Automation, healthcare, metal additive manufacturing (AM), and bioprinting were the four big topics discussed at the 2021 Additive Manufacturing Strategies (AMS) summit. The two-day virtual event gathered experts for panel discussions to talk about trends, challenges, and experiences in fields where no other technology has been as disruptive as AM. During the three-hour bioprinting segment, industry leaders exchanged views on current and future trends and shed some light on the hurdles preventing the bioprinting ecosystem from achieving its full potential, such as limited funding and high-cost technologies.
Currently, the major end-users of bioprinting technologies are universities and research institutes, paving the way for future users in the medical field, like hospitals that will require bioprinters in their operating rooms (ORs). Although we have seen breakthrough advances in technologies, processes, and biomaterials, the path toward realizing 3D printed organs for human transplant is still not entirely outlined. To help unravel some of the major questions surrounding bioprinting, a panel of experts discussed the future applications of the technology, a roadmap of the advances we could see in the next three decades.
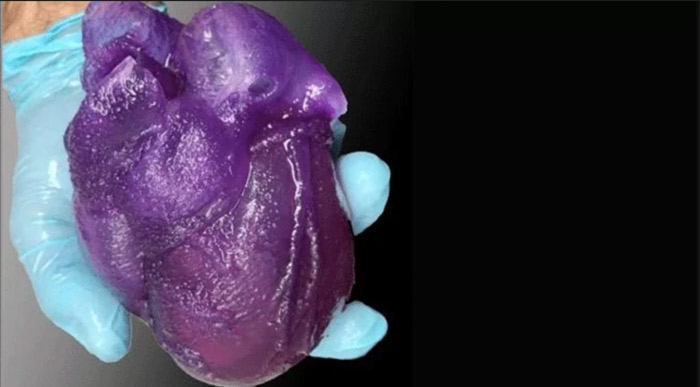 Researchers develop 3D printed human heart model for surgical planning using FRESH. Image courtesy of Adam Feinberg/Carnegie Mellon University.
Researchers develop 3D printed human heart model for surgical planning using FRESH. Image courtesy of Adam Feinberg/Carnegie Mellon University.Leading expert in biomedical engineering and creator of the promising Freeform Reversible Embedding of Suspended Hydrogels (FRESH) technique, Adam Feinberg, considered “we could have a bioprinted heart in an animal in 12 years.” However, the regulatory process to take something like that fully into the clinical phase is difficult to pin down, he determined.
Resources are crucial to the evolution of bioprinting and the commercialization of the technology, said Feinberg. The other panelists agreed, with more resources and a clear timeline to a product, the dynamic for new and existing companies changes completely. Keith Murphy, Co-founder and Executive Chairman of pioneering bioprinting firm Organovo, anticipated more funding would take bioprinting from bench to bedside much quicker – especially if we talk about a billion-dollar figure. Federal government involvement would be ideal in this scenario, Murphy went on, generating a snowball effect and possibly attracting other investors from the private sector.
An early indication that the federal government could support bioprinting initiatives came from President Joe Biden. Feinberg said he was excited to hear him mention 3D bioprinted organs during the introduction of the White House science team. On January 16, 2021, Biden described the members leading the science group as remarkable individuals that are “asking us to imagine 3D printers restoring tissue after traumatic injury, or hospitals printing organs for transplants.”
 Human vasculature model created using Print to Perfusion process. Image courtesy of United Therapeutics.
Human vasculature model created using Print to Perfusion process. Image courtesy of United Therapeutics.For the recently appointed VP of Regenerative Medicine at 3D Systems, Katie Weimer, the outlook is very optimistic. Fully aligned with the company’s end goal to print an unlimited supply of transplantable human lungs, Weimer sees a viable path toward creating functional organs not so far ahead. It is not an “unsolvable moonshot” but rather “an engineering problem,” she said. Instead, Feinberg believes it is not all clear sailing in this environment just yet.
“I think bioprinting a functional tissue or organ is a real objective, but still in the context of something like a moonshot. While we are developing amazing engineering technologies, we don’t understand the biology yet. This falls back to the field of tissue engineering, which has been promising these new technologies and solutions to diseases for 30-plus years, but there is still very little on the market. The success of bioprinting is a combination of technology enabling us to ask new questions about biology and hitting roadblocks as we develop these technologies,” explained Feinberg.
 Particle3D is creating tailor-made 3D printed bone implants. Image courtesy of Particle3D.
Particle3D is creating tailor-made 3D printed bone implants. Image courtesy of Particle3D.The long-term goal of creating transplantable organs might seem decades away, but the commercial landscape of other bioprinting endeavors looks bright for several companies today. At AMS 2021, attendees learned that Particle3D, Aspect Biosystems, Rokit Healthcare, Viscient Biosciences, Dimension Inx, and Poietis have a more linear time-to-market path, particularly because their products are not currently constrained by complex vascular networks or cellular functionality required for organ transplantation. During a panel moderated by 3DPrint.com’s Executive Editor, Joris Peels, the Chief Commercial Officer of biomedical company Particle3D, Casper Slots, revealed their current development of 3D printed biodegradable human bone implants could get to market by 2022, followed by second-generation implants in 2024.
Similarly, Aspect Biosystems combines microfluidics and bioprinting to transform drug discovery and development as well as cellular therapy, aiming to treat various diseases, such as Type 1 diabetes or unforeseen liver toxicity. The biotech company from Vancouver is targeting a middle ground in the broad space of bioprinting, just between cell-free, biocompatible implants and the creation of an organ.
Aspect’s Chief Scientific Officer, Sam Wadsworth, said they are bioprinting rationally designed structures with cells that perform a particular biological function that can affect or cure a disease, but are not trying to recreate the full complexity of an organ. Wadsworth suggested that, for example, in diabetes, they won’t need to print an entire pancreas to release insulin that controls blood glucose in a patient. Even though there are extra regulatory challenges, he hopes to see clinical trials for printed cell-containing implants starting in the next two years and coming into clinical use within five years.
 Aspect Biosystems combines microfluidics and bioprinting to transform drug discovery and development and cellular therapy. Image courtesy of Aspect Biosystems.
Aspect Biosystems combines microfluidics and bioprinting to transform drug discovery and development and cellular therapy. Image courtesy of Aspect Biosystems.Another company exploring the middle ground in bioprinting is South Korea’s Rokit Healthcare. While discussing the next steps for bioprinting in a clinical-stage, Rokit’s Senior Bio-Consultant Da-Yae Lee described how Rokit leverages the technology as an automation method for personalized fabrication using autologous bioinks. The regenerative medicine company has been consulting with hospitals and healthcare professionals and treating patients on four fronts: diabetic foot ulcer, osteoarthritis, kidney disease, and specific chronic aging illnesses. For bioprinters to work in the OR, they have to be compact, combine a bioreactor-type function and a sterilization beaker inside the device instead of requiring separate biosafety cabinets. Rokit’s clinical studies experience has led it to create 4D regeneration platforms to help treat patients.
During the event, attendees also heard from bioprinter manufacturers, like global leader Cellink and scientific developer Sciperio. While Swedish-based Cellink has focused efforts on reducing the price of bioprinting devices from the quarter-million mark to a $10,000 base price, making it accessible to hundreds of labs worldwide, Sciperio is building customized bioprinters. The research arm of nScrypt, Sciperio, has created unique models for the U.S. Department of Defense, academia, private companies, and even a bioprinter for the International Space Station.
 NASA astronaut Andrew Morgan works on setting up the BioFabrication Facility by nScrypt and Techshot, to test-print tissues in the International Space Station. Image courtesy of NASA.
NASA astronaut Andrew Morgan works on setting up the BioFabrication Facility by nScrypt and Techshot, to test-print tissues in the International Space Station. Image courtesy of NASA.In developing countries, where most research labs lack the funds to access high-end machines, many startups have begun commercializing their own accessible platforms. CEOs from Armenia-based Foldink and Brazil’s TissueLabs discussed additional challenges faced in their home countries. Although funding is an issue for labs everywhere, it’s even more serious in developing nations that have not invested in innovation or the talent pool needed for the technology to thrive. Researchers everywhere want to fabricate better structures, and now, they have the tools to get there.
As bioprinting aims to amplify and accelerate tissue engineering and regenerative medicine, it has the potential to transform the medical field, revolutionizing the way healthcare professionals treat patients. In 2021, AMS dedicated an entire afternoon to sharing technologies and knowledge in bioprinting, focusing on new products and innovative materials. The grounded perspective of the experts and their views on the democratization of bioprinting technology was enlightening and a clear timeline of the future of bioprinting.
Subscribe to Our Email Newsletter
Stay up-to-date on all the latest news from the 3D printing industry and receive information and offers from third party vendors.
Print Services
Upload your 3D Models and get them printed quickly and efficiently.
You May Also Like
The Convergence of Vision and Experience: AMS and AMUG
During the last few weeks, I spent time on the ground at both the Additive Manufacturing Strategies Forum (AMS) and the Additive Manufacturing Users Group (AMUG) meeting. What stands out...
HP’s Open Industrial Filament System Made North American Debut at AMUG 2026
At Formnext 2025, HP Additive Manufacturing revealed a major step in its broader portfolio expansion strategy with the announcement of a new 3D printer, the HP Industrial Filament 600 High...
The Magic of AMUG as Reported by a First-Time Attendee
There’s a special kind of magic about AMUG. I’ve heard about it for years, but never experienced it myself until last week. It’s different than what you see at some...
Würth Additive Group & B9Creations Announce Strategic Partnership at AMUG 2026
Last week, hundreds of additive manufacturing users (and a few journalists, like me!) met in Reno, Nevada to attend the 2026 Additive Manufacturing Users Group (AMUG) Conference. I’ll share more...






























