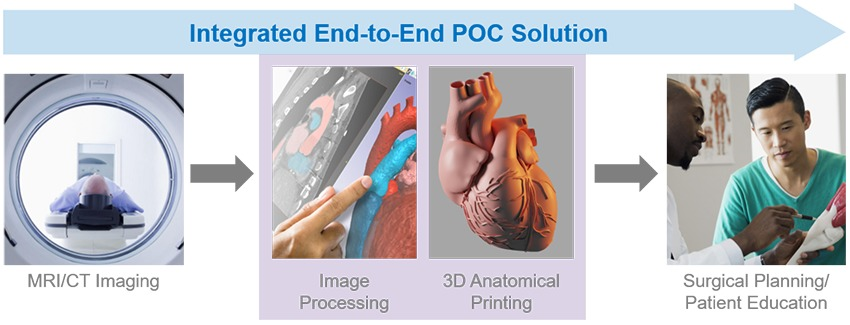
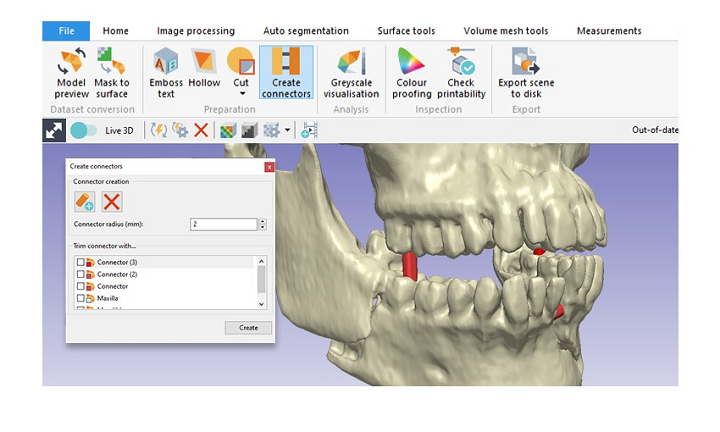
Synopsys, with its worldwide headquarters located in California and a score of other locations all across the globe, is focused on smart technology, and with more than 30 years in business, I’d say the company pretty well knows what it’s doing. Its Simpleware software seems tailormade for the medical industry, as it ” offers complete 3D image segmentation and model generation solutions for going from scans to 3D models,” though it can also be used for non-destructive testing and inspection of industrial parts and materials. But back to medical—Synopsys has announced that its Simpleware ScanIP Medical software solution is now available to use for medical 3D printing and diagnostic decision-making, as it has officially received FDA 510(k) clearance.
According to the FDA, the software’s 510(k) Indications for Use are the following:
“Simpleware ScanIP Medical is intended for use as a software interface and image segmentation system for the transfer of medical imaging information to an output file. It is also intended as pre-operative software for diagnostic and surgical planning.
“For these purposes, output files can also be used for the fabrication of physical replicas using traditional or additive manufacturing methods. The physical replicas can be used for diagnostic purposes in the field of orthopedic, maxillofacial and cardiovascular applications.
“The software is intended to be used in conjunction with other diagnostic tools and expert clinical judgment.”
With this newly received clearance, the Simpleware software can now be used in diagnostic DICOM-3D printing workflows, and Synopsys joins a very small club of other software programs to have received this kind of clearance from the FDA. In addition to its new 510(k) clearance, Simpleware ScanIP Medical is also CE and ISO 13485:2016-certified as a medical device for working with medical imaging data.
“Simpleware software offers a fast, easy-to-use solution for processing medical image data into 3D models for pre-surgical planning and 3D printing. Use the software to generate watertight STLs for direct export to 3D printing applications. Get more from patient-specific images by combining image and CAD data for evaluating implant positions. Take advantage of tools for visualizing and measuring medical image data, as well as for exporting meshes suitable for FE/CFD simulation,” the Synopsys website states.
This FDA-cleared Simpleware solution is advantageous for point-of-care (POC) 3D printing for many reasons. First, it can be used to reliably create high-quality, accurate anatomical models, which physicians can then use for surgical planning purposes, and with its intuitive interface and quick access to features and tools, the software is easy to learn and use. Simpleware ScanIP Medical is also customizable, so plug-ins and scripting can be used to automate repeatable tasks. Finally, the software includes a direct link to bundled 3D printers so they can all work together, and all licenses come with full support from the expert team of Synopsys application engineers.
“Our technology has always been cutting-edge and best-in-class for working with 3D images and generating 3D models, and it is great to now have regulatory clearance that includes point-of-care 3D printing,” said Kerim Genc, PhD, Business Development Manager at Synopsys. “We are excited to deploy these solutions within a point-of-care clinical space, and to leverage our deep technical expertise to provide much-needed solutions to existing bottlenecks in this area.”
As part of its FDA 510(k) clearance for diagnostic 3D printing, Synopsys is working with several other 3D printing companies to offer an integrated, end-to-end POC solution that covers the entirety of the DICOM-to-3D print workflow. For orthopedics and maxillofacial printing, the company is working with the XRIZE 3D printer and Rizium One material, in addition to the Stratasys J750, J750 DAP, J5 MediJet, and a host of Vero materials, including Vero Clear and Vero WhitePlus. The J750 and J750 DAP are also used for cardiac applications, but with the Agilus30Clr material.
If you’re interested in a fully supported trial version of the complete Synopsys Simpleware software suite for diagnostic 3D printing purposes, you can sign up here.
(Images courtesy of Synopsys)
Subscribe to Our Email Newsletter
Stay up-to-date on all the latest news from the 3D printing industry and receive information and offers from third party vendors.
Print Services
Upload your 3D Models and get them printed quickly and efficiently.
You May Also Like
Reinventing Reindustrialization: Why NAVWAR Project Manager Spencer Koroly Invented a Made-in-America 3D Printer
It has become virtually impossible to regularly follow additive manufacturing (AM) industry news and not stumble across the term “defense industrial base” (DIB), a concept encompassing all the many diverse...
Inside The Barnes Global Advisors’ Vision for a Stronger AM Ecosystem
As additive manufacturing (AM) continues to revolutionize the industrial landscape, Pittsburgh-based consultancy The Barnes Global Advisors (TBGA) is helping shape what that future looks like. As the largest independent AM...
Ruggedized: How USMC Innovation Officer Matt Pine Navigates 3D Printing in the Military
Disclaimer: Matt Pine’s views are not the views of the Department of Defense nor the U.S. Marine Corps Throughout this decade thus far, the military’s adoption of additive manufacturing (AM)...
U.S. Congress Calls Out 3D Printing in Proposal for Commercial Reserve Manufacturing Network
Last week, the U.S. House of Representatives’ Appropriations Committee moved the FY 2026 defense bill forward to the House floor. Included in the legislation is a $131 million proposal for...