Companies, organizations and individuals continue to attempt to lend support to the COVID-19 pandemic supply effort. We will be providing regular updates about these initiatives where necessary in an attempt to ensure that the 3D printing community is aware of what is being done, what can be done and what shouldn’t be done to provide coronavirus aid.
HP has provided some interesting details to 3D Printing Media Network, about the extent to which it has been involved in manufacturing medical supplies. It has produced over a million parts for COVID-19 protection with Multi Jet Fusion, including over 200,000 face shields for Sant Pau Hospital in Barcelona. To do so, it has partnered with such large companies as Lamborghini, Volkswagen-Seat, Ferrari, Skoda. Among the parts printed is one that connects a continuous positive airway pressure (CPAP) device to an oxygen mask, as well as one that connects to a diving mask, various face masks and parts for the Leitat1 emergency ventilator. For the U.K. alone, HP and its partners have made more than 50,000 face shields, mask adjusters and hands-free door openers.
SPEE3D has begun deploying its antimicrobial ACTIVAT3D copper printed devices, starting with the Northern Territory Department of Trade, Business and Innovation which is replacing door handles with ACTIVAT3D copper handles. According to SPEE3D and its testing partner, these copper parts are able to kill 96% of SARS-CoV-2 virus within two hours of contact, compared to stainless steel which had no impact.
RIZE is also producing face shields for workers at Cardiovascular Medicine, PC, in Davenport IA and Moline IL, emphasizing the US Pharmacopeia Class VI certification its RIZIUM ONE filament has achieved. This is considered the highest level of biocompatibility, such that it has no adverse impact on the wearer’s skin after long periods of use. The company also claims that this material does not trap moisture that can contain the virus, like other common 3D printing plastics, such as ABS and nylon.
An Duong, a Rolls-Royce worker making face shields in the U.K. covered in a previous update, is now asking for help. Having donated over 700 masks to National Health Services staff, Duong is hoping for donated filaments and funds, via GoFundMe. In return, he is planning to send one donor, chosen via random name drawing, a printed Jet-M1 models.
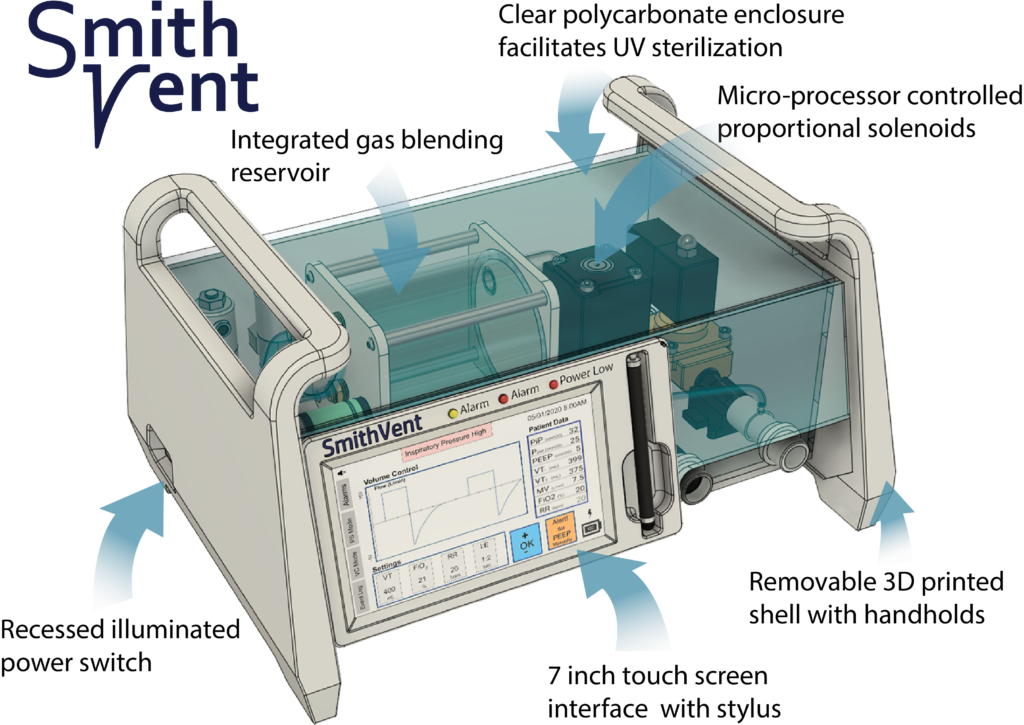
Stratasys has reported that over 200 teams have submitted designs for GrabCAD’s CoVent-19 Challenge and that the finalist round has begun, with seven teams selected to build working prototypes of rapidly deployable, minimum viable ventilators. After that, judging will proceed, led by 12 anesthesiology resident physicians from Massachusetts General Hospital. The finalist projects chosen are the following: SmithVent, CORE Vent, InVent Pneumatic Ventilator, RespiraWorks, OP Vent, Lung Evolve, and the Baxter Ventilator. A winner will be selected by the beginning of June, which will see them attempt to bring the device to market with FDA approval.
The Consumer Technology Association (CTA) and other trade groups are attempting to push the United States Trade Representative (USTR) Office to broaden tariff exemptions on products during the pandemic from imported ventilators, oxygen masks and nebulizers to include 3D printers, drones, robots and personal computers that are made in China.
The groups suggest that these technologies are important to managing the COVID-19 outbreak, with the CTA saying in a letter to the USTR Office, “These tariffs are not only a barrier to the entry of necessary products, they are a tax on businesses and consumers that has become ever more harmful as many enter ‘survival mode’.”
Dassault Systemés has published some of the efforts of its Open COVID-19 Community, including the work of Inali, a non-profit in India, that has developed what it suggests is a safe, affordable ventilator in only eight days. It should be noted that the device is of the Ambu bag variety, like the Leitat1, which can be dangerous due to the high incidence of delivering too much air to the patient and leading to aspirating vomit into their airways or lungs and even dying.
As the pandemic continues to grip the world, we will continue to provide regular updates about what the 3D printing community is doing in response. As always, it is important to keep safety in mind, remain critical about the potential marketing and financial interests behind seemingly good humanitarian efforts from businesses, and to do no harm.
Subscribe to Our Email Newsletter
Stay up-to-date on all the latest news from the 3D printing industry and receive information and offers from third party vendors.
Print Services
Upload your 3D Models and get them printed quickly and efficiently.
You May Also Like
Ceramitec 2026 3D Printing Wrap Up
I was surprised by just how many 3D printing companies and 3D printing projects there were at Ceramitec this year. I came away from the show convinced that the ceramics...
RusselSmith Brings Additive to Ghana’s Maritime Sector
Nigerian firm RusselSmith is in talks with Ghana’s Maritime Authority (GMA) to bring 3D printing to Ghana’s maritime sector. The Commonwealth Enterprise and Investment Council (CWEIC) helped make the discussion...
The Blueprint for Industrial Serial Production
Lithoz is kicking off the 2026 trade fair season with a powerful showcase of real-world applications of ceramic AM serial production capabilities alongside new material upgrades. The company is stepping...
Fraunhofer ILT Tests 3D Printed Titanium Reactors to Generate Hydrogen Onboard Heavy Vehicles
The Aachen-based Fraunhofer Institute for Laser Technology (Fraunhofer ILT) is to research titanium aluminide hydrogen reactors and heat exchangers. The hope is that lightweight, better-performing 3D printed components will make...