Handheld Bioprinter to Treat Skeletal Muscle Injuries
If having bioprinters available to medical practitioners and hospital workers could have a huge impact on their work, then just imagine what handheld bioprinters would do for the treatment of traumatic injuries. These new devices, a few of which we have encountered in the past couple of years, are not all that common and usually highly customized; however, researchers have realized how important they could be for trauma care. The idea behind handheld bioprinters is that they could deliver the technology straight to the patient, targeting the desired surface thanks to their mobility. In the past, we have reported several projects of handheld bioprinters delivering cells directly onto bone and cartilage during surgery, as well as to treat corneal ulceration, and mostly for healing burn wounds via cartilage and skin regeneration. The technology has great potential, which is why, earlier this year, a group of biomedical engineers from the University of Connecticut‘s School of Dental Medicine developed a handheld 3D bioprinter that could revolutionize the way musculoskeletal surgical procedures are performed.
The bioprinter enables surgeons to deposit scaffold materials to help support cellular and tissue growth directly into the defect sites within weakened skeletal muscles. Developed by Ali Tamayol, an associate professor at the biomedical engineering department at the University of Connecticut, the technology is capable of 3D in situ printing of adhesive scaffolds. The expert even considers it a paradigm shift in the rapid yet precise filling of complex skeletal muscle tissue defects.

In situ bioprinting of cell-laden GelMA hydrogels for the treatment of VML injuries (Image: ACS Appl. Bio Mater. 2020, 3, 3, 1568-1579)
Tamayol’s research was recently published in the American Chemical Society journal, in an article titled “In Situ Printing of Adhesive Hydrogel Scaffolds for the Treatment of Skeletal Muscle Injuries.” According to the investigators, current methods for reconstructive surgery have been largely inadequate in treating volumetric muscle loss (the traumatic or surgical loss of skeletal muscle which results in functional impairment), adding to the fact that the geometry of skeletal muscle defects in this type of injuries varies on a case-by-case basis. They consider that as a result, 3D printing technology has emerged as an up and coming solution to help reconstruct muscle. However, they also claim that the time and facilities needed for imaging the defect site, processing to render computer models, and print a suitable scaffold prevent immediate reconstructive interventions post-traumatic injuries.
To overcome these challenges, this new research proposes that gelatin-based hydrogels are printed directly into the defect area and cross-linked on site.
“The printer is robust and allows proper filling of the cavity with fibrillar scaffolds in which fibers resemble the architecture of the native tissue,” indicated Tamayol. “This is a new generation of 3D printers than enables clinicians to directly print the scaffold within the patient’s body, and best of all, this system does not require the presence of sophisticated imaging and printing systems.”
The handheld, partially automated, bioprinter is an extrusion-based device capable of continuously extruding biomaterials and includes an integrated ultraviolet (UV) light source for cross-linking of the extruded bioink capable of in situ printing of adhesive scaffolds in order to overcome the challenges associated with the treatment of a volumetric muscle loss injury. The researchers claim that the platform can print photo-cross-linkable hydrogels such as gelatin methacryloyl (GelMA) for this type of injuries immediately. GelMA is a collagen-derived biomaterial that closely mimics the extracellular matrix (ECM) of native skeletal muscles, furthermore, GelMA adheres to body tissues and has been used as a bioadhesive, yet they indicated that they are the first to investigate the adhesion of GelMA hydrogels to skeletal muscle.
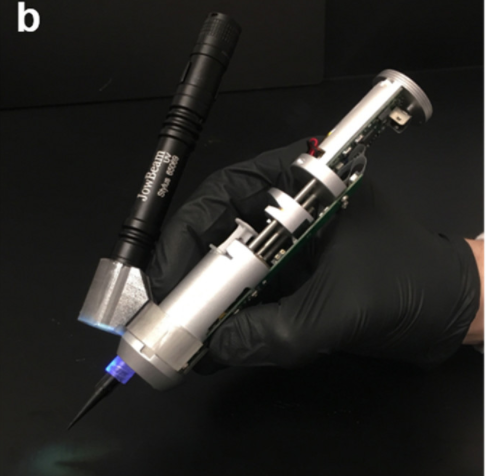
The handheld 3D bioprinter equipped with a UV light source for in situ cross-linking of the printed scaffolds (Image: ACS Appl. Bio Mater. 2020, 3, 3, 1568-1579)
The study suggests that in situ printing of GelMA is expected to eliminate the need for additional surgeries and solve the challenges of hydrogel-based scaffold implantation, and the scaffolds from the bioprinter adhere precisely to the surrounding tissues of the injury and mimic the properties of the existing tissue, eliminating the need for any suturing.
Indranil Sinha, a co-author and plastic surgeon at Brigham and Women’s Hospital (a Harvard Medical School Teaching Hospital) with expertise in treating muscle injuries, said: “A good solution currently does not exist for patients who suffer volumetric muscle loss. A customizable, printed gel establishes the foundation for a new treatment paradigm that can improve the care of our trauma patients.”
It is clear from the published material that implanting the hydrogel-based scaffolds successfully requires a very specific biomaterial to be printed that will adhere to the defect site. And while bioprinted scaffolds mimicking skeletal muscles have been created in vitro, the researchers suggest that these have not been successfully used on an actual subject, leaving the existing 3D bioprinting technology with a few problems.
Instead, Tamayol’s solution fixes the problem, since the successfully printed gelatin-based hydrogel bioink effectively adhered to defect sites when tested on mice with volumetric muscle loss injury. In fact, the mice showed a significant increase in muscle hypertrophy following Tamayol’s therapy, that is, an increase and growth of muscle cells.
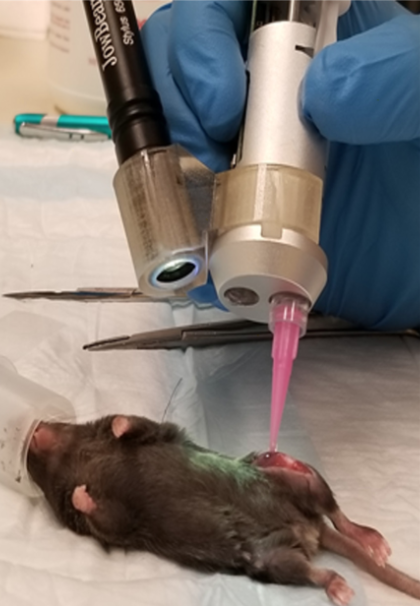
Surgical implantation of GelMA hydrogels via in situ printing into mice with a volumetric muscle loss injury (Image: ACS Appl. Bio Mater. 2020, 3, 3, 1568-1579)
The team of 15 experts was able to prove that this bioprinter is compatible with various types of bioinks, including photo-cross-linkable hydrogels and polymeric solutions conventionally used in tissue engineering and bioprinting, and that the bioprinter enabled printing on nonflat surfaces, which cannot be achieved using regular stationary bioprinters. They also tested the feasibility of in situ printing of GelMA hydrogel for the treatment of muscle injuries and realized that the printing pressure and applied shear stress using the handheld bioprinter had no adverse impact on the viability and proliferation of myoblasts (the embryonic precursors of myocytes, also called muscle cells).
The research, funded by the National Institutes of Health and The Gillian Reny Stepping Strong Center for Trauma Innovation, proved that the handheld printer will change the rapid yet precise filling of complex skeletal muscle tissue defects.
Although the new system is in the early stages of testing, it may eventually provide a way to treat patients with serious injuries. Tamayol and Sinha have already filed a patent on this technology for the treatment of musculoskeletal injuries. And as part of his profound interest in the field, Tamayol also recently developed a smart bandage to help clinical care for people with chronic wounds. It seems that the suitability of the in situ printed bioink for the delivery of cells was successful when directly printed into the defect site of mice with volumetric muscular loss injury, let’s hope it can also work in the future to promote skeletal muscle growth in humans with this kind of traumatic injuries.
Subscribe to Our Email Newsletter
Stay up-to-date on all the latest news from the 3D printing industry and receive information and offers from third party vendors.
Print Services
Upload your 3D Models and get them printed quickly and efficiently.
You May Also Like
3D Printing News Briefs, May 30, 2026: RIMPAC 2026, Acquisition, Ceramic Implants, & More
We’re kicking things off with materials news in this weekend’s 3D Printing News Briefs. Then it’s on to a hybrid manufacturing system for a maritime exercise, an expansion of industrial...
The Hidden Cost of Lost Engineering Intent
Recent reporting on the UPS MD-11 engine separation on Flight 2976 has understandably focused on the immediate question: what happened? The investigation will determine that, as it should. Serious engineering...
Scientists Create Stretchy 3D Printed Implants for High Blood Pressure Treatment
Researchers at Pennsylvania State University (Penn State) say they may have found a softer, less invasive way to treat severe high blood pressure. In a new study published in the...
Conexeu Sciences Wishes to Make a Regenerative Breast Matrix Solution (and sell shares)
Conexeu Sciences wants to commercialize a regenerative breast matrix solution. For years, Australian firm BellaSeno and Lattice Medical have been working on similar solutions. In 2018, Lattice came out with...

















































