With more than 440 patents under his belt, American engineer and inventor Dean Kamen has been searching for technological solutions to medical and social welfare dilemmas since he was a teenager. Mostly known as the creator of the Segway, a two-wheel self-balancing transportation system that is now ending production after two decades on the market without ever achieving wide consumer acceptance, Kamen is already onto something new: pursuing the mass-production of human organs and tissues.
Solving some of healthcare’s most challenging problems is not new to Kamen. After founding his first medical device company in 1976 to manufacture and market the first wearable infusion pump, he went on to work with leading diabetes researchers to pioneer another first, this time a portable insulin pump. This was followed by an ongoing list of inventions, including new technology to help end-stage kidney patients, a stair-climbing wheelchair, and even a prosthetics arm system that allows wearers to have “near-natural” control of their artificial limbs.
 Dean Kamen (Credit: FIRST Twitter account)
Dean Kamen (Credit: FIRST Twitter account)However, nothing comes close to his latest venture: the scaling up of tissue engineering, and eventually, human organ fabrication. To that end, Kamen brought together over 100 public and private organizations to develop an entirely new and collaborative ecosystem for regenerative medicine. Leading a nonprofit organization known as the Advanced Regenerative Manufacturing Institute (ARMI), Kamen aims to mend a fragmented tissue engineering field that “lacks a mechanistic framework with which to translate laboratory discovery into manufactured products.”
“There is so much work that needs to be done at the intersection of engineering and medicine to create a whole industry and give people a place for organs, whether it’s your liver or your kidney or your lung,” said Kamen two years ago during a talk he gave at Tufts University, in Massachusetts. “Every one of the problems we work on is a really big need that if not solved, and solved soon, the world will be sick or people will die. There has to be a core technology, and there has to be a need, and if we succeed, it will make a major difference.”
The organization is headquartered at New Hampshire’s Manchester Millyard, a set of refurbished mill buildings that also house several of Kamen’s other companies. ARMI is one of 14 sites under the federal umbrella of the network of regional institutes Manufacturing USA and the United States Department of Defense (DoD) that is focused on catalyzing specific and promising advanced manufacturing technology areas.
In December 2016, the DoD awarded ARMI an $80 million grant to operate the public-private partnership BioFabUSA, which launched mid-2017. The consortium comprises over 150 companies, academic institutions, and not-for-profit organizations that claim to be researching and building the equipment needed to manufacture lab-grown organs on a large scale.
From bioprinting startups like Cellink and Xylyx Bio to academic institutions like the University of Massachusetts Lowell, Northwestern University, the University of Vermont, the University of Notre Dame, and more, they are all collaborating to support the development of tissue and organ manufacturing capabilities. With members eligible for funding, over $33.3 million has been approved for projects to manufacture tissue-engineered medical products so far.
“I am astounded by the 21st century science fiction done by scientists in the field of regenerative medicine. I am equally astounded by the science fiction in their labs, where the manual labor conducted by technicians is reminiscent of Louis Pasteur’s laboratory. It is amazing that these miracles can be performed without modern process controls, robotics, and sensors, but this field will need 21st century engineering and manufacturing to mature into an industry capable of manufacturing FDA-approved tissues at the scale they are needed,” described Kamen, executive director of BioFabUSA. “BioFabUSA will build the coalition of industry, academia, and government that I hope will make that happen and enable the next big advance out there.”
While ARMI’s mission is to make practical the large-scale manufacturing of engineered tissues and tissue-related technologies, the technical scope for BioFabUSA work includes innovations across five thrust areas:
- Cell selection, culture, and scale-up
- Biomaterial selection and scale-up
- Tissue process automation and monitoring
- Tissue maturing technologies
- Tissue preservation and transport.
To carry this out, BioFabUSA claims to integrate innovative cell and tissue cultures with advances in 3D bioprinting, automation, robotics, and analytical technologies to create disruptive research and development tools and FDA-compliant volume manufacturing processes.
The institutes joining the ARMI-BiofabUSA group, have the opportunity to work together to develop industry standards and bring ground-breaking products to the clinic and market, with the ultimate goal of fabricating on-demand organs in the operating room (OR).
“The academic and commercial R&D community has done a tremendous job driving innovation in the field of regenerative medicine,” said Graham Chynoweth, startup mentor at ARMI/BioFabUSA, when the University of Vermont (UVM) joined the high-profile regenerative medicine consortium on January, 2018. “Now it is time to move from bench and clinical scale to commercial-scale manufacturing. New and different types of talent and expertise are needed for this transition to succeed. We are thrilled that UVM will be joining forces with us to support this transition and develop this talent pipeline.”
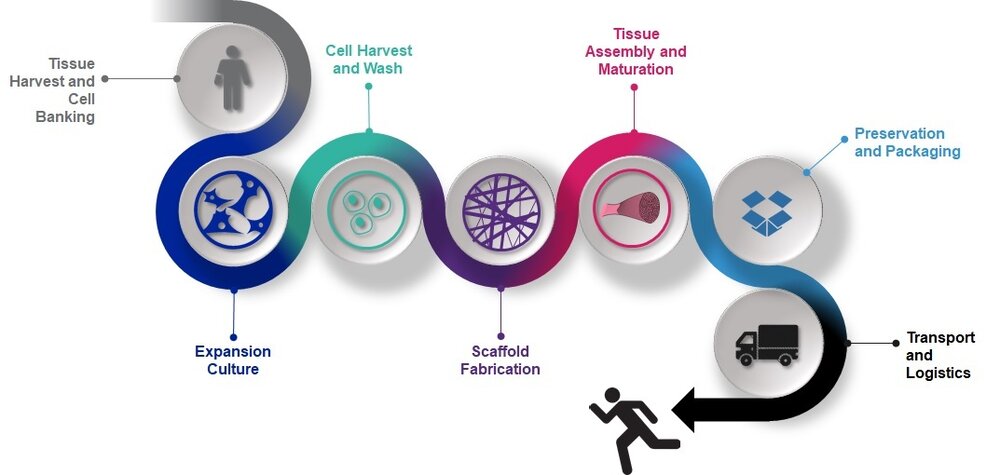 Tissue Foundry process (Credit: ARMI/BioFabUSA)
Tissue Foundry process (Credit: ARMI/BioFabUSA)Claiming to focus on creating state-of-the-art manufacturing innovations in biomaterial and cell processing for critical DoD and civilian needs, one of the projects under development at ARMI’s BioFabUSA is the Tissue Foundry prototype manufacturing platform. According to ARMI, the long-term goal of this project is to build a modular, flexible, GMP-compliant, closed, and fully automated manufacturing line for engineered tissues.
Yet, all that we know so far is that, in the short term, the team has focused on building a prototype modular manufacturing system for engineered tissues, which will serve to highlight capability gaps to refine BioFabUSA’s focus areas. The line is designed to have five modules, which will focus on seed pool culture, cell harvest and wash, scaffold fabrication, scaffold seeding and culture, and final packaging of the product. Ultimately, the manufacturing line will have a common core, but be modular enough to allow for the manufacture of a wide variety of tissue-engineered medical product constructs, and will be used for process validation, early phase clinical manufacturing and will form the basis for technology transfer activities. We expect more information to become available once the end of the DoD five-year contract concludes in 2021.
 At the DoD Manufacturing Institute, attendees can get a virtual reality tour of ARMI’s Tissue Foundry Line, the output of the line is this bone-ligament-bone construct (Credit: ARMI/Rockwell Automation)
At the DoD Manufacturing Institute, attendees can get a virtual reality tour of ARMI’s Tissue Foundry Line, the output of the line is this bone-ligament-bone construct (Credit: ARMI/Rockwell Automation)This is one of the many development activities necessary to achieve the organization’s goal of a scalable, modular automated, and closed tissue manufacturing line. Last year, Kamen suggested the new Tissue Foundry project could lead to the manufacture of tissue to be tested in animals before human clinical trials and hoped animal trials might start sometime this year.
While working on this project for four years, many companies have become members of ARMI and contributed technology and funding. This includes industrial control system developer Rockwell Automation, which committed $10 million to ARMI to develop ways to scale up the production of new technologies to produce tissues, such as growing artificial skin for grafts. Regenerative medicine company, CollPlant, supplied its expertise in bioinks, based on its rhCollagen, as a building block for 3D bioprinting tissues and organs. While Louisville-based Advanced Solutions—another ARMI member with a subsidiary at the Millyard campus—provided its six-axis robot, the BioAssemblyBot 3D bioprinter, to build biological structures, such as growing blood vessels outside of the human body. Another disclosed project comes from a team of biomedical engineers at Boston University, who designed a low cost, single-use bioreactor prototype that can grow tissue that is compatible with the Tissue Foundry fabrication line.
 Low cost, single-use bioreactor for ARMI’s Tissue Foundry project (Credit: Boston University/DEKA R&D)
Low cost, single-use bioreactor for ARMI’s Tissue Foundry project (Credit: Boston University/DEKA R&D)In addition to research, ARMI’s BioFabUSA is also focused on workforce development designed to create a new generation of employees to fill skilled, high paying jobs in regenerative medicine that barely exist today. In partnership with the University of Minnesota and its 3D Bioprinting Facility, BioFabUSA is developing educational programs to support the 3D bioprinting skills required to train future workers. These programs involve exposing young students to 3D printing, especially focused bioprinting courses for two- and four-year degree programs and graduate school, Veterans education, and even intense one-week workshop courses for industry professionals. In fact, one of the advantages to undergraduate and graduate students at the universities that are part of the BioFabUSA ecosystem are the internship and employment opportunities available to them.
Even though a lot of the information about the ongoing projects at ARMI-BioFabUSA has not been completely disclosed yet, Kamen and the consortium members are convinced that by working together, they will play an important role in the future of medical technology to advance the tissue manufacturing industry for curing certain diseases, like diabetes, renal failure, and limb loss. We’ll be on the lookout for more information coming out of ARMI-BioFabUSA in the future.
Subscribe to Our Email Newsletter
Stay up-to-date on all the latest news from the 3D printing industry and receive information and offers from third party vendors.
You May Also Like
3D Printing News Briefs, April 13, 2024: Robotics, Orthotics, & Hypersonics
In 3D Printing News Briefs today, we’re focusing first on robotics, as Carnegie Mellon University’s new Robotics Innovation Center will house several community outreach programs, and Ugogo3D is now working...
Rail Giant Alstom Saves $15M with 3D Printing Automation Software 3D Spark
3D Spark has entered into a three-year deal with the rail giant Alstom. Alstom, a transport behemoth with annual revenues of $16 billion, specializes in the manufacture of trains, trams,...
Meltio Expands Global Reach with New Partnerships in the Americas and Europe
Spanish 3D printing manufacturer Meltio has expanded its sales network across the globe. With the addition of three new partners in the United States, Brazil, Argentina, and Italy, Meltio aims...
3D Printing Webinar and Event Roundup: April 7, 2024
Webinars and events in the 3D printing industry are picking back up this week! Sea-Air-Space is coming to Maryland, and SAE International is sponsoring a 3D Systems webinar about 3D...































