3D Printing Multi-Drug Delivery Systems for Improved Gastrointestinal Health
International researchers are looking into ways to promote better human health by improving gastrointestinal health, outlining their findings in the recently published ‘A core-shell multi-drug platform to improve gastrointestinal tract microbial health using 3D printing.’
3D printing is connected with the medical field today in countless ways as researchers and manufacturers continue to innovate, encouraged by the potential for an infinite number of designs to allow for medical models and instruments, devices such as implants and prosthetics, and accompanying software, hardware, and materials. In this study, the authors branch out into a more unique and complex area of medicine, creating core-shell structure fibers via electrohydrodynamic 3D printing.
These types of fibers can be fabricated in a variety of shapes and sizes, and have to ability to deliver and release drugs; in fact, the researchers state that the biphasic drug release profile in simulated intestinal fluid offers ‘a burst release within the first 12 h and a slower sustained release up to 72 h.’
Bacteria living in the gastrointestinal tract can be both good and bad. It may be responsible for providing vitamins, nutrients, and digestion, but in turn, can also be hazardous to human health when there is an imbalance—causing a range of health issues from obesity to diabetes, and even worse—cancer. With suitable probiotics, however, and resulting gastrointestinal health, many patients see their health improve overall.
The fibrous membranes used in this study were successful in showing the potential for improved gastrointestinal health, coupled with the electrohydrodynamic (EHD) 3D printing. The authors chose this technique due to the following advantages:
- Enabling of micro to nanoscale fiber engineering
- Control and development of complex structures
- Precise printing of stacked and aligned fibers
- Personalized drugs can be created
“… cellulose acetate (CA) and polyacrylic resin II (PRII) were used as excipient materials of enteric-specific medicine due to its good biocompatibility [29], nontoxicity [28], and pH dependent [30] characteristics,” explained the authors. “The CA encapsulating stachyose (CA-STA) was used as shell while PRII loading proteoglycan (PRII-PRO) was used to form the core of the fibrous structures.”
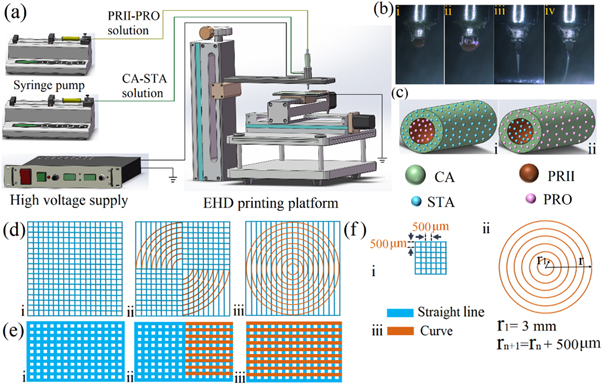
Schematic diagram of core–shell EHD 3D printing system (a); forming process of continuous core–shell stable-jet mode at different applied voltages (b): i at 0 kV, ii at 1 kV, iii (front view) at 2.5 kV and iv (side view) at 2.5 kV when fiber was printed at 20 mm s−1, respectively; core-shell fiber with two types of drug-loading models (c): i—PRII-PRO/CA-STA, ii—PRII-ST/CA-PRO A; top view of patterned fibrous membrane with various geometries (d): i—square, ii—semi-circle, iii—whole-circle; the front view of the patterned membrane (e) corresponding to i, ii, and iii of (d); characteristics of membrane (f): i, the space between two printed fibers; ii, the radius of curves, and iii was the printed fiber with straight line and/or curve.
The EHD printer was comprised of:
- Programmable X-Y-Z movement stage
- High power voltage supply
- Two syringe pumps
- Coaxial stainless-steel nozzle (inner and outer needles)
For the study, the authors created a square grid, semi-circle, and a whole circle using 10-layer deposition with an average thickness of 61.5 ± 2.6 μm. The printed fibers were examined with optical microscopy, as the team replaced PRO with Rhodamine B into the loaded PRII-core layer. Diameters of the inner and outer shells were 10.8 ± 3.7 μm and 61.2 ± 6.2 μm.
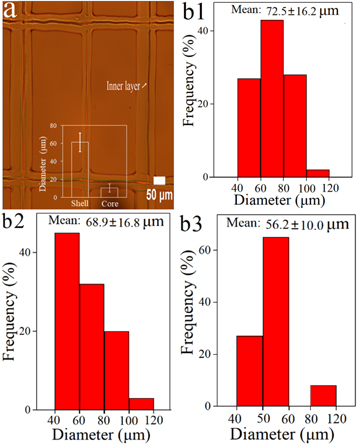
The optical images of printed fibrous membrane with square geometry (a); diameter distribution of fibers with various geometries (b1) square, (b2) semi-circle, and (b3) whole-circle.
The enteric capsule, tested a delivery system for oral drugs, was comprised of the sample fibers.
“The in vitro drug release was carried using PBS solution (pH = 7.4) to mimic intestinal fluid,” explained the researchers. “The LC for STA and PRO was 3.67 ± 0.1% and 1.1 ± 0.02%, respectively; and the EE was 90.3 ± 1.7% and 95.3 ± 0.5%, respectively. This confirms each drug was efficiently loaded into outer layer and inner layer, respectively.”

Optical images (a1)–(c1) and scanning electron images (a2)–(c2) of printing fibers loading STA and PRO with various patterns: (a1) and (a2), square; (b1) and (b2), semi-circle; (c1) and (c2), whole-circle.
For each sample, thickness was measured before tensile testing was performed, noted respectively for the square, semi-circle, and whole-circle membranes at 64.4 ± 2.6 μm, 61.9 ± 4.2 μm, and 58.1 ± 2.5 μm.
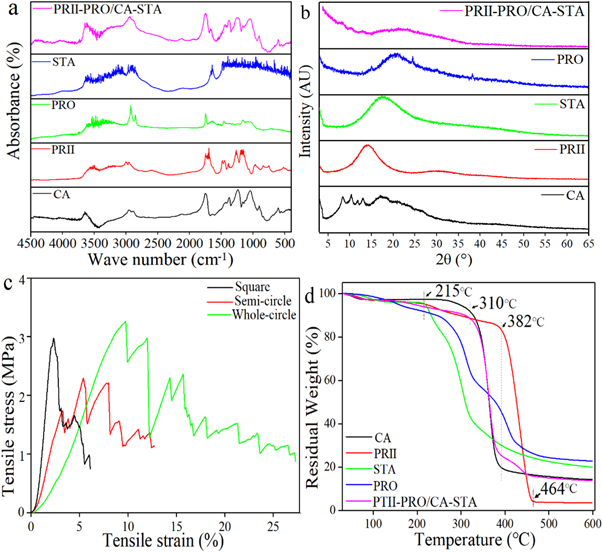
FT-IR spectra (a), XRD spectra (b), tensile strain (c) and TGA results (d) of printed fibers and raw materials.
Because zig-zag patterns were prominent in the membrane sandwich structure, the research team notes them as critical elements in deciding mechanical properties of the 3D printed samples. Tensile strengths and broken strains increased, however, as the circle proportion increased—attributed to circular fibers transforming into a straight line when under the pressure of tensile force. Maximum tensile strength was also noted as ‘greater than 0.22 MPa,’ the gastrointestinal pressure of in humans.
“The printed drug-loaded membrane possessed good biocompatibility on L929 cells and excellent improvement effects on Bifidobacterium bifidum, alongside adequate inhibition effects on E. coli. Compared to the control, the proliferation ratio of Bifidobacterium bifidum can be raised to 294.2% and 242.7%, respectively, at the treatment of 0.1 g PRII-STA/CA-PRO and PRII-PRO/CA-STA,” concluded the researchers.
“The inhibition effects on E. coli was 37.0% and 27.5%, at the treatment of 0.1 g PRII-STA/CA-PRO and PRII-PRO/CA-STA, respectively. This result confirmed the feasibility of improving proliferation of probiotics (Bifidobacterium bifidum) combining with inhibiting growth of pathogenic (E. coli) in a single dosage for using dual drug loading. The core–shell structures developed here can be extremely advantageous in efficient therapy for gastrointestinal conditions.”
What do you think of this news? Let us know your thoughts! Join the discussion of this and other 3D printing topics at 3DPrintBoard.com.

(a) Water contact angle images of fibrous membrane loading drug in the manner of PRII-PRO/CA-STA at various time point: 0 s, 30 s and 60 s, (first column in figure 5(a): top view of the patterned membrane with various geometries; red dot presents the position of water drop for water contact angle test); (b) figure (a) expressed with a column chart.
Subscribe to Our Email Newsletter
Stay up-to-date on all the latest news from the 3D printing industry and receive information and offers from third party vendors.
Print Services
Upload your 3D Models and get them printed quickly and efficiently.
You May Also Like
HP’s MJF 1200 Targets Entry-Level AM — And Could Shift the Competitive Landscape
HP has launched an entry-level MJF solution, the HP Multi Jet Fusion 1200 3D Printer Solution, which will be available in 2027. What is the 1200 exactly? What does it...
Metal Base & Scrap Labs Usher in an Ultra-Low-Cost Sub-$10,000 Metal LPBF Evolution
Above, an image of a part coming off the ScrapLabs Scrap 1. Dutch firm Metal Base started a low-cost metal LPBF revolution last month. An ultra-low-cost metal laser powder bed...
Most 3D Printing Metals Are Adapted. This One Was Designed with AI
A research team from the University of South China and Purdue University developed a new type of steel designed specifically for 3D printing, using machine learning to guide the process....
Nikon AM Synergy Gets Defense Innovation Unit FORGE Contract
Nikon AM Synergy has received an Other Transition Agreement (OTA) contract from the U.S. Department of War (DoW) Defense Innovation Unit (DIU). The Foundry for Operational Readiness and Global Effects...










































