 It’s hard to keep up with 3D bioprinting technology. What seemed, not that long ago, like a futuristic dream is now advancing at an incredible speed. Numerous organizations, from corporations to universities, are developing their own variations on the technology, each slightly different, but all aimed at the same goal – to 3D print viable human organs. The majority of the cells and tissue developed so far remains in the laboratory, used for pharmaceutical testing and further study, but we’re getting closer and closer to being able to implant 3D printed tissue into the human body.
It’s hard to keep up with 3D bioprinting technology. What seemed, not that long ago, like a futuristic dream is now advancing at an incredible speed. Numerous organizations, from corporations to universities, are developing their own variations on the technology, each slightly different, but all aimed at the same goal – to 3D print viable human organs. The majority of the cells and tissue developed so far remains in the laboratory, used for pharmaceutical testing and further study, but we’re getting closer and closer to being able to implant 3D printed tissue into the human body.
While the field may be advancing quickly, the process of creating 3D printed tissues and cells in the lab is a relatively slow and deliberate one. A person unfamiliar with the technology may picture doctors pulling out a 3D printer, turning it on, and – voila! – a 3D printed ear! In reality, 3D printed cells must be carefully cultivated and allowed to grow into viable tissue in a process that takes days at least. But a new development by researchers based at Australia’s University of Wollongong could speed that process up – with a tool that, in essence, is a biological 3Doodler.
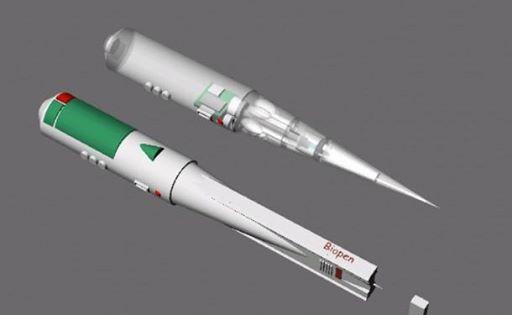
The BioPen arose out of a collaboration between researchers at the UoW-based Australian Research Council Centre of Excellence for Electromaterials Science (ACES) and orthopedic surgeons at St. Vincent’s Hospital Melbourne. The device would allow surgeons to repair damaged bone and cartilage by “drawing” new cells directly onto bone in the middle of a surgical procedure. A team led by ACES Director Professor Gordon Wallace developed the pen, which was then transferred to St. Vincent for researchers to work on optimizing it for clinical trials.
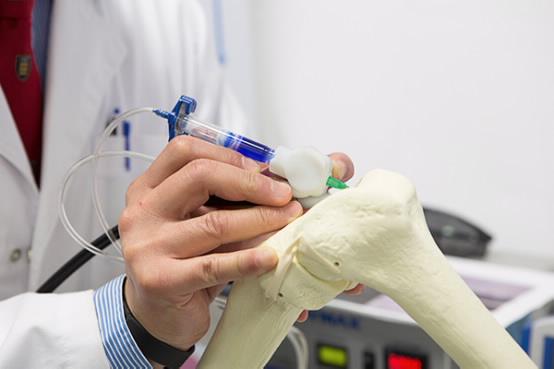 How it works: the pen is loaded with a bio-ink comprised of stem cells inside a biopolymer such as alginate, a seaweed extract, which is in turn protected by a second layer of hydrogel. The ink is then extruded onto the bone surface and solidified by a UV light embedded in the pen. Once they are drawn onto the bone, they will multiply inside the patient’s body, differentiating themselves into nerve, muscle and bone cells and eventually growing into tissue.
How it works: the pen is loaded with a bio-ink comprised of stem cells inside a biopolymer such as alginate, a seaweed extract, which is in turn protected by a second layer of hydrogel. The ink is then extruded onto the bone surface and solidified by a UV light embedded in the pen. Once they are drawn onto the bone, they will multiply inside the patient’s body, differentiating themselves into nerve, muscle and bone cells and eventually growing into tissue.
The technique could revolutionize how surgeons repair cartilage, in particular. For certain types of injuries, it’s difficult or impossible for surgeons to discern the exact shape of the area requiring an implant, making it extremely difficult to design an artificial cartilage implant before surgery. With the BioPen, surgeons could simply fill in the damaged area with the hydrogel solution.
“This type of treatment may be suitable for repairing acutely damaged bone and cartilage, for example from sporting or motor vehicle injuries,” said Professor Peter Choong, Director of Orthopaedics at St. Vincent’s. “Professor Wallace’s research team brings together the science of stem cells and polymer chemistry to help surgeons design and personalise solutions for reconstructing bone and joint defects in real time.”
The cell solution could also be further customized by adding drugs to boost healing and regrowth. The BioPen prototype was 3D printed in medical grade plastic and titanium to be lightweight and easy to sterilize.
“The combination of materials science and next-generation fabrication technology is creating opportunities that can only be executed through effective collaborations such as this,” said Professor Wallace. “What’s more, advances in 3D printing are enabling further hardware innovations in a rapid manner.”
So far, cells produced by the BioPen have shown a survival rate of over 97%. The full research study has recently been published in the journal Biofabrication.
Below, Professor Choong demonstrates how the BioPen works. What do you think of the technology? Do you think it will catch on with surgeons? Discuss in the 3D Printing BioPen forum over at 3DPB.com.
Subscribe to Our Email Newsletter
Stay up-to-date on all the latest news from the 3D printing industry and receive information and offers from third party vendors.
You May Also Like
Profiling a Construction 3D Printing Pioneer: US Army Corps of Engineers’ Megan Kreiger
The world of construction 3D printing is still so new that the true experts can probably be counted on two hands. Among them is Megan Kreiger, Portfolio Manager of Additive...
US Army Corps of Engineers Taps Lincoln Electric & Eaton for Largest 3D Printed US Civil Works Part
The Soo Locks sit on the US-Canadian border, enabling maritime travel between Lake Superior and Lake Huron, from which ships can reach the rest of the Great Lakes. Crafts carrying...
Construction 3D Printing CEO Reflects on Being Female in Construction
Natalie Wadley, CEO of ChangeMaker3D, could hear the words of her daughter sitting next to her resounding in her head. “Mum, MUM, you’ve won!” Wadley had just won the prestigious...
1Print to Commercialize 3D Printed Coastal Resilience Solutions
1Print, a company that specializes in deploying additive construction (AC) for infrastructure projects, has entered an agreement with the University of Miami (UM) to accelerate commercialization of the SEAHIVE shoreline...































