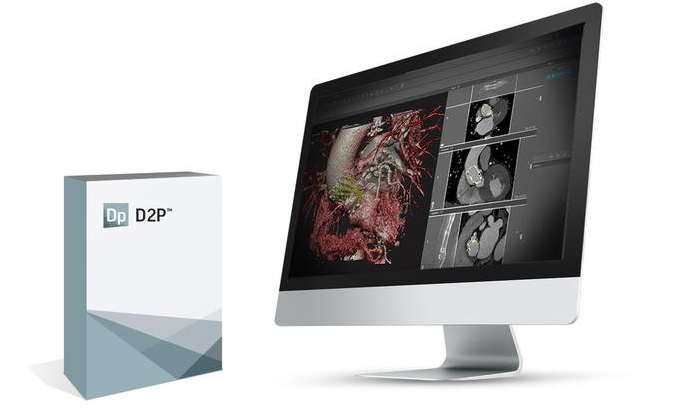
3D Systems has long been involved with using 3D technology to change and save lives in the medical industry. Last summer, the company introduced a new On Demand Anatomical Models service, which allows medical professionals to order 3D printed anatomical models from their own files. The service lets users upload 3D model files, which can be prepared with the company’s D2P (DICOM to PRINT) software. The technology makes it possible to use medical imaging data to accurately, and quickly, produce digital 3D anatomical models.
3D Systems just announced that it has received additional 510(k) clearance from the FDA for its D2P solution, which makes it the only company that provides a solution which combines its own hardware and software to 3D print patient-specific anatomic models for diagnostic purposes in a wide range of medical specialties, such as cardiovascular, craniofacial, gastrointestinal, genitourinary, musculoskeletal, and neurological.
“The capabilities offered in D2P give the healthcare professional an unprecedented toolset for deeper medical understanding across most medical specialties. Our proprietary advanced visualization technology is changing how medical imaging data is used in a variety of formats such as virtual reality and 3D printed physical models,” stated Ran Bronstein, vice president, chief research and operation officer, 3D Systems.
D2P uses deep learning-driven automatic segmentation tools so that medical practitioners can speed up the process in using medical imaging data to accurately produce digital 3D anatomic models. The software includes the latest in virtual reality visualization, and deep learning image processing technology, which helps decrease the amount of time it typically takes to make this kind of 3D model. In order to help facilitate important surgical planning and comprehension ahead of the surgery, for both surgeons and their patients, the D2P technology has an embedded volumetric VR solution that allows the user to get instant views of patient scans in a 3D environment.
“We are used to going into surgery with uncertainties and an arsenal of contingency plans. With 3D printed models and enhanced 3D visualization tools such as VR, we are able to gain a better understanding of the surgery and pathology we are going to treat. This allows us to come up with a more precise surgery plan designed to deliver a better surgical outcome,” explained Dr. Solomon Dadia, deputy director of the orthopedic-oncology department and director of the 3D surgical center at Souraski Medical Center in Tel Aviv, Israel.
 In accordance with FDA guidelines that were introduced two years ago for 3D printed patient-specific anatomic models, models of diagnostic-quality have to be an output of a Class II regulated medical device software. As previously mentioned, 3D Systems is the only company that offers both its own compatible 3D printers and a software solution that meet this requirement. This additional FDA clearance brings 3D Systems squarely into the Point of Care (POC) market, and its continued need for in-house manufacturing of 3D printed anatomic models made with a reliable, accurate 3D segmentation solution.
In accordance with FDA guidelines that were introduced two years ago for 3D printed patient-specific anatomic models, models of diagnostic-quality have to be an output of a Class II regulated medical device software. As previously mentioned, 3D Systems is the only company that offers both its own compatible 3D printers and a software solution that meet this requirement. This additional FDA clearance brings 3D Systems squarely into the Point of Care (POC) market, and its continued need for in-house manufacturing of 3D printed anatomic models made with a reliable, accurate 3D segmentation solution.
Discuss this story and other 3D printing topics at 3DPrintBoard.com or share your thoughts in the Facebook comments below.
[Images: 3D Systems]Subscribe to Our Email Newsletter
Stay up-to-date on all the latest news from the 3D printing industry and receive information and offers from third party vendors.
You May Also Like
Gorilla Sports GE’s First 3D Printed Titanium Cast
How do you help a gorilla with a broken arm? Sounds like the start of a bad joke a zookeeper might tell, but it’s an actual dilemma recently faced by...
Nylon 3D Printed Parts Made More Functional with Coatings & Colors
Parts 3D printed from polyamide (PA, Nylon) 12 using powder bed fusion (PBF) are a mainstay in the additive manufacturing (AM) industry. While post-finishing processes have improved the porosity of...
$25M to Back Sintavia’s Largest Expansion of Metal 3D Printing Capacity Since 2019
Sintavia, the digital manufacturing company specializing in mission-critical parts for strategic sectors, announced a $25 million investment to increase its production capacity, the largest expansion to its operations since 2019....
Velo3D Initiates Public Offering in a Bid to Strengthen Financial Foundations and Drive Future Growth
Velo3D (NYSE: VLD) has been among a number of publicly traded 3D printing firms that have attempted to weather the current macroeconomic climate. After posting a challenging financial report for 2023,...