Organovo Explains Why They’ve Raised an Additional $40M – Cites Potential for $400M in Annual Revenue
 As we’ve mentioned in the past, the rollercoaster ride, known all too well to Organovo (NYSEMKT: ONVO) shareholders, has been quite bumpy. The stock price has dropped considerably following an announcement on June 18 that the company was raising an additional $40 million in a secondary offering valued at just $4.25 per share. While dilution oftentimes is viewed by current shareholders as a negative action, ultimately Organovo views this move as one which will help the company prosper in many ways moving forward.
As we’ve mentioned in the past, the rollercoaster ride, known all too well to Organovo (NYSEMKT: ONVO) shareholders, has been quite bumpy. The stock price has dropped considerably following an announcement on June 18 that the company was raising an additional $40 million in a secondary offering valued at just $4.25 per share. While dilution oftentimes is viewed by current shareholders as a negative action, ultimately Organovo views this move as one which will help the company prosper in many ways moving forward.
Likely stemmed by the recent drop in their share price, the company has put forth a rather detailed explanation for this secondary offering, explaining why they’ve taken this route, and what it will mean for their 3D printed human tissue moving forward. Organovo’s decision to raise the additional funding was a move aimed at achieving three main goals:
- Expand on the total number of 3D printable tissue types they are able to offer commercially, and expand the market for the current liver tissue they are already offering.

- Bring additional institutional investors on board. By providing large blocks of new shares to institutions via this offering, the company hopes that the overall share price will stabilize, with less volatility taking place.
- Continue to build a strong relationship with investment banks, hopefully garnering the attention of further analyst coverage, thus attracting longer-term investors rather than short-term flippers.
“Organovo regularly emphasizes to our investors that our 3D bioprinting capability is a platform technology. We have been consistent in communicating to our shareholders that we will take advantage of financing opportunities when we believe they can offer long term shareholder benefit,” explained the company. “We have often repeated this after discussing that we had two years of capital in hand under our previous operating plan, that we would seek to be opportunistic if we could do more.”
So how will these additional funds be utilized according to the company?
While their exVive3D™ Human Liver Tissue has only begun to hit the market and their kidney tissue is still many months out, the company expects a significant increase in demand in the years to follow, meaning they will have to expand their capacity to produce these 3D printed tissues. In order to take complete advantage of the opportunities and keep up with demand, they foresee the need to open a second manufacturing facility sometime in the near term. This, of course, will require a significant capital allocation.
The company also foresees numerous other products coming online within the next few years, which will need further investment and R&D. They specifically point to their 3D skin models, which are currently being funded, through the point of model validation, via their partnership with L’Oréal. This funding is only for models related to cosmetic use, and if Organovo wishes to take advantage of their 3D printed skin tissue for other uses, a larger R&D budget is necessary.
“The opportunity to use it [3D skin model] more broadly requires a further investment, to commercialize the model for wider use, make it relevant for dermatology pharma use, etc,” explained the company.
Additionally, they wish to advance their 3D cancer model offering at a more rapid pace, meaning this additional capital could play a major role in its development and commercialization, sooner rather than later.
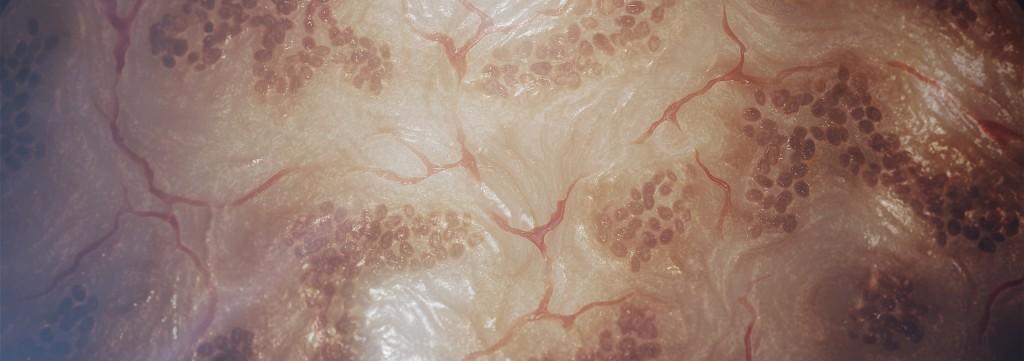 Finally, Organovo says that they see tremendous opportunity in not only 3D printing healthy liver and kidney tissue, but also diseased tissue. They are looking to ultimately print tissue which is unhealthy, perhaps kidney tissue which has polycystic disease, or a liver fibrosis model. In doing so they could eventually begin screening for drug candidates themselves, exponentially increasing their possible revenue opportunities.
Finally, Organovo says that they see tremendous opportunity in not only 3D printing healthy liver and kidney tissue, but also diseased tissue. They are looking to ultimately print tissue which is unhealthy, perhaps kidney tissue which has polycystic disease, or a liver fibrosis model. In doing so they could eventually begin screening for drug candidates themselves, exponentially increasing their possible revenue opportunities.
“For $46M raised in August 2013, we have been building what we believe can grow to be two $100M+ annual revenue first to market tissue franchises,” explained the company. “One way to think of the recent financing is to consider point number two [investment in new tissue buildouts] in terms of potential uses of funds: we can now choose to develop four tissues that have the potential to deliver $400M+ per year in top line revenue instead of two tissues at $200M+.”
Certainly it’s never fun for investors to experience dilution, but if you believe Organovo’s reasoning for the dilution, the additional funding seems to have the potential to greatly increase their bottom and top lines in the long run.
Are you an Organovo shareholder? What are your thoughts on this recent secondary offering and its potential impact down the line? Discuss in the Organovo funding forum thread on 3DPB.com.
Subscribe to Our Email Newsletter
Stay up-to-date on all the latest news from the 3D printing industry and receive information and offers from third party vendors.
Print Services
Upload your 3D Models and get them printed quickly and efficiently.
You May Also Like
Bambu Lab A1 Used to Directly 3D Print Copper Electroplated Parts
Maker Dzingof was doing tonnes of electroplating of desktop and other 3D prints years ago with his Metalizzr project. I’ve been playing, a lot less successfully, with electroplated 3D prints...
3D Printing News Briefs, October 1, 2025: Bambu Lab Store, Shape-Morphing Materials, & More
In today’s 3D Printing News Briefs, Bambu Lab has opened its first retail store, and Meltio is inaugurating its first international additive manufacturing reference site. A consortium has launched a...
3D Printing News Briefs, August 13, 2025: Public Utility, Rocket Engine, Brains, & More
In today’s 3D Printing News Briefs, AML3D’s large-scale metal 3D printing system is now online at the largest public utility in the U.S. Moving on, Bright Laser Technologies is 3D...
Soft Robotics is Finally Coming Together: Northwestern Researchers Make Strides Towards a Better Actuator
Soft robotics for a long time was a lot of nice videos, papers, and very little in the way of useful technology. It was kind of an engineering Esperanto: a...










































