China: Zhejiang University Researchers Evaluate Trends in Bioink Printability
 As Chinese researchers from Zhejiang University point out in their recently published article, ‘Trends on physical understanding of bioink printability,’ the future of bioprinting is only as promising as the available materials. Authors Jun Yin, Dengke Zhao, and Jingyi Liu state that even with the increasing popularity of bioprinting among the scientific research and medical community, the expansion of new bioinks—and especially the development of them as raw materials—is being ‘largely ignored.’
As Chinese researchers from Zhejiang University point out in their recently published article, ‘Trends on physical understanding of bioink printability,’ the future of bioprinting is only as promising as the available materials. Authors Jun Yin, Dengke Zhao, and Jingyi Liu state that even with the increasing popularity of bioprinting among the scientific research and medical community, the expansion of new bioinks—and especially the development of them as raw materials—is being ‘largely ignored.’
Such efforts are required to propel bioprinting further, and the scientists emphasize previous work by Huang’s research group at University of Florida (Appl. Phys. Rev. 2018, 5, 041304) as it offers a good understanding of details surrounding bioink and its printability issues today. Bioprinting is showing itself to be useful in applications beyond just growth of tissue in the lab, to include success in regenerative medicine, drug screening, and physiological modeling.
Raw materials include:
- Living cells
- Extracellular matrix materials
- Cell media
- Other additives
Again, the researchers emphasize that much more attention must be paid to printability issues, focusing on droplet-based bioprinting and filament-based bioprinting.
“For Newtonian bioinks, the successful droplet formation usually consists of liquid ejection and stretching, breakup/pinch-off, contraction and breakup of tails, and recombination of primary and satellite droplets. For non-Newtonian bioinks, four breakup types, namely front pinching, hybrid pinching, exit pinching, and middle pinching, have been identified based on the first pinch-off location.
“Moreover, cell-laden bioink has a smaller ejected fluid volume, lower droplet velocity, and longer breakup time compared to a hard bead (polystyrene)-laden suspension.”
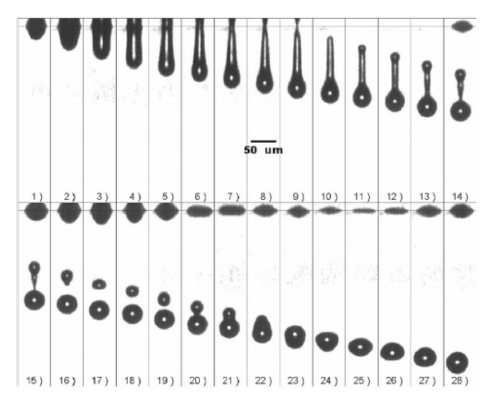
Figure 1 – Representative images of successful droplet formation process during DOD inkjet printing of Newtonian glycerol solutions [4].
Reprinted with permission from Ref. [4], Copyright 2006, American
Institute of Physics
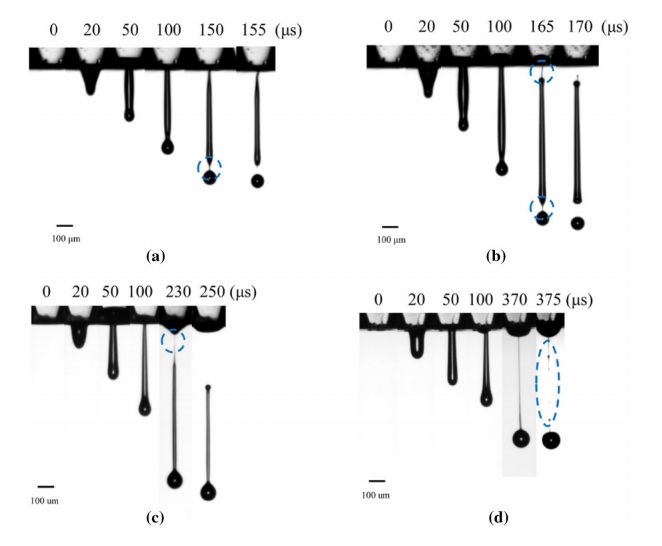
Figure 2A. Four pinch-off types during DOD printing of alginate solutions: a. front pinching, b. hybrid pinching, c. exit pinching, and d. middle pinching [5]. Reprinted with permission from Ref. [5], Copyright 2017, American Chemical Society
“The Z number (the inverse of Ohnesorge number) and capillary number (Ca) were used along with the Weber number (We) to capture printing conditions’ infuence by phase diagrams of Newtonian bioinks (Fig. 3). The Weissenberg (Wi) and Deborah (De) numbers were utilized for the evaluation of non-Newtonian bioinks printability in the phase diagram (Fig. 4).”

Figure 3 – Example phase diagram during DOD inkjet printing of particle-filled inks [6]. The solid curves indicate contours of equal maximum droplet spreading. The dotted lines indicate the boundaries of fluid properties typical of inkjet printers, and the bold line in the upper-right corner indicates the onset of splashing. Reprinted with permission from Ref. [6], Copyright 2003, Cambridge University Press
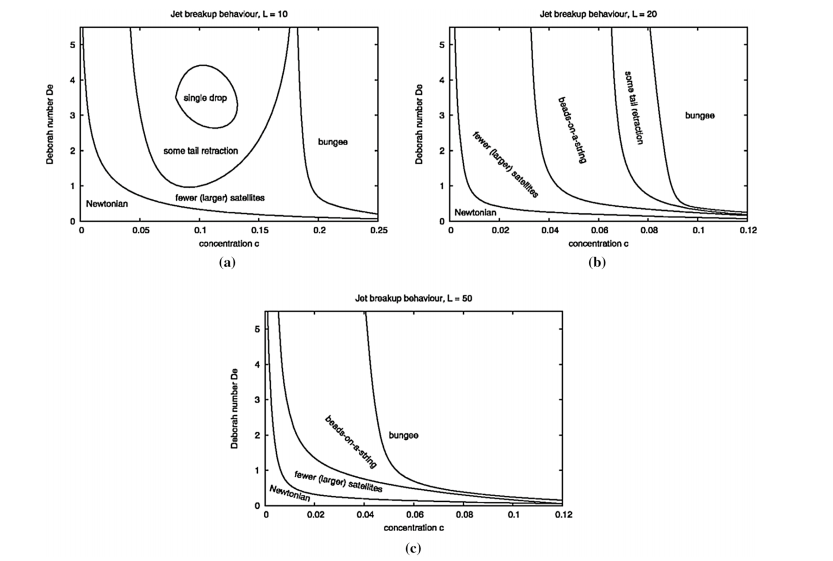
Figure 4 – Phase diagrams gained via numerical simulations showing the jet breakup behavior in different regions of the viscoelastic parameter space, aL = 10, bL = 20, cL = 50. In which, c and L represents the concentration and molecule extensibility of non-Newtonian inks, respectively [7]. Reprinted with permission from Ref. [7], Copyright 2010, Springer Nature
“During self-supporting rapid solidification printing, operating conditions usually determine the flament size and printing resolution, including nozzle diameter, dispensing pressure, pathway height, path width, and print speed.”
The team assessed seven different types of filament for extrusion printing, and illustrated printability with dimensionless ratios and storage modulus ratio—along with the Oldroyd number demonstrating yield area dimensions.
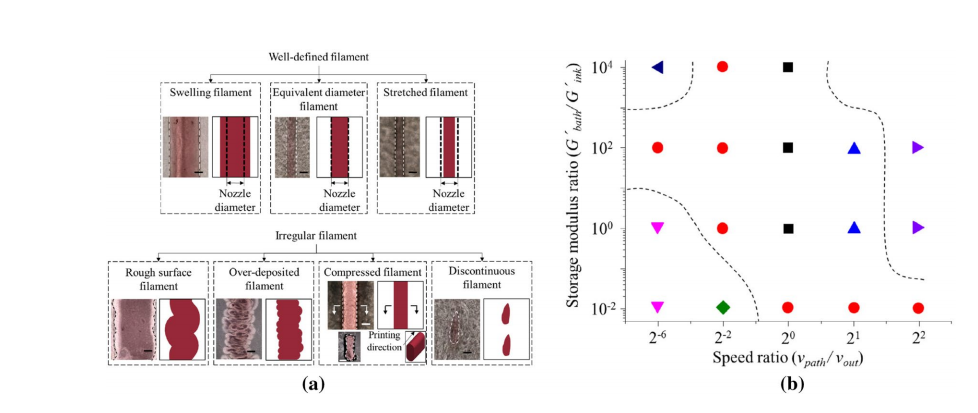
Fig. 5A Different types of flaments during support bath-enabled printing. b Phase diagram during support bath-enabled printing [8]. Reprinted with permission from Ref. [8], Copyright 2017, Elsevier
The study of materials in 3D printing may be much more fascinating you ever realized, mainly due to the potential for so many different types of innovation. While a variety of polymers, metals, and alternative materials are available, the study of bioinks continues to grow along with the science of bioprinting, leading to incredible strides in the medical field. Find out more about current advancements in bioink here.
Discuss this article and other 3D printing topics at 3DPrintBoard.com.
[Source / Images: Trends on physical understanding of bioink printability]Subscribe to Our Email Newsletter
Stay up-to-date on all the latest news from the 3D printing industry and receive information and offers from third party vendors.
Print Services
You May Also Like
New Business: Temporary, Migratory, & Modular 3D Printed Architecture
If we look at potentially emerging 3D printing businesses, then architecture has not been fully explored. Yes, there is a lot of house 3D printing going on worldwide. From deployable...
3D Printing News Briefs, April 19, 2025: Material Extrusion Standard, Metal Powder, & More
In today’s 3D Printing News Briefs, we’re covering a proposed standard for material extrusion, before moving on to business and metal powder. We’ll end with a commercial store’s robotic 3D...
Japan Unveils World’s First 3D Printed Train Station
Japan is now home to what we believe is the world’s first train station built with 3D printing technology. Located in Arida City, just south of Osaka, the new Hatsushima...
restor3d Raises $38M to Expand 3D Printed Orthopedic Implants
Backed by $38 million in new funding, restor3d is pushing ahead with the launch of four personalized implant lines, set to roll out in 2025 and 2026. This latest venture...

























