It appears as though 3D printing is finally being taken seriously from a medical perspective. Over the last two months we have seen a flurry of activity when it comes to 3D printed medical products receiving FDA approval in the United States. Just last week the pharmaceutical company Aprecia earned FDA approval for their 3D printed ZipDose tablets of their seizure medication SPRITAM levetiracetem, and in the weeks prior to that, two other companies, joimax® and Oxford Performance Materials, earned approval for 3D printed medical implants.
It appears as though 3D printing is finally being taken seriously from a medical perspective. Over the last two months we have seen a flurry of activity when it comes to 3D printed medical products receiving FDA approval in the United States. Just last week the pharmaceutical company Aprecia earned FDA approval for their 3D printed ZipDose tablets of their seizure medication SPRITAM levetiracetem, and in the weeks prior to that, two other companies, joimax® and Oxford Performance Materials, earned approval for 3D printed medical implants.
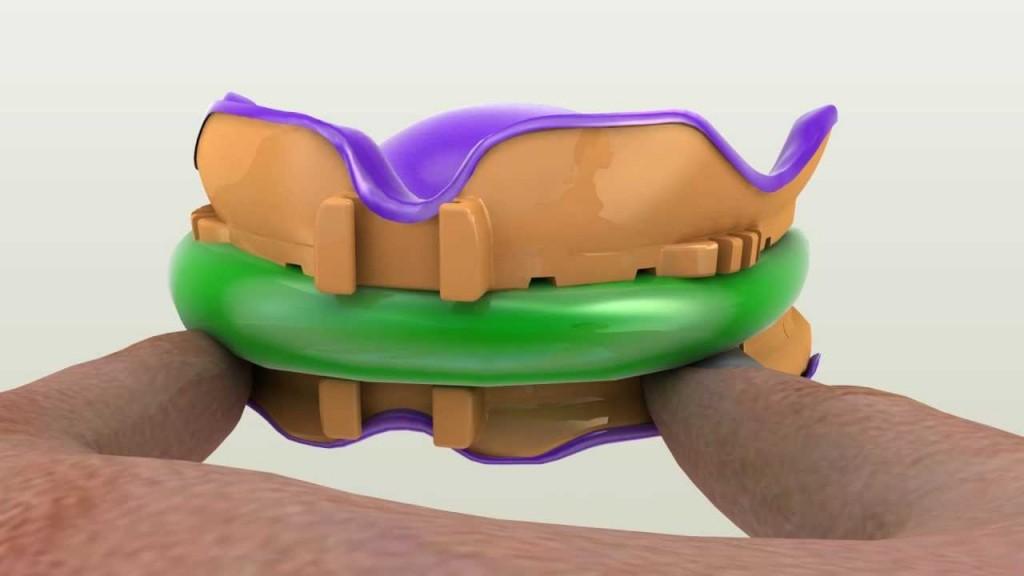
With all the hoopla surrounding the Aprecia news, another major FDA accomplishment was achieved by a small denture company called DENTCA, which has gone relatively under the radar. The Los Angeles-based company officially earned 510(k) approval for the use of their new 3D printable material as a denture base.
“After several years in development DENTCA’s new 3D printable denture base is finally cleared for use. We are very excited to begin applying this technology to continue revolutionizing the denture world,” explained Dr. Jason Lee, a creator of the DENTCA technology. “The material is a light-cured resin indicated for fabrication and repair of full and partial removable dentures and baseplates; which will eventually replace traditional heat-cured and auto polymerizing conventional denture making methods. By improving the manufacture process with the help of precise 3D printers, we will be making the denture production process quicker, more accurate and more predictable.”
The material used within an SLA 3D printing process to create the denture bases has passed all the required tests for cytotoxicity, irritation, genotoxicity, sensitization, acute toxicity and material characterization for biocompatibility in accordance with the FDA Blue Book Memorandum #G95-1 and International Standard ISO 10993-1.
“This clearance completely revolutionizes the denture manufacture process, which has barely changed in over 100 years,” explained DENTCA’s CEO Mr. Sun Kwon. “The new developments will create incredible possibilities for patients, as doctors will soon be able to 3D print final dentures at their office, allowing the manufacture process to be done in one day everywhere in the world. Stay tuned for new and exciting updates.”
The technology behind DENTCA’s products allows for the fabrication of full denture cases 2.5 times faster than traditional production methods. DENTCA’s precision and accuracy with their CAD/CAM technology, combined with 3D printing, is able to eliminate human error while also drastically cutting down on the number of visits and amount of chair-time that each patient has to invest prior to receiving their dentures. Additionally, the fabrication turnaround time was reduced from 30 days to just 5.
This is major achievement for the company, and one which could certainly play a significant role within the denture industry. Let us know your thoughts on DENTCA’s recent accomplishments in the DENTCA 3D printable Denture Base forum thread on 3DPB.com.
Subscribe to Our Email Newsletter
Stay up-to-date on all the latest news from the 3D printing industry and receive information and offers from third party vendors.
You May Also Like
3D Printing Webinar and Event Roundup: April 21, 2024
It’s another busy week of webinars and events, starting with Hannover Messe in Germany and continuing with Metalcasting Congress, Chinaplas, TechBlick’s Innovation Festival, and more. Stratasys continues its advanced training...
3D Printing Webinar and Event Roundup: April 14, 2024
We’re starting off the week’s 3D printing webinars and events at ASTM AMCOE’s 11th Snapshot Workshop and MACH Exhibition. Stratasys continues its advanced training courses, SME is holding a virtual...
Polly the Duck to Receive 3D Printed Bill Prosthetic
In Williamson County, Texas, a story of resilience, innovation, and cross-community effort is unfolding, illustrating the bond between humans and wildlife. All Things Wild, a wildlife rehabilitation center, has been...
3D Printing News Briefs, April 3, 2024: Kickstarter FDM 3D Printer, Artificial Eyes, & More
In 3D Printing News Briefs today, we’re talking about an FDM 3D printer on Kickstarter, advancements in artificial eye creation, and 3D printed solenoids for electromagnets. Then we’ll move on...