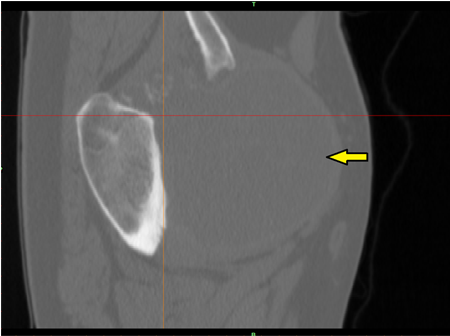
The medical field is, rightly so, a highly regulated one, and you can’t really get momentum for a product until it’s received clearance from the U.S. Food and Drug Administration (FDA). Last summer, specialized medtech company Onkos Surgical was cleared by the FDA to market its 3D printed collar implants for musculoskeletal oncology and complex orthopedic limb salvage surgery, and now the company has announced that it has earned the coveted FDA 510(k) clearance for its My3D personalized pelvic reconstruction system, which includes 3D printed implants, models, instruments, and an advanced surgical planning tool.
“This clearance is a major milestone for Onkos Surgical as we continue to leverage our experience and expertise in 3D planning and printing to optimize patient specific solutions for complex orthopaedic conditions. We founded the company with the belief that patients with these challenging conditions deserve solutions designed specifically for them,” Onkos Surgical Co-Founder and CEO Patrick Treacy stated in a press release. “Our My3D platform and Digital Ecosystem enable us not only to provide that personalized solution faster, but also to lay the foundation for future clearances in musculoskeletal personalization.”
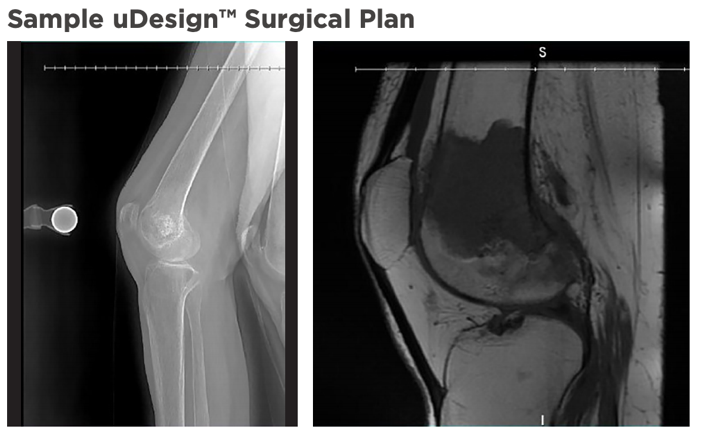
Onkos says that because of the FDA clearance, the pelvic reconstruction platform can meet surgical requirements in just six weeks. The company also claims that the My3D system is “the first of its kind” to include not only 3D printed patient-specific implants, models, and instruments, but also access to an advanced tool to use for planning purposes in the face of disease, deformity, trauma, and revisions that couldn’t be fixed by other treatments. The platform is used by surgeons to develop these personalized medical devices for surgeries.
“This offering from Onkos Surgical will greatly advance how I, and my colleagues treat these patients. Patients with these conditions of the pelvis have many clinical challenges. Historically, our implant options are mass produced and may not be best suited for the individuality that each patient requires. With this platform, Onkos has developed a process that allows me to virtually plan the surgery in advance and delivers a patient specific implant and instruments in a matter of weeks. It changes the way I can treat my patients,” said Matthew Seidel, MD, orthopedic surgeon with HonorHealth Orthopedics.
The My3D solution includes 3D printed patient-specific implants, with “unique” features, for acetabular reconstruction, as well as advanced reconstruction of several pelvic regions. The company says these features help with anatomic restoration accuracy, as well as encouraging soft tissue and bone attachment and growth, the latter of which is known as ossification.
My3D also offers access to the Onkos uDesign digital ecosystem, which “enables a personalized planning approach for each patient,” according to the website. Patient images, like CT scans, can be sent through a cloud-based, secure, HIPAA-compliant portal, and segmented by high-quality image processors in order to create 3D models of both hard and soft tissue. Then the surgeon, together with ONKOS technical experts, reviews the models in a virtual planning session to come up with the best pre- and intra-operative surgical solutions and ensure better patient outcomes.
In February of 2023, 3DPrint.com and SmarTech Analysis will once again be holding our Additive Manufacturing Strategies summit in New York City. This event will include keynote presentations and panels focused on nine vertical topics that are critical in the continually-growing world of additive manufacturing, including 3D printing for healthcare. Register now to join us in these important discussions!
(Images via Onkos Surgical)
Subscribe to Our Email Newsletter
Stay up-to-date on all the latest news from the 3D printing industry and receive information and offers from third party vendors.
You May Also Like
3D Printing Financials: Fathom Struggles in Financial Quicksand During Critical Transition
Facing a year of key transitions and financial pressures, Fathom (Nasdaq: FTHM) has filed its annual report for 2023 with the U.S. Securities and Exchange Commission (SEC). The document outlines...
Latest Earnings Overview for Australian 3D Printing Firms Titomic and AML3D
Australian 3D printing manufacturing firms Titomic (ASX: TTT) and AML3D (ASX: AL3) reported their financial results for the period from July to December 2023, marking the first half of their...
3D Printing Webinar and Event Roundup: April 7, 2024
Webinars and events in the 3D printing industry are picking back up this week! Sea-Air-Space is coming to Maryland, and SAE International is sponsoring a 3D Systems webinar about 3D...
3D Printing Financials: Unpacking Farsoon and BLT’s 2023 Performance
In the Chinese 3D printing industry, two companies, Farsoon (SHA: 688433) and Bright Laser Technologies, or BLT (SHA: 688333), have recently unveiled their full-year earnings for 2023. Farsoon reported increases...