Safety Recomendations for 3D-Printed COVID-19 Medical Devices, Part One
One of the most important issues that we’ve repeatedly stressed in relation to the 3D printing community’s efforts to fulfill the need for medical supplies amid the COVID-19 pandemic is the need for safety. To understand the precautions that should be taken in relation to various classes of medical devices that are being 3D printed, we spoke with experts from the industry and researched the regulatory guidelines of the U.S. Food and Drug Administration (FDA).
We are still working to expand our knowledge base on this topic, but this is what we’ve learned so far.
Work with medical professionals: In part, we are trying to delineate between the services that can be provided by hobbyists as compared to more expert 3D printing users. To start, we should note that, regardless of the device, designers, engineers and machine operators should work with medical professionals when developing new designs or producing parts. Even if the part is as simple as a face shield, it is important to find a hospital that needs it first, before you start printing. #GetUsPPE has created a script that one can follow to ask hospitals if they need your help and supplies. Please do not needlessly approach your local hospital. Work with local makers and businesses first to find out if they perhaps already have established what your local healthcare providers need. First, check online and contact all of the 3D printing-related businesses locally before you attempt to contact a hospital.
Use medically tested and validated designs: If a part already exists and has been tested and validated, such as headbands for face shields, it is not necessary to reinvent the wheel and a 3D printer user can simply print acceptable designs. The National Institute of Health (NIH) in the U.S. has developed an online file repository for just such a purpose. There are already numerous face shield designs on this site. Please be aware that all files on the site have not yet been tested or validated. Just being on the site does not mean that it works or has been tested. Read the comments underneath a file, as well as the description, to be sure what the limitations and specifics are. On the detail page of the file in question, it should be clearly labeled that the file has been reviewed. Some files on the site have multiple versions floating about elsewhere. Any advice or recommendation on the site must be seen as being specific to the one file in question, not others. Be careful when using different slicing settings, machine settings or opening and reviewing the file in a CAD or 3D modeling program as this may alter the part.
Face shields are safe for hobbyists to make: Based on regulatory classification and the guidance of the U.S. Food and Drug Administration, some products must be limited to facilities with a Quality Management System in place. However, because face shields do not meet this threshold, they are deemed acceptable for hobbyists to print them. In fact, we have come across a number of medical professionals specifically requesting these devices, suggesting their utility. Please do try to use skin approved materials and only obtain materials only from reputable vendors. Please work in a sanitary and clean way and also try to see if you are able to sterilize or clean these masks properly. You not washing your hands properly could have fatal consequences for a person with a compromised immune system.
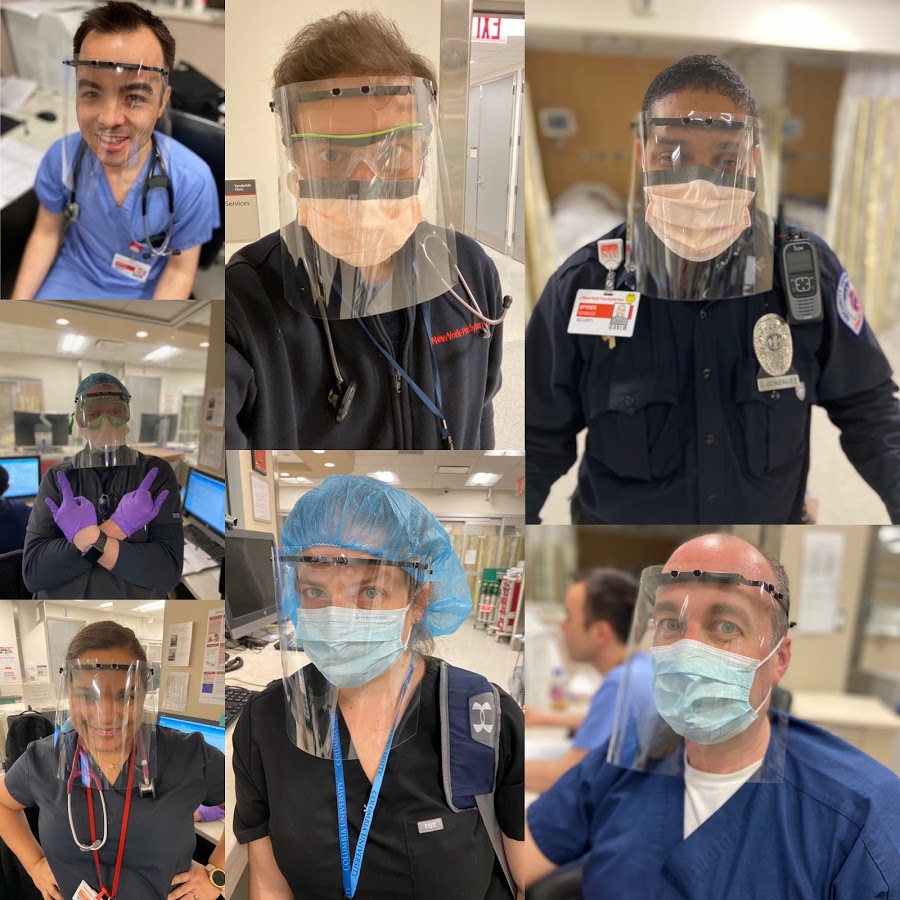
Healthcare professionals at Columbia University Medical Center/New York-Presbyterian Hospital using 3D printed face shields thanks to the MatterHackers Maker Response Hub. Image courtesy of MatterHackers.
Face masks are NOT safe for hobbyists to make for medical professionals: When it comes to face masks, the 3D-printed variety will likely be considered a last resort based on the FDA’s existing guidance. Exceptions include situations in which medical device manufacturers and/or industrial manufacturers work specifically with hospitals and governments to provide 3D print them (as occurred in the Czech Republic, for instance).
The FDA has provided guidelines for face mask usage based on the capacity that a hospital is experiencing, with many facilities facing surge scenarios as a result of the COVID-19 outbreak being told to implement the most extreme methods of face mask supply conservation. This means:
- Canceling “all elective and non-urgent procedures and appointments for which a face mask is typically used by [healthcare personnel (HCP)].”
- Using masks beyond shelf-life dates, re-using masks, and prioritizing masks specific activities, such as contact with an infected patient.
- Alternatives to face masks when supplies have run out, such as face shields, ventilated headboards, and filtration fans, and homemade face masks.
The FDA has provided similar guidelines for protective eyewear, isolation gowns, and N95 respirators.
Face masks are potentially safe for hobbyists to make for NON-MEDICAL uses: Because all face masks with filtration capabilities, such as N95 respirators, are meant to be reserved for medical personnel directly interacting with COVID-19 patients, as discussed above, the general public and non-medical workers must rely on other means to prevent the spread of the disease.
The FDA and Centers for Disease Control and Prevention (CDC) are both recommending that the public wear cloth masks. Though not to the extent possible with surgical masks and N95 respirators, homemade masks prevent the spread of germs by limiting the number of microorganisms expelled by wearers. As a Cambridge University study on the topic explains,
“Face masks reduce aerosol exposure by a combination of the filtering action of the fabric and the seal between the mask and the face. The filtration efficiency of the fabric depends on a variety of factors: the structure and composition of the fabric, and the size, velocity, shape, and physical properties of the particles to which it is exposed. Although any material may provide a physical barrier to an infection, if as a mask it does not fit well around the nose and mouth, or the material freely allows infectious aerosols to pass through it, then it will be of no benefit.”
That said, 3D printer users may be hoping for better protection than can be provided with cloth masks. Essentium, for instance, is providing 3D-printed masks with filtration to local police and firefighters in Texas. The same medical regulations may not apply outside of hospital settings, but we have regulatory bodies to cover industrial goods for a reason. Issues beyond the transmission of viruses need to be considered, such as whether or not a material is allergenic or toxic.
Materials and Workflows Matter: Medical device manufacturers have received regulatory approval for their devices because they have set up the proper facilities with the proper machines and materials for producing their goods. Not all materials and operating environments are suitable for every type of item being 3D-printed in response to coronavirus supply shortages.
3D Systems, for instance, has a Healthcare division that manufactures biocompatible implants, surgical guides, and other devices that have been tested, validated and regulated to meet the safety requirements for use with patients. As Ben Johnson, director of Product Development at 3D Systems Healthcare, explained,
“Face shields likely can be printed in FDM just fine and serve the need of a health care provider just fine, where you can disinfect them. Other types of applications, like face masks or diagnostic tools or other medical device components are likely not acceptable to be manufactured from those types of materials and processes because they wouldn’t meet the essential requirements of those parts.”
While a tabletop fused deposition modeling (FDM) machine is not an unacceptable technology for making emergency supplies, hobbyists don’t typically have the kind of testing and rigor that’s required for specific types of devices, Johnson pointed out. The company uses selective laser sintering (SLS) and nylon to produce many devices like surgical guides and anatomical models and does so within a controlled environment that has been registered with regulatory authorities including the US FDA to make medical devices. The company uses its specific variety of nylon because it is non-cytotoxic (not toxic to cells) and non-allergenic to the user.
According to Johnson, it is less about the 3D printing technology and more about the workflow, processing of parts and the management of that processing:
“What you see in [3D Systems’ Littleton Healthcare Technology Center] is a facility that has controls from everything on how you operate, the equipment, how you manufacture the parts, how you clean the parts, and how you package and ship the parts. And that’s the type of control that’s required for the manufacture of medical devices, like surgical masks and face masks that are compliant with standards. It’s less about the technology and more about how you deploy that technology to make medical devices that are safe and effective.”
Disinfection vs Sterilization: Most of the devices being made by the 3D printing community to not need to be sterilized, which involves placing parts into an autoclave and exposed to pressurized saturated steam at temperatures of approximately 121 °C (250 °F) for about 15–20 minutes. Additionally, all of the medical devices being printed in response to the virus, must be disinfected by healthcare staff, which can involve immersion in a solution of water and household bleach or wiping down with alcohol, but depends on the actual device itself. For instance, N95 masks degrade when exposed to alcohol, but could be disinfected using warm humid heat and ultraviolet germicidal irradiation or hydrogen peroxide vapor. Likewise, these kinds of processes when undertaken should not be improvised and be as tightly controlled as is possible.
For those wishing to provide supplies using 3D printing in the U.S., designs can be uploaded to the NIH 3D Print Exchange for validation, while manufacturers can visit the America Makes website to lend manufacturing capabilities. Hobbyists can work with #GetUsPPE to connect with hospitals. Johnson suggested that going through the America Makes/NIH/VA coalition in the U.S. might be the best way to participate because “files and designs will get tested and validated by the VA with America Makes helping to connect the dots between those in need and those that can provide. So it’s great for public visibility of a government-controlled type of forum for people to upload design files and for people to figure out how they can help.”
Menno Ellis, senior vice president and general manager of Plastics for 3D Systems, made the following suggestion of people hoping to lend their support to the supply efforts:
“I would impress upon people that as they consider making things, that they really look at the available documentation for their equipment and their materials or do research before they start printing things; particularly things that are going to come into direct contact with patients or care providers. There is a real risk of doing harm from items made by just grabbing something off the shelf and promoting it as a solution.”
We will be following up this article with an interview with a member of the Veterans Affairs team. We welcome all comments and criticism regarding the information provided here and look forward to expert readers to provide us with further information regarding the safety of 3D printed medical devices made in response to COVID-19-related supply shortages. Feel free to email the author at michael@3dprint.com.
Subscribe to Our Email Newsletter
Stay up-to-date on all the latest news from the 3D printing industry and receive information and offers from third party vendors.
Print Services
Upload your 3D Models and get them printed quickly and efficiently.
You May Also Like
Creality Marks 12 Years with KliTek and AI-Powered Ecosystem Expansion
For 12 years, Creality has advanced accessible 3D printing technologies, enabling global users to turn ideas into tangible creations. What began as a desktop 3D printer manufacturer has evolved into...
Asia AM Watch: Creality Launches $177M Hong Kong IPO as HKEX Trading Debut Nears
Creality has formally launched its Hong Kong initial public offering (IPO), marking one of the biggest public market moves by a 3D printing company in 2026 and offering a new...
How AtomForm’s 12-Nozzle System Cuts Multi-Color FDM Transition Waste by Up to 90%
Pull up the print stats on multi-color FDM jobs. The number that stings isn’t time; it’s material efficiency. On six-color models, single-nozzle systems consume significant filament during transition flushing, expelling...
3D Printed Weapons Keep Showing Up in Crime
In the past few weeks, activity around 3D printed weapons in the U.S. has increased across several fronts. States including California, Colorado, New York, and Washington are moving forward with...

















































