As the COVID-19 pandemic reached the U.S., fears of ventilator shortages spread across states, who requested far higher numbers of the breathing machines than the national stockpile maintained. According to the Trump administration, the country is now at a point where there are a sufficient number to treat patients with severe respiratory issues resulting from COVID-19, and the U.S. is now shipping ventilators to other countries that are running on short supply.
However, if the administration’s reporting is inaccurate or the situation worsens, we have seen that there may be ways around the shortage. For instance, states could share ventilators based on supply and need and there may be a number of viable, low-cost alternatives waiting in the wing, including simple methods for converting existing equipment into ventilator systems to completely novel and untested, but easy-to-assemble machines. Putting aside the question of whether or not ventilators are the safest method for treating severe respiratory problems, the crisis has raised the question of why these systems, as complicated as they are, are so hard to manufacture.

An open source ventilator designed by Nvidia’s chief scientist, Billy Dally. Image courtesy of OP-Vent.
Professor Joshua Pearce at Michigan Technological University suggests that a key inhibitor to the rapid manufacture of ventilators, other medical equipment, and goods in general is intellectual property (IP).
“The pandemic is exposing the absurdity of the IP system,” Pearce told 3DPrint.com. “There is no ‘fair use’ for patents. This means even if you know how to make something that someone needs and you have the ability, you may not do so without infringing. This is even if it is for yourself or a family member. Even if you never read the patent with crazy wide and vague claims. Normally this is just an inconvenience, but as the conventional proprietary manufacturing system and supply chains have been unable to meet the demand for PPE and other COVID-19 related products, an IP tragedy is unfolding. People are dying unnecessarily because IP restricts anyone from making critical products without a license.”
In previous papers, Pearce has argued that patent rights should be severely weakened, by at least 25 to 50 percent. According to Pearce, the main economic reasons behind patent law suggest that inventors need to recoup costs associated with research and commercialization, but that new technologies, such as 3D printing, synthetic biology and cloud computing shrink these costs to the point that IP protection is no longer as needed.
More importantly, “the IP system is well-known to retard innovation,” Pearce said. “Now antiquated IP laws are not only getting in the way of innovation—they are killing people. It is time to take a hard look at getting rid of IP entirely.”
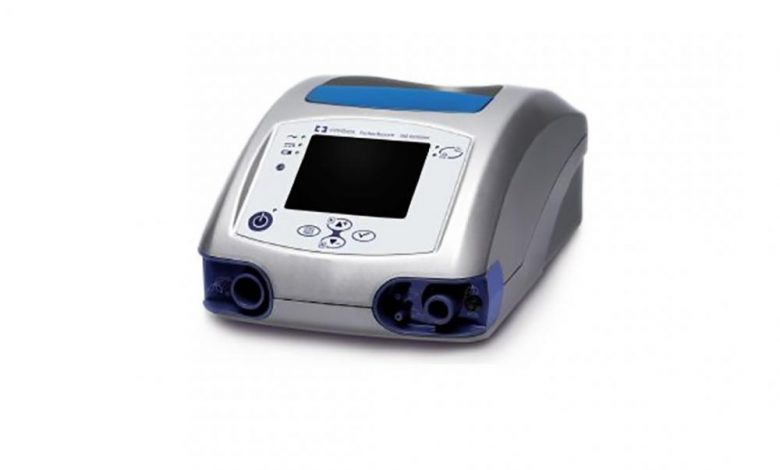
The PB560 is a portable ventilator system from Medtronic that is less expensive and complicated than its other machines. Image courtesy of Medtronic.
As a result of the projected ventilator shortage, at least one manufacturer of the machines did decide to make the designs for their technology available to the public, but in a limited manner. Medtronic is a publicly traded medical device manufacturer with annual revenues of roughly $30 billion, with over 86,000 employees across 140 countries. This includes Ireland, where, despite being a U.S. company, it was able to establish its legal registration by acquiring a manufacturer called Covidien in one of the largest tax inversion operations in history.
In March, the manufacturer opened up the design files (including product and service manuals, requirements documents, manufacturing documents, schematics, 3D CAD files, and software source-code files) for its Puritan Bennett 560 ventilator system. A company representative told 3DPrint.com that this model was chosen in particular due to its size and relative complexity compared to other models in its line:
“Ventilator manufacturing is a complex process that relies on a skilled and specialized workforce, an interconnected global supply chain, and a rigorous regulatory regime to ensure patient safety. Medtronic sources more than 1,500 components to make Puritan Bennett 980 (PB980) and Puritan Bennett 840 (PB840) ventilators. By contrast, the Puritan Bennett 560 (PB560) is a smaller, more compact ventilator that we think will help participants across industries to evaluate options for rapid ventilator manufacturing. The PB560 is a more portable ventilator with a turbine pump that provides pressure from room air and external oxygen. All three ventilators are suitable for institutional settings. The PB560 can be used at home.”
The goal, according to the representative was so that “participants across industries to leverage their own expertise and resources to evaluate options for rapid ventilator manufacturing to help doctors and patients dealing with COVID-19.” So far, the company has received over 100,000 registrations for the design files.
Beyond these quotes, the company mostly rehashed what it had already shared in its press releases and on its website. Professor Pearce pointed out that, as admirable as the corporation’s decision to share the files for its PB560 ventilator is, there may be limits to Medtronic’s generosity.
“When Medtronic first announced an open ventilator project, they garnered a lot of good press. At the time, however, they did not release the CAD, BOM, software, etc. to actually fabricate it, which looked a lot like ‘open washing’,” Pearce said. “Medtronic has now released these documents under a permissive license, which is not what the [Open Source Hardware Association] would consider an open hardware license. All ventilators made from the designs must be labeled with a warning noting that it was built in response to COVID-19 and is only to be used to address this pandemic. The term ends either when the World Health Organization’s official Public Health Emergency of International Concern (PHEIC) is declared over, or on October 1, 2024, whichever comes first. This is obviously sub-optimal and is likely to restrict distribution and cause waste. That said, it is a lot more than any of the other ventilator companies have done.”
While Medtronic was able to make the designs for one of its lower-cost ventilators freely available to the public now, the cost may have been the time it took to do so. In 2007, the Biomedical Advanced Research and Development Authority, newly established by the U.S. Department of Health and Human Services, invested public funds into private companies to develop ventilators in the case of a nationwide flu pandemic. Among the recipients was a small firm called Newport, who was set to develop a $3,000 ventilator.
Before it could deliver one, it was acquired by Covidien, the same company that, once acquired in 2015, gave Medtronic its Irish tax status. According to a New York Times story on the subject, both “government officials and executives at rival ventilator companies said they suspected that Covidien had acquired Newport to prevent it from building a cheaper product that would undermine Covidien’s profits from its existing ventilator business.” It is therefore possible that Medtronic could have released a low-cost ventilator sooner, using Newport’s technology.
This tale is discussed in a recent paper by Pearce titled “Distributed Manufacturing of Open-Source Medical Hardware for Pandemics,” in which he describes the extent to which open source medical supplies could be used to address such pandemics as the one currently taking place, as well as a possible roadmap for establishing open source protocols for such lifesaving technology. In studying the top 20 technologies requested by the Government of India to handle the COVID-19 outbreak, Pearce found that only 15 percent of the equipment for open source development was freely available. The paper recommends the following five actions be taken in order to make open source medical hardware available in order to tackle supply issues during such a pandemic:
- “technical development of a wide range of open source solutions for all medical supplies and devices
- policies that protect the productivity of laboratories, makerspaces and fabrication facilities during a pandemic
- streamlining the regulatory process
- developing Good- Samaritan laws to protect makers and designers of open medical hardware, as well as to compel those with knowledge that will save lives to share it
- requiring all citizen-funded research to be released with free and open source licenses”
In addition to making medical supplies open source, 3D printing is a key method for the proliferation of inexpensive devices, as has been demonstrated by the outpouring of supplies made in response to the pandemic. Pearce’s lab has reported many times on the cost savings made possible with open source 3D printing, but he believes that these medical supplies now being produced will make this value that much more clear.

Medical supplies being produced by students and staff at Michigan Tech. Image courtesy of Michigan Tech.
Without low-cost Newport-style ventilators available ahead of the pandemic, we’ve instead seen an array of efforts attempt to address possible supply issues, including potentially dangerous projects by hobbyists, promotional CAD modeling campaigns, and many viable machines, including a mostly 3D printable ventilator from Pearce’s own research group at Michigan Tech in Open Sustainability Technology.
When asked about other ventilator manufacturers who have so far chosen not to open up their designs, Pearce said that the flood of low-cost systems now being developed will ultimately hurt the big IP holders.
“They are being short sighted by protecting fictional future profits at the expense of human lives. When all the dust settles on this pandemic, the ventilator market is going to be flooded with high-performance low-cost open source ventilators. I know this because between the 1st and 2nd version of my open source ventilator review article, a medical team in Spain published a design and there are designs under review now in the special issue of HardwareX…As these open source ventilator designs make their way through the FDA approval and add features, greedy ventilator IP hoarders are going to be sitting on a stack of useless IP.”
Subscribe to Our Email Newsletter
Stay up-to-date on all the latest news from the 3D printing industry and receive information and offers from third party vendors.
You May Also Like
Gorilla Sports GE’s First 3D Printed Titanium Cast
How do you help a gorilla with a broken arm? Sounds like the start of a bad joke a zookeeper might tell, but it’s an actual dilemma recently faced by...
Nylon 3D Printed Parts Made More Functional with Coatings & Colors
Parts 3D printed from polyamide (PA, Nylon) 12 using powder bed fusion (PBF) are a mainstay in the additive manufacturing (AM) industry. While post-finishing processes have improved the porosity of...
$25M to Back Sintavia’s Largest Expansion of Metal 3D Printing Capacity Since 2019
Sintavia, the digital manufacturing company specializing in mission-critical parts for strategic sectors, announced a $25 million investment to increase its production capacity, the largest expansion to its operations since 2019....
Velo3D Initiates Public Offering in a Bid to Strengthen Financial Foundations and Drive Future Growth
Velo3D (NYSE: VLD) has been among a number of publicly traded 3D printing firms that have attempted to weather the current macroeconomic climate. After posting a challenging financial report for 2023,...































