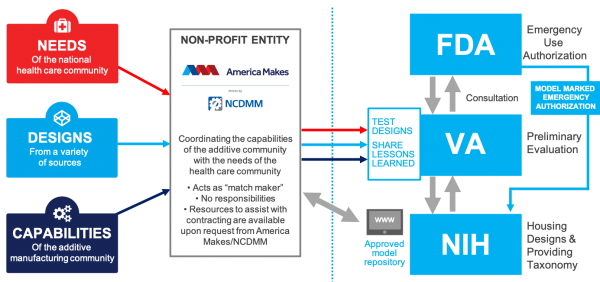
The U.S. Food and Drug Administration (FDA) is now working with the Department of Veterans’ Affairs, the National Institutes of Health (NIH), and the 3D printing trade group America Makes to provide some coherence and regulation to the work of additive companies and individuals to address the COVID-19 outbreak in the U.S.
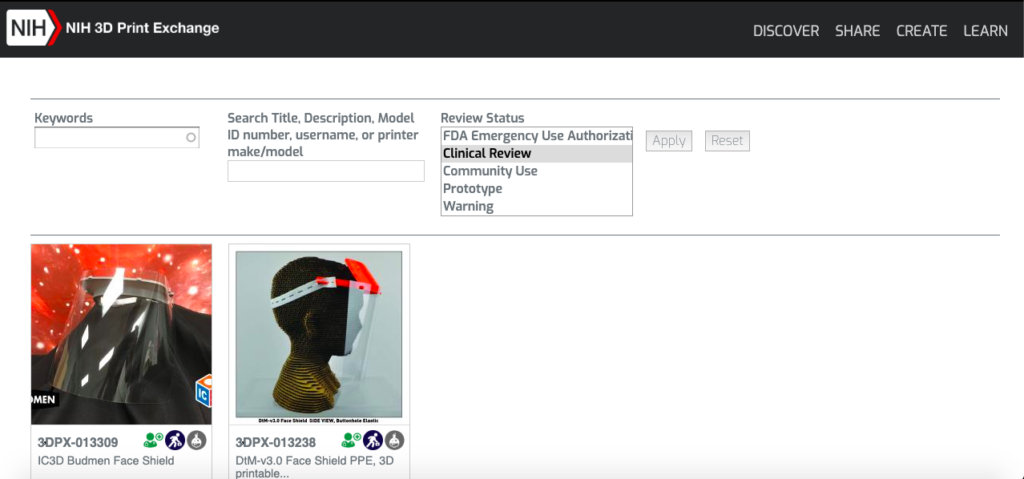
The result is the COVID-19 Health Care Needs and Additive Manufacturing (AM) Capabilities Repository, an online library where AM companies can upload designs to be reviewed and placed on the NIH 3D Print Exchange. When uploaded through the America Makes submission form, 3D printable designs will be fast tracked for review. According to the organization, this approval process is meant to help determine the safety and reliability of these devices in a healthcare setting.
The site will include information necessary for AM and healthcare providers that will ensure they meet medical standards for production. Through this process, the FDA is processing Emergency Use Authorization Applications to streamline the availability of safe medical devices. So far on the NIH repository, two face shields are under clinical review, while numerous others are prototypes in progress. This raises the question of just how much innovation needs to be dedicated to face shields when there are already so many designs available. The next step for these shields is FDA Emergency Use Authorization.
Meanwhile, the VHA has established an ecosystem meant to connect hospitals and clinics within the VA healthcare system that have 3D printing design and engineering expertise so that they can provide that expertise to others. As a part of the VHA Innovation Ecosystem, lessons and resources can be shared with frontline employees and the 3D printing community. Individuals can also volunteer to print, design, or provide feedback for in-demand equipment & resources.
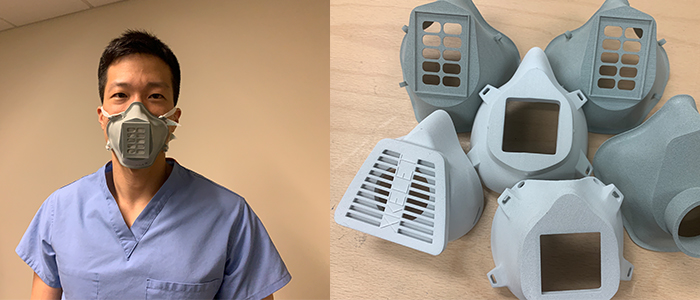
Left Image: VA physician Dr. William Chang evaluates a prototype 3D printed mask, designed by VA staff. Right Image: Different mask designs printed and awaiting clinical evaluation. Image courtesy of VHA.
Within this larger effort, the FDA and its partners are establishing other resources to aid in the development, production and distribution of 3D-printed medical devices as well. This includes a key FAQ page on 3D printing medical devices, accessories and parts during the pandemic. Here is a summarized list of what is discussed on the page:
- The FDA has issued previous guidance on 3D printed medical devices that should be considered during the pandemic.
- 3D-printed personal protection equipment (PPE) will not provide the same protection as FDA-cleared surgical masks and N95 respirators, but if they are used, the FDA has provided information about how to make them as effective as possible.
- Safety considerations that should be taken into account for 3D printed masks include:
- checking the mask for seals
- ensuring that the wearer can breathe through the mask and any makeshift filters
- using caution in surgical environments
- knowing the mask may not be an adequate filter for preventing the transmission of disease
- safely disposing of the masks.
- Complex products cannot be easily printed
- Those printing parts should ensure they match originals as much as possible and ensure they fit and work properly.
- Those printing parts should work with relevant medical device manufacturers.
- The FDA is willing to discuss the possibility of 3D printing entire medical devices and should be contacted at COVIDManufacturing@fda.hhs.gov to learn more.
- The FDA’s Emergency Use Authorization (EUA) includes 3D printed tubing for multiplexing ventilator use.
Other critical information shared by the FDA includes studies examining and optimizing the decontamination and reuse of respirators during public health emergencies. Additionally, ASTM International is offering free access to its Standards related to COVID-19. The FDA has also provided a link to Cambridge University’s research into the efficacy of household materials as filters used during a flu pandemic.
We will examine all of this information in greater detail to better understand what is and isn’t needed from 3D printing providers to address medical supply shortages, as well as the safety precautions involved, and provide further updates in subsequent articles.
Subscribe to Our Email Newsletter
Stay up-to-date on all the latest news from the 3D printing industry and receive information and offers from third party vendors.
You May Also Like
3D Printing News Briefs, April 27, 2024: Research, Digital Dentistry, Cycling, & More
We’re starting today’s 3D Printing News Briefs with some research into 3D printed luminescent quantum-dot polymer architectures and free-form laser beam shaping, and then on to an open source 4-axis...
HP & INDO-MIM Collaborate to Boost Metal 3D Printing in India
HP Inc. and INDO-MIM, a US- and India-based supplier of metal injection molding (MIM) powders and contract manufacturer, have announced that the two companies will collaborate to accelerate additive manufacturing...
3D Printing News Briefs, February 17, 2024: Shot Blasting, Service Bureaus, & More
In today’s 3D Printing News Briefs, we’re starting out with post-processing, as SKZ Würzburg is using a shot blast system from AM Solutions for its research. Moving on to business,...
3D Printing News Unpeeled: Not That Kind of Organ 3D Printing
GKN Aerospace will create a 150 jobs in Trollhattan Sweden with an investment of $60 million part of which comes from the Swedish Energy Agency’s Industriklivet initiative. The investment will...