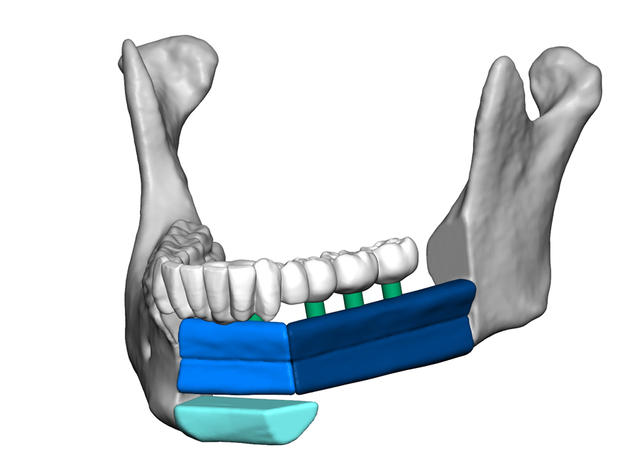
A veritable pioneer in 3D printing for personalized medicine applications, 3D Systems (NYSE:DDD) created its Virtual Surgical Planning (VSP) system for craniomaxillofacial (CMF) and orthopaedic cases years ago, and it’s helped change the lives of many patients. VSP is a service-based approach, pairing 3D printing with surgical simulation and medical imaging to provide personalized surgery to patients, and has been used in over 120,000 unique cases. 3D Systems is now sharing some exciting news about VSP: the Food and Drug Administration (FDA) has approved 510(k) clearance for maxillofacial surgical guides 3D printed out of its LaserForm ® Ti and DuraForm ® ProX PA materials.
VSP works like this: the surgeon gets things started, meeting online with a biomedical engineer from 3D Systems to present the surgical plan they hope to use, and together they simulate and plan out the procedure. The meeting results in a digital plan, consisting of patient-specific, 3D printable templates, anatomical model, and surgical guides, that’s then sent directly to the OR. It’s been shown that using 3D printed guides can reduce the number of hours in surgery, and when they’re made for VSP purposes, with these newly cleared materials, the guides have a lot more design flexibility, with better rigidity, strength, and performance, and can help, according to a 3D Systems press release, “enhance visibility and access in the surgical site” for reconstructive and maxillofacial procedures.
The company announced an exclusive partnership with Stryker’s CMF division two years ago, meant to provide innovative maxillofacial surgical solutions, and this new FDA clearance is just the latest success for their mutual objectives.
“Through close collaboration with surgeons and Stryker’s CMF division, we’ve uncovered opportunities to refine VSP guide designs that leverage additional capabilities in our materials portfolio. Our expert biomedical engineers are now able to design surgical guides tailored to the surgeon’s needs with enhanced properties that can help improve accuracy and facilitate procedures in ways not previously possible,” stated Menno Ellis, Executive Vice President, Healthcare Solutions, 3D Systems. “Our powerful VSP System continues to transform surgery – enabling better patient outcomes.”
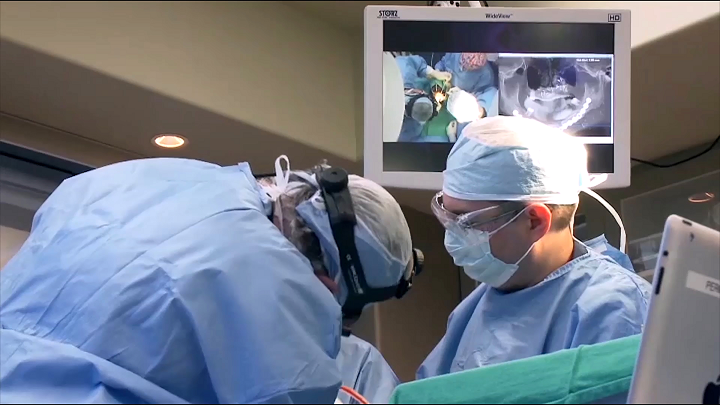
Surgeons delivering enhanced surgical treatments using 3D Systems’ VSP® (Virtual Surgical Planning) which delivers accurate, patient-specific solutions for better treatment outcomes.
Using these new materials, 3D Systems can print customized surgical guide designs, that are stronger and more durable but less bulky, that will help surgeons improve their performance. For example, according to mechanical tests, titanium marking and cutting guides printed out of the company’s LaserForm Ti are 20 times stronger than guides fabricated using traditional methods of manufacturing, and fibula cutting guides made with this titanium material have shown to be 70% thinner than regular guides, which provides easier access to the surgical site.
Nylon guides made with DuraForm ProX PA are also thinner, and can hold up better under forces applied during surgery, because they have been shown to feature 88% higher toughness than other guides. This allows surgeons to achieve “a close, snap-like, fit to patient anatomy” not possible with normal nylon materials.
Using tailored guides made with these two materials can help surgeons be more accurate when performing drilling and cutting operations, and they’ve also been validated with a larger number of options for either automated or manual cleaning methods, as well as more steam sterilization cycles, which is obviously very important when dealing with tools for surgical applications.
As an added bonus, the newly FDA-cleared materials, as well as 3D Systems’ new surgical guide designs, are also compatible with Stryker’s Facial iD range of patient-matched plating solutions. So maxillofacial surgeons can rest easier, knowing they have a solid, yet flexible, end-to-end solution for patient-matched treatment with 3D Systems VSP. Using patient-specific implants, surgical planning, and better marking and cutting guides made with the newly FDA-cleared LaserForm Ti and DuraForm ProX PA materials, 3D Systems provides “an unmatched portfolio of material and guide design options.”
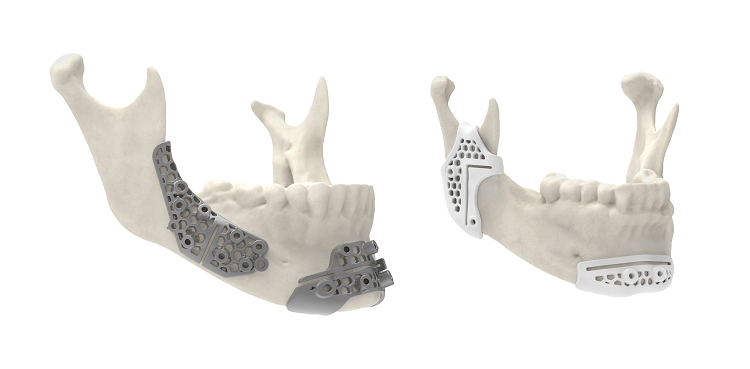
Maxillofacial surgical guides 3D printed using 3D System’s LaserForm® Ti and DuraForm® ProX PA materials allow for more innovative designs that improve performance.
(Source/Images: 3D Systems)
Subscribe to Our Email Newsletter
Stay up-to-date on all the latest news from the 3D printing industry and receive information and offers from third party vendors.
You May Also Like
Further Understanding of 3D Printing Design at ADDITIV Design World
ADDITIV is back once again! This time, the virtual platform for additive manufacturing will be holding the first-ever edition of ADDITIV Design World on May 23rd from 9:00 AM –...
3D Printer Maker EVO-tech Reborn as NEVO3D — Once More With Feeling
EVO-tech was a 3D printing service and original equipment manufacturer established in 2013 and based in Schörfling am Attersee, Austria. The company produced high-quality material extrusion systems featuring linear bearings,...
3D Systems Brings 3D Printed PEEK Cranial Implant to the U.S. with FDA Clearance
For more than 10 years, 3D Systems (NYSE:DDD) has worked hand-in-hand with surgeons to plan over 150,000 patient-specific cases, and develop more than two million instruments and implants from its...
CDFAM Returns to Berlin for Second Annual Symposium
The second CDFAM Computational Design Symposium is scheduled for May 7-8, 2024, in Berlin, and will convene leading experts in computational design across all scales. Building upon the first event...