Cristina Gentili recently presented a thesis, ‘3D Printed Instrumented Packaging for Implantable Devices,’ to the Centre of Bio-Inspired Technology at the Imperial College London. While there is much research focused on the creation of 3D printed medical models, surgical planning devices, and implants, this study centers on the importance of effective, biocompatible packaging for medical electronic implants. Such devices offer critical monitoring for patients who may be suffering from one or more serious conditions like heart disease, diabetes, and other chronic health issues.
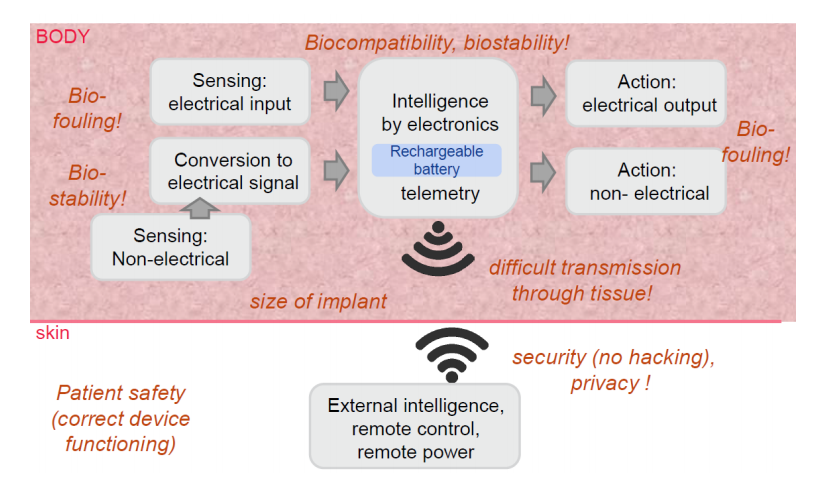
Pointing out that the human body ‘is a critical site,’ Gentili states that device packaging is of the utmost importance due to risk of rejection of damage to tissue. Packaging must offer the following:
- Biocompatibility
- Biostability
- Strength
- Smooth, soft texture
Materials such as titanium, ceramic, or glass are often used for device packaging as they are able to offer proper resistance against moisture and can also hold up against stress and wear.
Production processes today may be extremely expensive however, as well as complicated and challenging, leaving Gentili to suggest more affordable—and fast—production of polymeric packages via digital light processing (DLP). There are still obstacles to overcome in such manufacturing, though, such as adhesion issues, high porosity, and inferior surface quality.
“Active implantable medical devices are complex systems under extremely tight design constraints. Requirements for implants involve general common design rules and principles, but application specific requirements, as well,” states Gentili. “For instance, the device specific function, sensing mechanism, placement in the body, implantation time… and so on, have all to be taken into account in the whole design phase.”
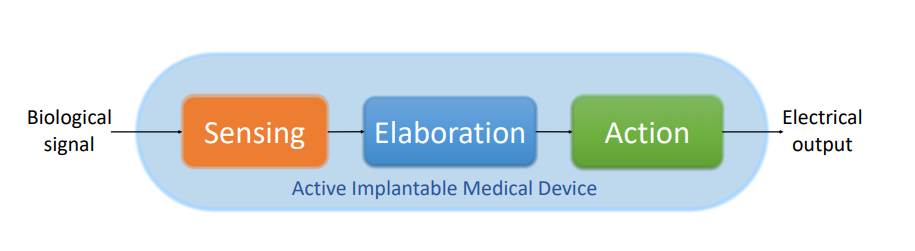
Active implantable medical devices are divided into two categories: diagnostic and therapeutic. Typically used for monitoring, diagnostic AIMDs detect sugar levels, blood pressure, inner ocular pressure, and other physiological signals. Therapeutic AIMDs often act as pacemakers, neurostimulators, recorders, modulators, and more. Main features required are:
- Ability to receive/sense electric signals
- Ability to analyze input
- Actuation, depending on signals arriving from human tissue, resulting in action and electrical output
One of the most difficult challenges in creating packaging for such devices is the hermeticity requirement. More traditional methods for measurement would include exposure in a pressurized helium chamber; however, that technique is not suitable for polymers as they are permeable to helium.
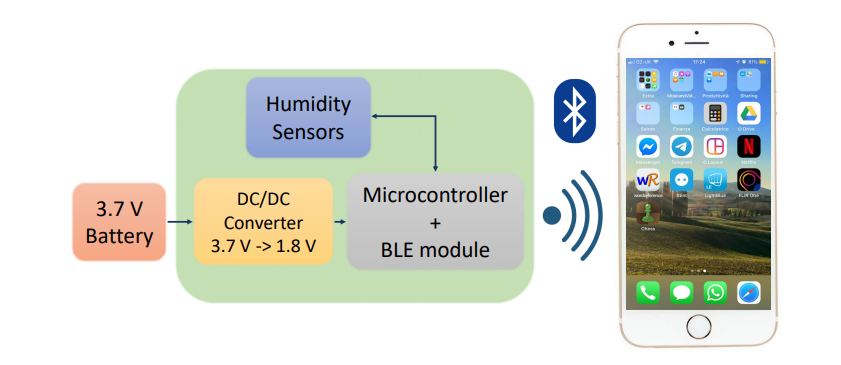
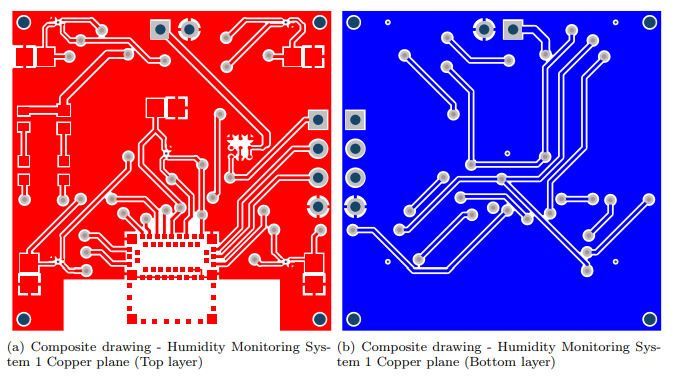
An in situ monitoring system was used to measure humidity and temperature, with data shared through Bluetooth. These techniques offer ‘the best sensitivity’ regarding leak detection and permeation of the implant package. The downside in using these systems, however, is that an electronic structure must accompany such sensors. For this study, the system was designed in a 29×29 mm printed circuit board.

Sensor measurements sent by Bluetooth to the iPhone App. (a) Characteristic values read and notification. (b) Log view.
“… the advantages are that any package material can be used and there are no sensibility limitations due to cavity dimensions,” states Gentili. “In fact, in situ methods are independent on the volume. Furthermore, it is possible to detect the outgassing of internal materials, as well.”
“For what concerns the estimated lifetime inside the human body organism, to allow a temperature variation of maximum 4◦C, it turns that the maximum relative humidity allowed in the cavity, to not make condensation happen, is about 80%,” concluded Gentili. “This upperlimit was reached after 4065 minutes of test at 77◦C, which are equivalent to about 37 days (considering sensors tolerances) at human body temperature.
“As a last remark, it has been highlighted that corrosion problems due to ionic contamination in printed circuit board assemblies may further lower the maximum relative humidity level. Therefore, considering this last safer limit, the implant lifetime is estimated to be about 20 days. Hence, the final conclusions assert that the developed package structure is not suitable for a long term implantation. To make it acceptable for at least three months inside the human body organism, it actually needs improvements concerning both the substrate permeability and the sealing process.”
What do you think of this news? Let us know your thoughts! Join the discussion of this and other 3D printing topics at 3DPrintBoard.com.
[Source / Images: ‘3D Printed Instrumented Packaging for Implantable Devices’]
Subscribe to Our Email Newsletter
Stay up-to-date on all the latest news from the 3D printing industry and receive information and offers from third party vendors.
You May Also Like
Gorilla Sports GE’s First 3D Printed Titanium Cast
How do you help a gorilla with a broken arm? Sounds like the start of a bad joke a zookeeper might tell, but it’s an actual dilemma recently faced by...
Nylon 3D Printed Parts Made More Functional with Coatings & Colors
Parts 3D printed from polyamide (PA, Nylon) 12 using powder bed fusion (PBF) are a mainstay in the additive manufacturing (AM) industry. While post-finishing processes have improved the porosity of...
$25M to Back Sintavia’s Largest Expansion of Metal 3D Printing Capacity Since 2019
Sintavia, the digital manufacturing company specializing in mission-critical parts for strategic sectors, announced a $25 million investment to increase its production capacity, the largest expansion to its operations since 2019....
Velo3D Initiates Public Offering in a Bid to Strengthen Financial Foundations and Drive Future Growth
Velo3D (NYSE: VLD) has been among a number of publicly traded 3D printing firms that have attempted to weather the current macroeconomic climate. After posting a challenging financial report for 2023,...