation: March 17, 9:30 a.m. – 12:45 p.m. CT, Great Hall A
Title: Monocrotaline Toxicity in 3D Bioprinted Human Liver Tissue
Presenter: Umesh M. Hanumegowda, MVSc Ph.D. DABT, Bristol-Myers Squibb
Poster: 3562: Poster Board – P254
The Organovo Exhibit Booth can also be visited from March 14-16, from 9:15 a.m. – 4:30 p.m. CT, in Booth 1701, at CC Exhibit Hall.
We will be releasing more information regarding the content of the presentations next week, so stay tuned to find out more about what the Organovo team and their group of presenters had to say. Their latest developments are indeed important for everyone to learn about as with their innovations they will be able to accelerate drug discovery processes, allow for faster and more affordable treatment, as well as improving toxicology procedures and preclinical drug testing. They also have additional products in development.
You can find out more about this company and their use of 3D human tissue in research and therapeutic applications here. Discuss in the exVive3D Human Liver Model Presentations at SOT forum over in 3DPB.com.
Subscribe to Our Email Newsletter
Stay up-to-date on all the latest news from the 3D printing industry and receive information and offers from third party vendors.
Print Services
Upload your 3D Models and get them printed quickly and efficiently.
You May Also Like
Asia AM Watch: China’s 5 Million-Printer Export Year Signals Desktop AM at Scale
For years, a lot of the discussion around China and additive manufacturing has focused on industrial competition. Can Chinese companies move into higher-end markets? Can they challenge Western machine makers...
Creality Launches Filament Maker M1 & Shredder R1, Letting Makers Reuse Waste, Cut Costs, and Create Their Own Filament
From Printing Objects to Shaping Materials Desktop 3D printing has made on-demand creation more accessible than ever. Yet one critical part of the process remains fixed: the material itself. Most...
Bambu Lab 3D Prints Miniature Playground City for Kids in China
Bambu Lab has partnered with meland to open what they describe as China’s first 3D printing creativity center for children. The new space, officially named “meland x Bambu Lab,” launched...
Bambu Lab Says 2025 Was a Breakout Year: 10 Million Monthly Users and Real Business Growth
Chinese 3D printer maker Bambu Lab reported strong results for 2025, showing that the company’s push into community and small-business 3D printing is working. The numbers suggest consumer 3D printing...
































