ICAM 2021: Keynotes on 3D Printing in Healthcare & Aerospace
At last month’s International Conference on Additive Manufacturing (ICAM) 2021 in Anaheim, California, hosted by ASTM International’s Additive Manufacturing Center of Excellence (AMCOE), a wide variety of topics were covered, including feedstock challenges, 3D printing for defense applications, economics and sustainability considerations in AM, application of 3D printing in construction, and much more.
On the second day, Sam Onukuri, Head and Sr Fellow of 3D Printing Innovation at Johnson & Johnson, presented a keynote on Healthcare Transformation Enabled by 3D Printing – a Johnson & Johnson Case Study. As the event was a hybrid one, Onukuri virtually gave his keynote on the world’s largest healthcare company.
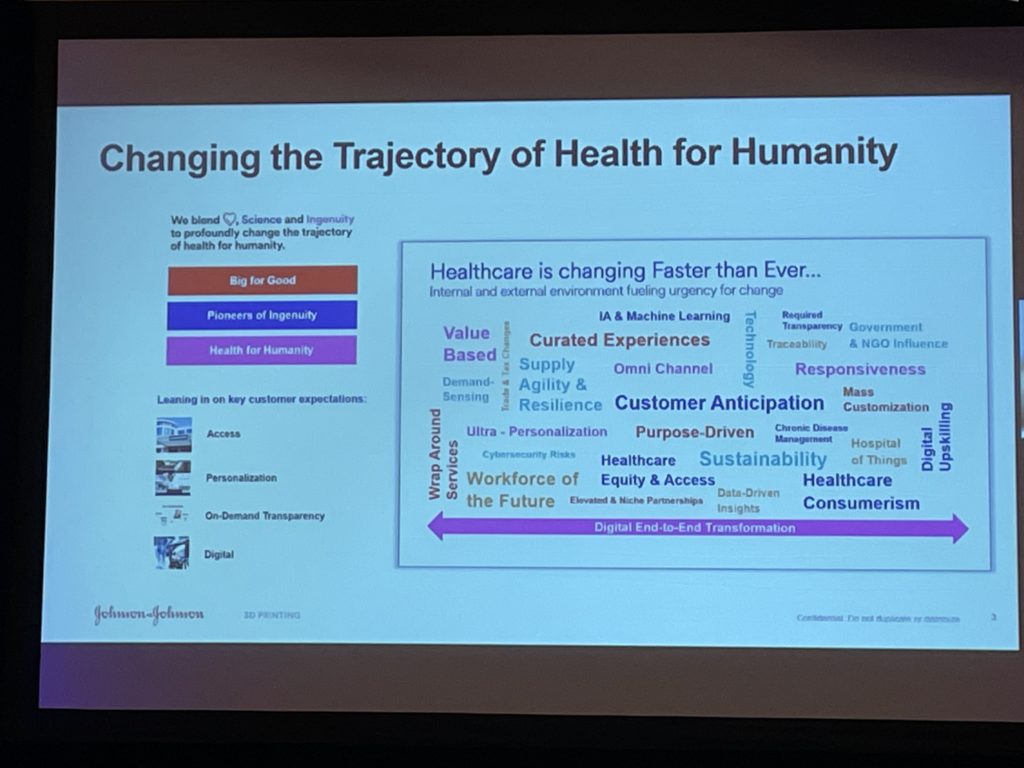
“If you think about the healthcare space, it’s definitely constantly evolving. A lot of things are happening,” Onukuri said. “One of our broad goals at J&J is to help change the trajectory of human health.”
He listed several of the problems that AM sees in the healthcare space, including reimbursement struggles, an aging population, lack of personalization, and high total cost of care, among others. By investing in technology areas such as 3D printing, bioprinting, and personalized medicine, Onukuri said we can “really transform some of the issues we have in healthcare,” and not only in the medical device space, but also in consumer health and pharmaceuticals.
“For all of these workflows, we think 3D printing has a platform of value across all of them,” Onukuri explained.
“We saw the value of the 3D printing platform when we first started, like ultra-personalization for implants and beauty products. 3D printing can definitely bring a lot of value here,” he continued. “The other area is the design possibilities compared to the traditional way of manufacturing, 3D printing brings a lot of design freedom for devices, concepts, various pharmaceutical solutions, and then global reach and access.”
The technology is scalable, he went on to say, and as such can offer a lot of flexibility, such as with point-of-care (POC) printing, which takes place at the clinical setting and is used to fabricate such things as anatomical models, a “very important application.” This scalability has also been valuable in improving the speed and efficiency of manufacturing, which we all know was helpful during the pandemic for tasks like making PPE. Finally, Onukui explained that J&J believes the “overall cost” of things can be improved with 3D printing.
“A lot of the medical implants we use have a lot of expensive materials, like titanium,” he said.
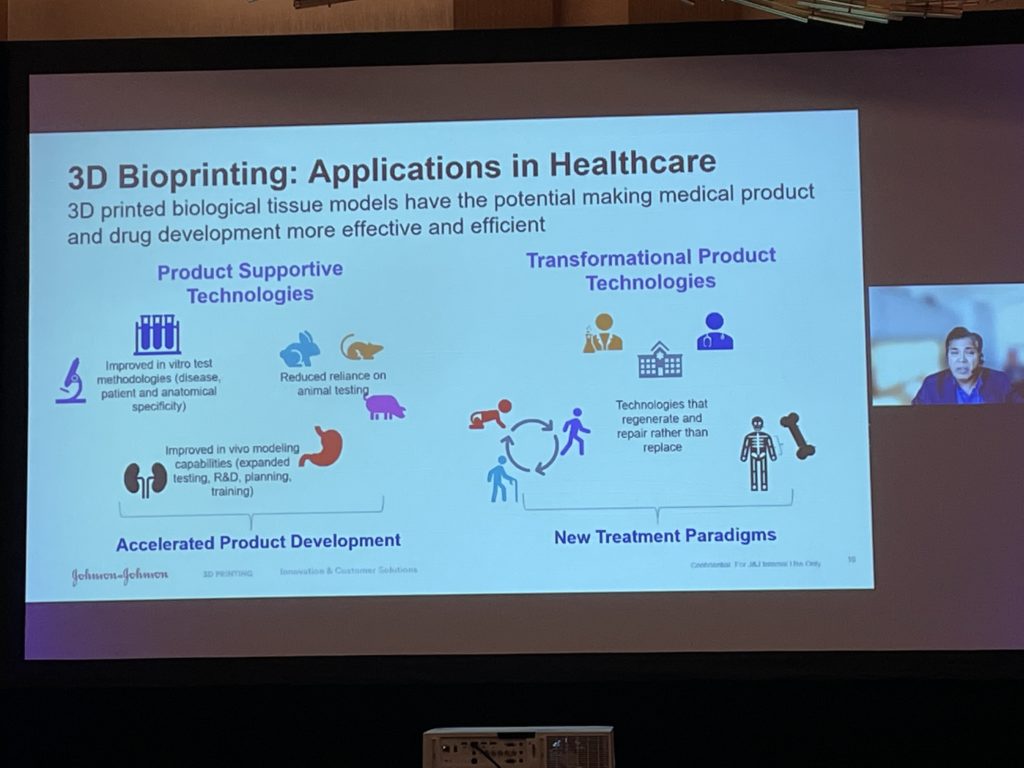
Onukuri went on to explain that J&J has begun to really focus on healthcare applications beyond orthopedics, such as 3D bioprinting, printed sensors and electronics, precision personalized medicine, and more.
“If I leave you with one thought, it’s that 3D printing is not just one thing,” he said. “It’s really a platform of technologies that can be exploited in a very agile manner, depending on the need.”
He said that J&J is really “taking this technology forward,” complete with roadmaps, in six main areas, the first of which is advanced metals and ceramics, followed by polymers and natural materials and then the pharmaceutical space.
“This one is really about printing dosage forms, either in multiple APIs or a single API, because we can get better release profiles and customization,” he said about 3D printing pharmaceuticals.
The next 3D printing innovation area that J&J is focusing on is 3D printed electronics and sensors, because the technology allows for fabrication of parts with very small footprints and high complexity, like sensors that offer 24/7 access to data and biometrics. The last two areas of focus are 3D bioprinting and tissue regenerative technologies, and finally 3D printing at the point-of-care.
“It’s really about bringing new business models,” Onukuri explained.
He provided the attendees with a few examples from these areas where 3D printing has benefitted J&J, the first of which was the CONDUIT interbody platform made with EIT cellular titanium printing.
“The benefits here can be seen at both the macro and nano level, as well as in better osseointegration,” he said.
Next he showed a slide of a TRUMATCH graft cage for a long bone, for which a personalized, PCL-based scaffold was used to print a custom cage, filled with healthy bone.
“It’s bioresorbable and personalized,” he said. “We could not have done this without a technology like 3D printing.”
J&J also sees a lot of value for 3D printing in the pharmaceutical space, from customizing drug dosages and combinations, shapes and sizes, and tailored release profiles. Onukuri explained that the technology made it possible to print the dosage forms locally, and deliver “these personalized therapeutics” right where they’re needed, which obviously keeps things more sustainable since less packaging and shipping is required this way.
“There’s a lot of big potential here, and lots of other companies are also looking in this space,” he said. “This is a great application of 3D printing offering some therapeutic value.”
Moving on to sensing technologies, he said that 3D printing makes it possible to create flexible, enhanced diagnostic and therapeutic capabilities that “allow data to be gathered for the healing process.” Then he said that J&J is piloting a point-of-care business model at several locations around the world, which helps educate patients about their anatomy and the overall surgical process.
“Potential benefits here include ultra-personalized care, shorter surgical time, and improved outcomes,” Onukuri said. “We can move into cutting guides, surgical templates, and implants and instruments as well. This is a good application for 3D printing from a business model perspective.”
Finally, he discussed the “rapidly evolving” 3D bioprinting space, which is beginning to move from the purely academic into the commercial space; J&J “definitely sees a big value here,” Onukuri stated. The technology is helping drive R&D and testing efforts, as well as expanding in vivo modeling, and can hopefully reduce the need for animal and human models in the future, in addition to regenerating and repairing instead of only replacing.
“Very broadly, our company cannot do it by ourselves,” he said. “We have a lot of partners, in the academic and government sectors, and technology, like with supply chain channel partners and strategic partners with a broad IP collaboration, and of course hospitals.”
Onukuri’s final example was the FDA-authorized VESper, or Ventilator Expansion Splitter, which was created in a matter of weeks, and not years, thanks to 3D printing.
“This is a very powerful technology,” he concluded. “I think we often see it as only a prototyping tool, but it can be very helpful, from creating novel designs to bringing new things to market.”
The next keynote presentation I attended was given by Venkat Vedula, Executive Director of Raytheon Technologies, on Accelerating Adoption of AM in Aerospace and Defense; Vedula, who was there in-person, explained that the company was formed last year, at the height of the pandemic, by combining the Raytheon Company and United Technologies Corporation aerospace businesses.
“We are using all seven ASTM-defined categories of additive manufacturing across our businesses, but this morning I’m focusing primarily on powder bed fusion,” he said.
Vedula explained that at Raytheon, they learned early on that when dealing with AM, it was necessary to look at the entire value stream, starting with the powder and how it was stored, transported, etc. Then it was on to build themes, part builds, and materials characterization, thermal processing and finishing, and finally quality and inspection and certification and industrialization.
He told attendees that Raytheon had been using 3D printing for a few decades, but pivoted to begin “actively pursuing” additive manufacturing when metal was available.
On a slide showing metal 3D printed parts made by Raytheon, Vedula pointed out two, highlighted in orange, that are currently on the International Space Station.
“In fact, the very first part we qualified back in 2008 has also been on the space station,” he said, about a3D printed impeller for water separation.
Raytheon has four AM use cases: optimized designs, part unitization, aftermarket parts, and part repair and substitution—the company’s primary use case. All the business drives for these use cases are some combination of performance, size and weight, design cycle, and cost and lead time reduction. Vedula said that build for additive and design tool chain are the “key drivers for cost reduction.”
“This is about how you can nest different parts together to bring down costs,” he explained.
Powder recycling and in-process monitoring and control will be “key for inspection methods and multi-laser large footprint machines,” and other considerations are identifying the right parts and valuable business cases for additive.
“No one should walk away thinking we’ll have a significant cost reduction for all parts made with traditional casting if they switch to additive manufacturing.”
Vedula explained that for Raytheon, qualification is partially based on part, part family, and process, and that “ultimately, the certification approach is no different than how we go about it for castings.” The company looks closely at a part, as well as the risks.
“We have an additive policy at our company for all we print internally, including design allowables,” he said. “Statistical process control is absolutely critical, but we have to have a physics-based understanding in looking at machine qualification.”
Vedula got into the physics a little, and showed the room some analytical process models for nickel, titanium, steel, and aluminum alloys that offer some insight into the additive process.
“These analytical models are multi-dimensional maps. This is the level of understanding you need to have control over the overall process.”
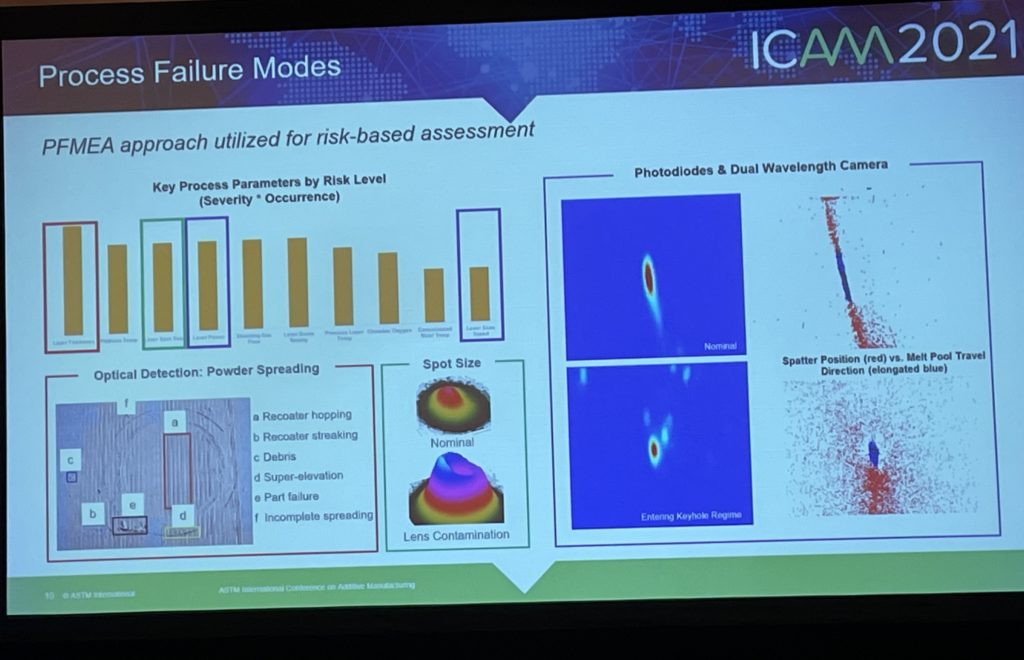
He explained that Raytheon uses the PFMEA approach for risk-based assessment, and showed some of the key process parameters in order of risk level. The company looks at a variety of factors, including laser spot size, gas flow, and spatter prediction, to assess the risk of 3D printing a certain part. Raytheon has a contract with the US Department of Energy to look at melt pool technology, as well as a project called ULTIMATE, or Ultrahigh Temperature Impervious Materials Advancing Turbine Efficiency.
“We also used the physics-based process to develop custom laser scan strategies for heat exchangers,” he said. “We were able to reduce the thickness by about 50% and decrease surface roughness by 50% also.”
Vedula said something that excites Raytheon about metal 3D printing in the aerospace sector is the TJ-150 Turbojet Attritable Engine by Pratt & Whitney’s GatorWorks prototyping arm, and explained that the company was looking at a “truly holistic approach for additive manufacturing for the whole engine.”
“The engine was roughly 100 or so parts, and we were able to move it down to a single additive part,” he said. “We were also able to use additive to achieve zero post-processing. It had no supports, and went straight into a successful engine test.”
As many know, lightweighting is an extremely important factor in aviation applications, so this 99% part count reduction is a big deal. In a successful test of the engine, it achieved twice its target mission life, no doubt in large part because of additive manufacturing. Additionally, while an engine redesign typically takes anywhere from 24-36 months using conventional manufacturing, it only took eight months for Raytheon to get through two design iterations and two engine tests using AM.
“For a broad adoption of AM, it really requires a scientific understanding of the process,” Vedula concluded.
After the keynote presentation was over, Kathie Morgan, the President of ASTM International, took the stage to tell us all some exciting statistics about what ended up being the organization’s largest event this year: more than 300 people were attending ICAM 2021 in-person, with another 500 joining in virtually.
“Hopefully next year, we’ll get 800 to 1,000 people at ICAM 2022,” she said.
Morgan explained that ASTM was established in 1898, specifically for standardizing railway steel rails, and now includes nearly 160 technical committees for multiple industry sectors, like robotics, oil and gas, and additive manufacturing.
“More than 13,000 standards developed by ASTM are used around the world, and we’re really proud that more than 8,000 of those experts come from 140 different countries,” she said. “So we’re a facilitator, and all these technology experts then exchange ideas, debate tech issues, reach a consensus, and develop some of the best standards in the world.”
ASTM’s Additive Manufacturing Center of Excellence (AMCOE) was established in 2018, and supports strategic R&D efforts, working to build trust in technology, close standardization gaps, drive quality, accelerate commercialization, and identifying and filling workforce gaps. 37 of ASTM’s over 13,000 published standards are AM-related, with another 77 currently in development.
Morgan said that ICAM began as a simple workshop in 2016 about AM fracture and fatigue, but over the next few years developed into a symposium, finally expanding into more than 25 symposia over five days, “which brings us to today.”
“Six parallel sessions over five days, eight keynotes, over 550 talks, 12 panels, and more than 800 attendees from all over the world that represent key stakeholder interest…we believe ICAM has created a venue to connect the community together and move the additive manufacturing industry forward.”
Morgan closed by stating that Diversity, Equity, and Inclusion (DEI) initiatives were of personal importance to her, and was happy to have both hosted and attended the Women in 3D Printing happy hour at ICAM the night before. She explained that ASTM’s DEI initiative starts with the words, “Every voice is unique, so every voice matters,” and said that the organization was “already beginning to see some positive impacts.
“Last year five women joined the Board of Directors,” Morgan said. “We are excited to support the Women in 3D Printing Mission. We sponsored the TIPE Conference last year, and will do it again this year, and keep making sure that women have a strong voice in the 3D printing industry.”
Subscribe to Our Email Newsletter
Stay up-to-date on all the latest news from the 3D printing industry and receive information and offers from third party vendors.
Print Services
Upload your 3D Models and get them printed quickly and efficiently.
You May Also Like
Where the Money Is Going: The New Infrastructure Landscape
This is Part 1 of a two-part PRO series examining where infrastructure investment is flowing and how those trends are reshaping manufacturing, energy, logistics, and additive manufacturing. Part 2, by...
3D Printing & Drone Dominance: Speed, Performance, and Derisking the Supply Chain
A shift is underway in drone manufacturing. Government programs like the U.S. Department of War’s Drone Dominance, a $1.1 billion effort to deliver low-cost, one-way attack (OWA) sUAS at scale,...
3D Printing Financials: Stratasys Bets on Defense and Drones as Printer Sales Slow
Stratasys (Nasdaq: SSYS) started 2026 with lower revenue and a larger loss as customers continued to slow down spending on new 3D printers. Still, the company pointed to stable recurring...
INDOPACOM Advanced Manufacturing Team Saves Thousands Per Week at Joint Exercise in the Philippines
In the summer of 2025, the US Indo-Pacific Command (INDOPACOM) opened a new advanced manufacturing facility at Schofield Barracks, the Hawaiian home of the US Army’s 25th Infantry Division. INDOPACOM...