Companies, organizations and individuals continue to attempt to lend support to the COVID-19 pandemic supply effort. We will be providing regular updates about these initiatives where necessary in an attempt to ensure that the 3D printing community is aware of what is being done, what can be done and what shouldn’t be done to provide coronavirus aid.
Now that 3D-printed medical supplies seem to have reached peak momentum, there has been enough time to gather data about these items and the larger additive manufacturing (AM) industry as it relates to the pandemic. Researchers from Deakin University in Australia have published a study about 3D-printed medical supplies, specifically looking at what was made using what technology.
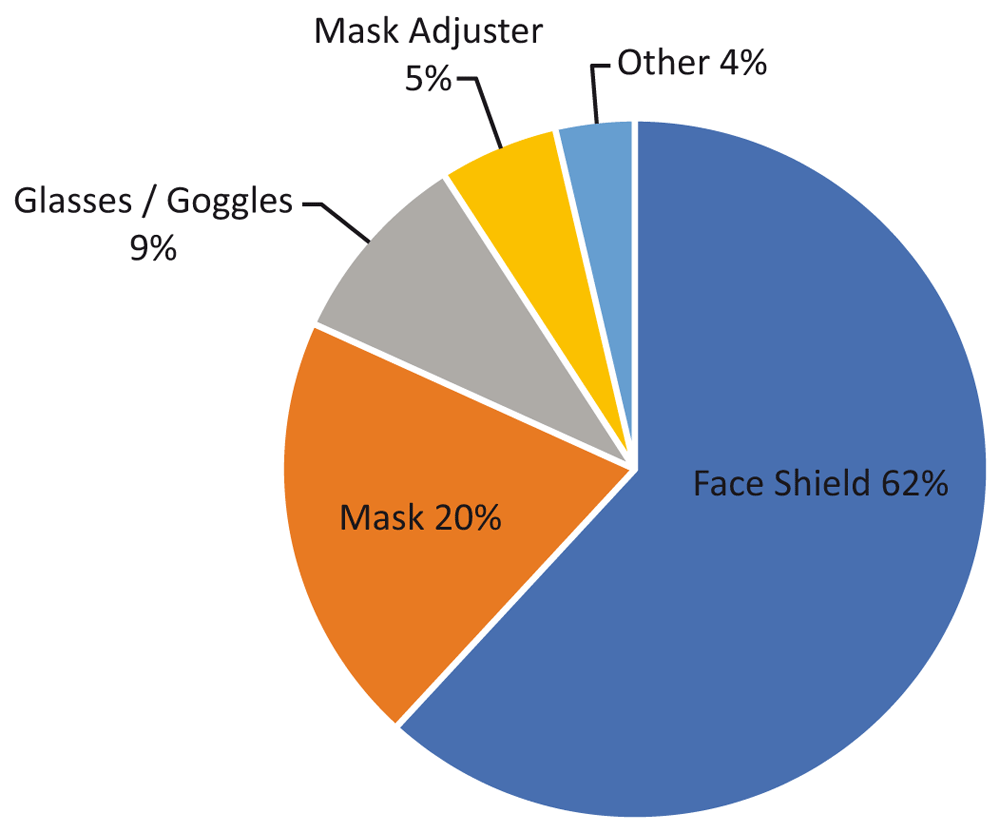
From the study: “Types of PPE products being 3D printed during February-March 2020.” Image courtesy of “A critical review of initial 3D printed products responding to COVID-19 health and supply chain challenges.”
The report, published in Emerald Open Research, lists the first 3D-printed items to address supply issues as occurring on February 11: a series of safety goggles for healthcare workers made by Hunan Vanguard Group in China and 3D-printed concrete isolation houses for Xianning Central Hospital. Once the World Health Organization declared the COVID-19 outbreak as a pandemic on March 11, 3D printing projects related to the disease grew at a massive rate, with 92 percent of projects occurring after that time.
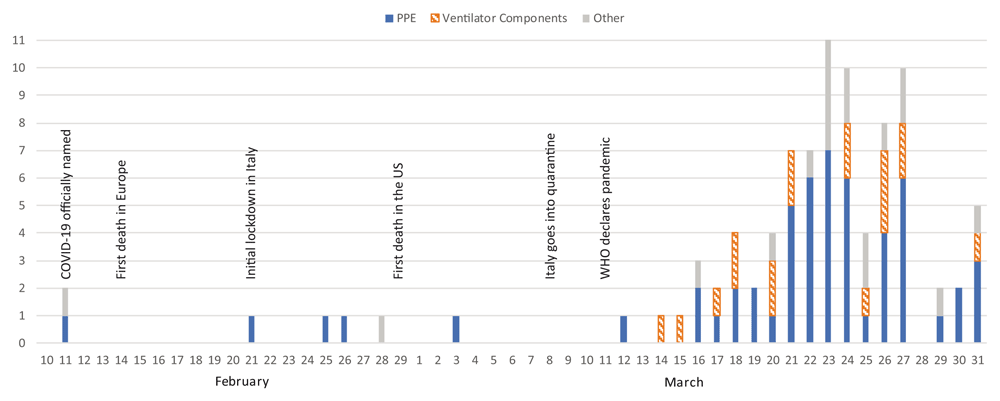
From the study: “COVID-19 3D printing projects mapped against date of release/publication PPE, personal protective equipment.” Image courtesy of “A critical review of initial 3D printed products responding to COVID-19 health and supply chain challenges.”
The authors looked at 91 3D printing projects set up since April 1, 2020, determining that about 60 percent of 3D-printed medical supplies was made up of personal protection equipment (PPE), 62 percent of which was face shields. Fused filament fabrication (FFF) was the primary mode of production. 20 percent were ventilator parts and 20 percent were for a variety of items, such as nasopharyngeal swabs and hands-free door openers.
FFF was more often used for face shields, while stereolithography and digital light processing were more often used for ventilator parts. Selective laser sintering was most used for hands-free door openers but was spread across multiple categories. Multi Jet Fusion was evenly distributed across item categories.
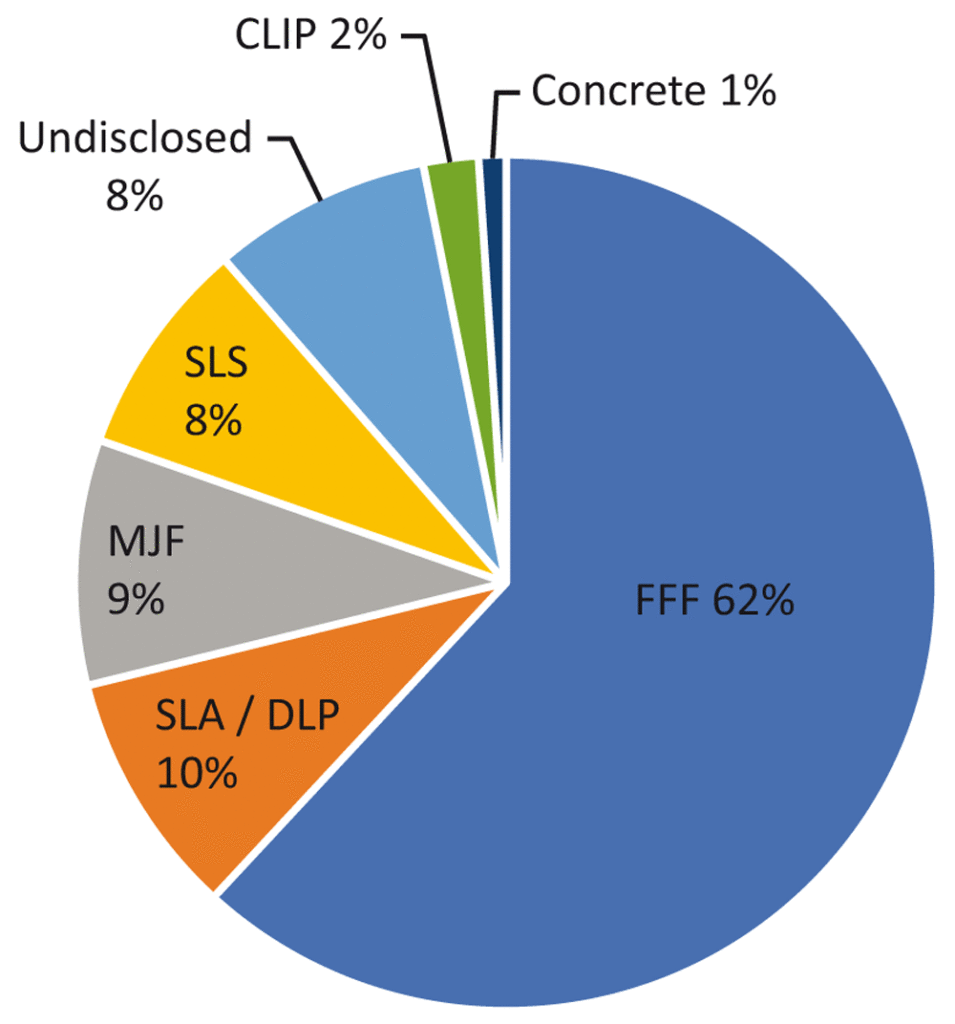
From the study: “3D printing technologies utilised in projects. FFF, fused filament fabrication; SLA, stereolithography; DLP, digital light projection; MJF, multi jet fusion; SLS, selective laser sintering; CLIP, continuous liquid interface production.” Image courtesy of “A critical review of initial 3D printed products responding to COVID-19 health and supply chain challenges.”
The authors noted that there was wide variation in designs for the same issue, exemplified by the 34 different face shields made, including some that were meant for different machines and printing processes. This could naturally cause confusion for health professionals unfamiliar with the varying technologies and systems. The authors even cited our own Joris Peels in highlighting the possibility that some of these efforts could have been “a marketing exercise that adds further confusion for those wanting to help 3D print supplies. The appearance of branding on many designs was evidence of this.”
The report suggests that project reporting peaked on March 23 with the authors hypothesizing as to what caused to reporting to die off: U.S. Food and Drug Administration announcements could have caused some teams to reconsider their projects; some efforts may have been merged for collaboration purposes; supply chains could have begun to stabilize by that point.
“Elements of manufacturing in a post-COVID-19 future may look very different to pre-COVID-19. 3D printing could be central to new ways of thinking about making and distribution, but only if it is successful in avoiding being undermined by hype,” the authors reflect. “Researchers, manufacturers and those with a vested interest in 3D printing must commit to building products designed for each additive manufacturing technology, with systems that maximise potential, whilst shouldering the responsibilities involved in producing viable, qualified products that can be relied on by society whether in a crisis or not.”
Others have begun analyzing the relationship between the pandemic and the 3D printing industry, including the European manufacturing trade group CECIMO, who published an article on recommendations for policymakers based off of its experience in acting as a hub for European efforts to 3D print medical supplies. In anticipation of a phase two for the fight against COVID-19, the organization has made the following suggestions:
- Fast-track standards and certification development for AM
- EU funding to allow Europe to attain autonomy in fabricating critical goods, using AM and other technologies
- Regulations that incorporate AM into existing industry
- Address the need for AM skills in the workforce
CECIMO naturally benefits from some of these recommendations, as it would likely play an important part in some if not all of them.
Outside of these reflections on the role of the 3D printing industry during the pandemic, we have already discussed what Joshua Pearce at Michigan Technological University has suggested, which is an increase in the use of open source designs and licenses in order to more quickly innovate and spread 3D printable supplies.
As the pandemic continues to grip the world, we will continue to provide regular updates about what the 3D printing community is doing in response. As always, it is important to keep safety in mind, remain critical about the potential marketing and financial interests behind seemingly good humanitarian efforts from businesses, and to do no harm.
Subscribe to Our Email Newsletter
Stay up-to-date on all the latest news from the 3D printing industry and receive information and offers from third party vendors.
You May Also Like
Gorilla Sports GE’s First 3D Printed Titanium Cast
How do you help a gorilla with a broken arm? Sounds like the start of a bad joke a zookeeper might tell, but it’s an actual dilemma recently faced by...
Nylon 3D Printed Parts Made More Functional with Coatings & Colors
Parts 3D printed from polyamide (PA, Nylon) 12 using powder bed fusion (PBF) are a mainstay in the additive manufacturing (AM) industry. While post-finishing processes have improved the porosity of...
$25M to Back Sintavia’s Largest Expansion of Metal 3D Printing Capacity Since 2019
Sintavia, the digital manufacturing company specializing in mission-critical parts for strategic sectors, announced a $25 million investment to increase its production capacity, the largest expansion to its operations since 2019....
Velo3D Initiates Public Offering in a Bid to Strengthen Financial Foundations and Drive Future Growth
Velo3D (NYSE: VLD) has been among a number of publicly traded 3D printing firms that have attempted to weather the current macroeconomic climate. After posting a challenging financial report for 2023,...































