Wake Forest Researchers Claim to Bioprint Skeletal Muscle Constructs With Neural Cell Integration, in Rats
 Scientists at Wake Forest Institute for Regenerative Medicine (WFIRM), in North Carolina, have found a way to advance the 3D bioprinting technique they developed to engineer skeletal muscle as a potential therapy for replacing diseased or damaged muscle tissue, moving another step closer to someday being able to treat patients.
Scientists at Wake Forest Institute for Regenerative Medicine (WFIRM), in North Carolina, have found a way to advance the 3D bioprinting technique they developed to engineer skeletal muscle as a potential therapy for replacing diseased or damaged muscle tissue, moving another step closer to someday being able to treat patients.
Skeletal muscles are responsible for moving the body. So when they are damaged, such as due to blunt trauma, combat injuries or burns, they are not regenerated but instead require reconstructive surgery and are replaced with muscle graft, usually using connective tissue or adipose tissue, which do not possess the contractile abilities of muscle tissue, so over time muscles atrophy and these muscle cells die. There have been various attempts to recover muscles, such as through cell therapy and the use of various biomaterials, however, none promote skeletal muscle repair and functional regeneration. According to WFIRM, going into the study, the researchers knew that effective nerve integration of bioengineered skeletal muscle tissues has been a challenge, but they are now giving new hope to patients.
In their recent paper called “Neural cell integration into 3D bioprinted skeletal muscle constructs accelerates restoration of muscle function“, Ji Hyun Kim, Ickhee Kim, Young-Joon Seol, In Kap Ko, James J. Yoo, Anthony Atala, and Sang Jin Lee published a study which investigates the effects of neural cell integration into the bioprinted skeletal muscle construct to accelerate functional muscle regeneration in vivo. That is, they applied the bioprinted constructs in a rat model with a defect injury of the tibialis anterior (TA) muscle and evaluated the functional outcomes of muscle tissue reconstruction and innervation.
“Being able to bioengineer implantable skeletal muscle constructs that mimic the native muscle to restore function represents a significant advance in treating these types of injuries,” said lead author Ji Hyun Kim, an instructor of WFIRM. “Our hope is to develop a therapeutic option that will help heal injured patients and give them back as much function and normalcy as possible.”
WFIRM acknowledges that institute scientists had previously demonstrated that the Integrated Tissue and Organ Printing System (ITOP), developed in house over a 14-year period, can generate organized printed muscle tissue that is robust enough to maintain its structural characteristics. Since then, the researchers have been developing and testing different types of skeletal muscle tissue constructs to find the right combination of cells and materials to achieve functional muscle tissue. In the current study, they investigated the effects of neural cell integration into the bioprinted muscle construct to accelerate functional muscle regeneration.
In this particular study, the muscle defect injury in the rodent was created by removing 40% of the TA muscle mass and other injuries that caused irreversible anatomical and functional deformity following six months post-injury. After creating the TA muscle defect, they implanted the bioprinted skeletal muscle constructs with muscle progenitor cells (MPC) into the defect sites for anatomical and functional skeletal muscle regeneration.
The team used its own custom bioprinting system to fabricate the skeletal muscle constructs. They describe the system as consisting of four dispensing modules (including cooling and heating units), a pneumatic pressure controller, XYZ stage and controller, a temperature controller, and a humidifier in a closed chamber. They used cell-laden hydrogel, acellular sacrificing hydrogel, and supporting polymer for the 3D skeletal muscle constructs, by loading the bioinks into different sterile syringes.
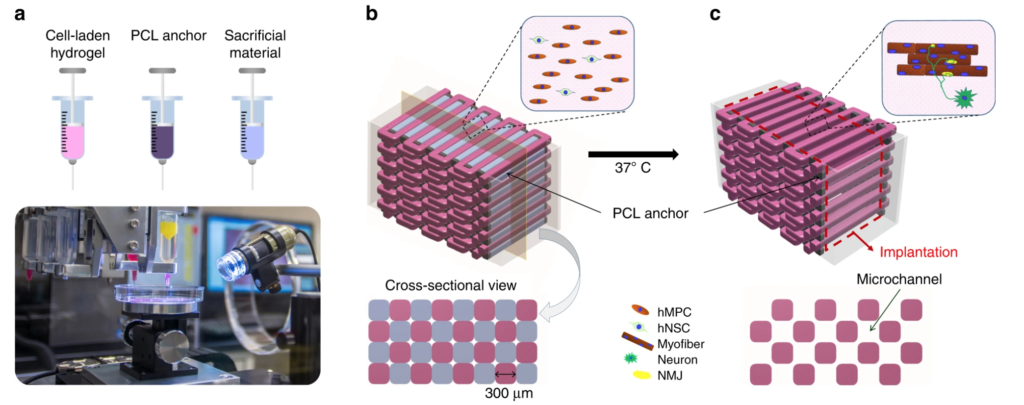
Bioprinting of human skeletal muscle constructs: three components were printed in a layer-by-layer fashion using WFIRM’s custom 3D bioprinting system (Image: WFIRM)
Study results state that the neural input into the bioprinted skeletal muscle construct shows the improvement of myofiber formation (a multinucleated single muscle cell), long-term survival, and neuromuscular junction formation in vitro. Moreover, they claim that the bioprinted constructs with neural cell integration facilitate rapid innervation and mature into organized muscle tissue that restores normal muscle weight and function in the rodent model. Also they suggest that the 3D bioprinted human neural-skeletal muscle constructs can be rapidly integrated with the host neural network, resulting in accelerated muscle function restoration.
The director and co-senior author of the paper, Anthony Atala, indicated that the “continued improvements in 3D bioprinting techniques and materials are helping us advance in our quest to make replacement tissue for patients.”
Atala and his team of scientists have been first in the world to implant lab-grown tissues and organs into patients. Starting in 1990 with most of their research and implanting the first structures at the end of that decade, using a 3D printer to build a synthetic scaffold of a human bladder, which they then coated with cells taken from their patients. In fact, Wake Forest’s WFIRM has been a pioneer in bioprinting for regenerative medicine for the past decades, growing tissues and organs and developing healing cell therapies for more than 40 different areas of the body, from kidney and trachea to cartilage and skin, and successfully implanting living, functional 3D printed human tissue into animals.
This particular ongoing line of research at WFIRM is supported and federally funded through the Armed Forces Institute of Regenerative Medicine, which, in turn, was supported by the U.S. Army, U.S. Navy, National Institutes of Health (NIH), U.S. Air Force, Veterans Administration, and partially by a Medical Technology Enterprise Consortium grant.
The team’s goal is to develop clinical therapies to treat wounded soldiers as well as benefiting the general population. Soldiers pose a particular challenge, considering that combat injuries are quite severe and wound treatment is in high demand, especially with so many conflict areas to watch out for in 2020, while Syria, Afghanistan, and Iraq are well known, a few other new ones might arise, and that means more injured soldiers.
In the paper, the team of researchers concluded that “the neural cell component can support the long-term cell survival, enhance myogenic differentiation, and induce neuromuscular junctions (NMJ) formation on the muscle fibers in the bioprinted skeletal muscle constructs in vitro, resulting in rapid restoration of muscle function in the rat TA muscle defect model.” They consider that with further advances, the NMJ preformed bioengineered skeletal muscle construct may be an effective therapeutic approach for repairing extensive skeletal muscle defect injuries with accelerated innervation capacity.
“These constructs were able to facilitate rapid nerve distribution and matured into organized muscle tissue that restored normal muscle weight and function in a pre-clinical model of muscle defect injury,” considered Sang Jin Lee, co-senior author and associate professor of WFIRM.
The WFIRM is advancing into novel bioprinting research that could deal with many of the problems that are common today, such as severe muscle injury, which is among the most common affliction in orthopedic practices around the world. Researchers are fighting against failure to gain functional recovery by using bioprinting technology which was developed on-site at their own labs to discover new ways to treat patients. With a successful in vivo trial, let’s hope the next stages bring the team one step closer to creating a solution for the many patients suffering from the condition.
Subscribe to Our Email Newsletter
Stay up-to-date on all the latest news from the 3D printing industry and receive information and offers from third party vendors.
Print Services
Upload your 3D Models and get them printed quickly and efficiently.
You May Also Like
AMPulse Asia: Creality IPO Headlines APAC 3D Printing Market Roundup
Asia’s additive manufacturing sector spent the back half of May moving capital and capacity, not just demos. Chinese desktop and consumer printer makers pushed onto public markets, metal powder producers...
Blue Origin’s New Glenn Explosion Comes During Major Manufacturing Push
Blue Origin‘s orbital New Glenn rocket exploded during a hot-fire test at Launch Complex 36 in Cape Canaveral on May 29, setting back the company’s launch ambitions at a time...
Aibuild Says New FETS Simulation Tool Is 10,000x Faster for AM
Aibuild has launched FETS for Additive Manufacturing, a Finite Element Thermomechanical Simulation tool that lets you simulate stress, distortion, thermal effects, and thermomechanical effects. The solution has been optimized for...
AI CAD Tools for 3D Printing: An Overview
There is a bevy of AI-to-CAD tools coming out. Some are finding users; some are raising millions in funding. Many new ones are coming out all the time, so we...


















































