Due to the ravaging effects of COVID-19 on the respiratory system of patients, ventilators are in high demand. However, due to the sheer capacity of hospitals, a lack of global coordination around manufacturing and distributing this complex medical equipment, and disruptions in the globalized supply chain, ventilators are also in short supply.
We’ve previously discussed a number of efforts to produce cheap ventilators from scratch, but what we have really been waiting for is for ventilator manufacturers to step up to the task. While GE Aerospace workers demand their employer shift all jet engine manufacturing to ventilator production, one of the largest makers of ventilators, Medtronic, has released the design specifications for its $8,000 PB560 ventilator system.
Over 200 pages of documentation, mechanical part drawings, all CAD files, manufacturing fixtures, printed circuit board drawings for multiple bills of materials for the PB560 have been published by Medtronic. This will allow engineers to develop systems for rapid production while Medtronic increases its own production of the machines. Having increased manufacturing capacity in Ireland by 40 percent, the company is also collaborating with Tesla to use its New York site to make more PB560 systems.

Medtronic employees in Ireland have increased ventilator production by 40 percent. Image courtesy of Medtronic.
As grateful as we might be for the work that businesses like Tesla is doing to aid in the manufacture of such critical equipment, it’s important not to lose sight of the fact that they are still performing potentially problematic business practices in other regards. Tesla, for instance, kept workers assembling its latest vehicles despite shelter-in-place orders from the California state government and two cases of COVID-19 among its office workers, all while laying off employees.
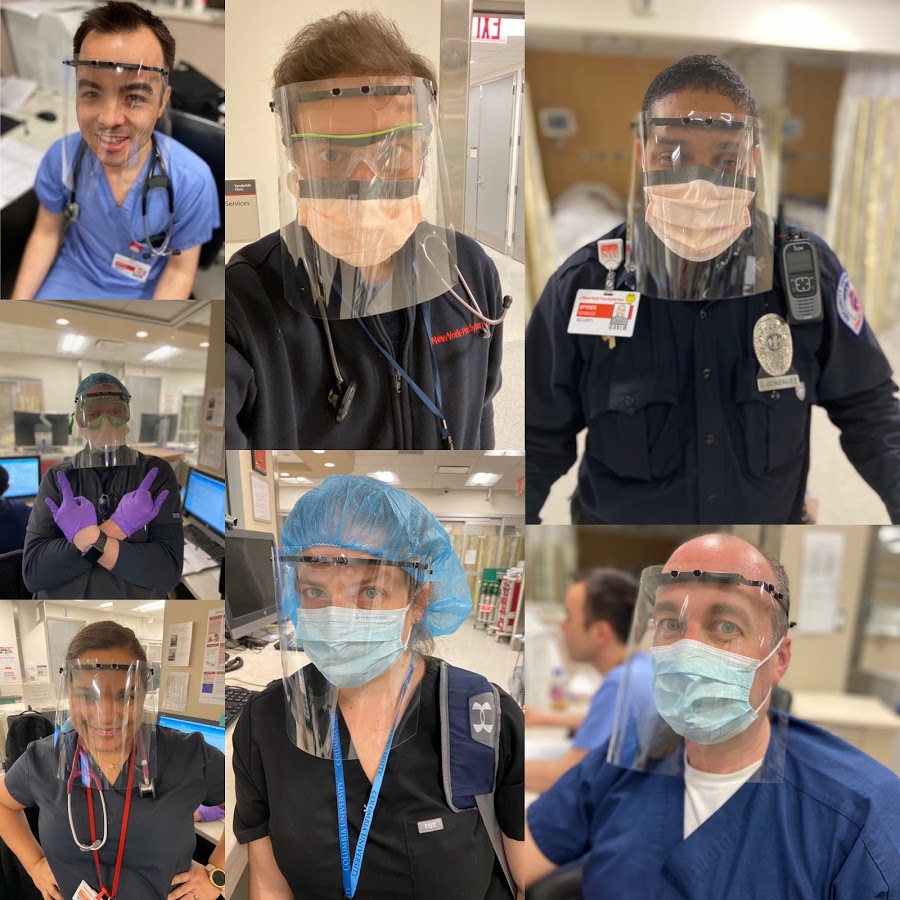
Healthcare professionals at Columbia University Medical Center/New York-Presbyterian Hospital using 3D printed face shields thanks to the MatterHackers Maker Response Hub. Image courtesy of MatterHackers.
Meanwhile, MatterHackers has seen overwhelming support from its Response Hub, in which over 3,500 volunteers have enlisted over 10,000 3D printers to produce personal protection equipment (specifically face shields) for multiple hospitals in the U.S. The company has a team of Engineers, EHS, and Emergency Response specialists that are validating designs to ensure the safety and effectiveness of the devices being made. The volunteers have produced over 3,500 face shields for Columbia University Medical Center, New York-Presbyterian Hospital in New York, Geisinger Health Systems in Pennsylvania, and Palo Alto Medical Center in California.
Similarly, the e-NABLE network (known for its work in 3D printed prosthetics), is addressing medical supplies needs by making face shields to help preserve N95 respirator masks. As listed on the e-NABLE website, volunteers from across the globe are producing shields for healthcare staff in New York, Ohio and Oregon in the U.S., as well as in Australia, Brazil, Columbia, Greece, Ireland, New Caledonia and Thailand.
We are in the process of speaking with sources about the safety of various 3D-printed medical devices. From what we gather, face shields featuring parts 3D printed using desktop extrusion printers are comparatively safe, as long as they are routinely disinfected and medical professionals are requesting them in huge numbers. The safety of other 3D printed devices are still questionable.
For instance, face masks (not shields) require greater precautions. WASP has designed customizable face masks and released a Blender add-on that makes it possible to design masks that can fit to the wearer’s face. Including an interchangeable filter, the mask is meant to fit comfortably and be clean and sterilized easily. So far, WASP is testing the device as it receives requests from police, city officials and supermarket employees to make the mask.
Because they would be worn by non-medical professionals, the safety requirements are not as stringent as if they were worn by medical staff. According to the FDA, homemade masks can prevent wearers from projecting particles and are, therefore, acceptable for the public to use so that surgical masks and N95 masks can be saved for healthcare providers.
Therefore, those who are developing these types of devices for 3D printing must work with medical professionals and regulatory bodies in order to ensure that they are adequate replacements when traditionally regulated devices are not available. In the U.S., this can be achieved by working through America Makes.
Otherwise, hobbyists might consider reading working with #GetUsPPE, which is collaborating with Nation of Makers and Open Source Medical Supplies to increase PPE stocks. The partners are first requesting people to donate their own PPE to local hospitals by following a helpful script. Beyond that, they are asking Makers with 3D printers, CNC machines, injection molding machines, sewing and textile equipment, and more to visit the initiative website to learn how to produce medically validated designs and follow proper instructions to make PPE for hospitals within their network.
Subscribe to Our Email Newsletter
Stay up-to-date on all the latest news from the 3D printing industry and receive information and offers from third party vendors.
Print Services
Upload your 3D Models and get them printed quickly and efficiently.
You May Also Like
The Convergence of Vision and Experience: AMS and AMUG
During the last few weeks, I spent time on the ground at both the Additive Manufacturing Strategies Forum (AMS) and the Additive Manufacturing Users Group (AMUG) meeting. What stands out...
HP’s Open Industrial Filament System Made North American Debut at AMUG 2026
At Formnext 2025, HP Additive Manufacturing revealed a major step in its broader portfolio expansion strategy with the announcement of a new 3D printer, the HP Industrial Filament 600 High...
The Magic of AMUG as Reported by a First-Time Attendee
There’s a special kind of magic about AMUG. I’ve heard about it for years, but never experienced it myself until last week. It’s different than what you see at some...
Würth Additive Group & B9Creations Announce Strategic Partnership at AMUG 2026
Last week, hundreds of additive manufacturing users (and a few journalists, like me!) met in Reno, Nevada to attend the 2026 Additive Manufacturing Users Group (AMUG) Conference. I’ll share more...































