Korea: 3D Printing Pregabalin Tablets for Successful & Controlled Release
In the recently published ‘Customized Novel Design of 3D Printed Pregabalin Tablets for Instra-Gastric Floating and Controlled Release Using Fused Deposition Modeling,’ Korean researchers examine more detailed use of 3D printing as a vehicle for patient-specific treatment in medications.
While there is attention being paid to the potential for 3D printing of pills, changes within the medical and pharmaceutical industry are still in the beginning stages, following on the heels of FDA approval for Aprecia’s 3D printed epilepsy pill, Spritam. Medication dispensers, and innovations like different shapes and designs of pills for kids have been introduced, but still, there is much to be explored within the realm of fabricating filaments and tablets. In this study, researchers created a new, floating sustained-release system via FDM 3D printing.
Active ingredients for the filaments include:
- Hypromellose acetate succinate (HPMCAS)
- Polyethylene glycol (PEG 400)
- Pregabalin
Tablets were printed with infill percentages of 25%, 50% and 75%, featuring a bottom layer that is enclosed—but a top layer that is partially open. Filaments and tablets were characterized by:
- Fourier-transform infrared spectroscopy (FTIR)
- Differential scanning calorimetry (DSC)
- X-ray powder diffraction (XRPD)
- Thermogravimetric analysis (TGA)
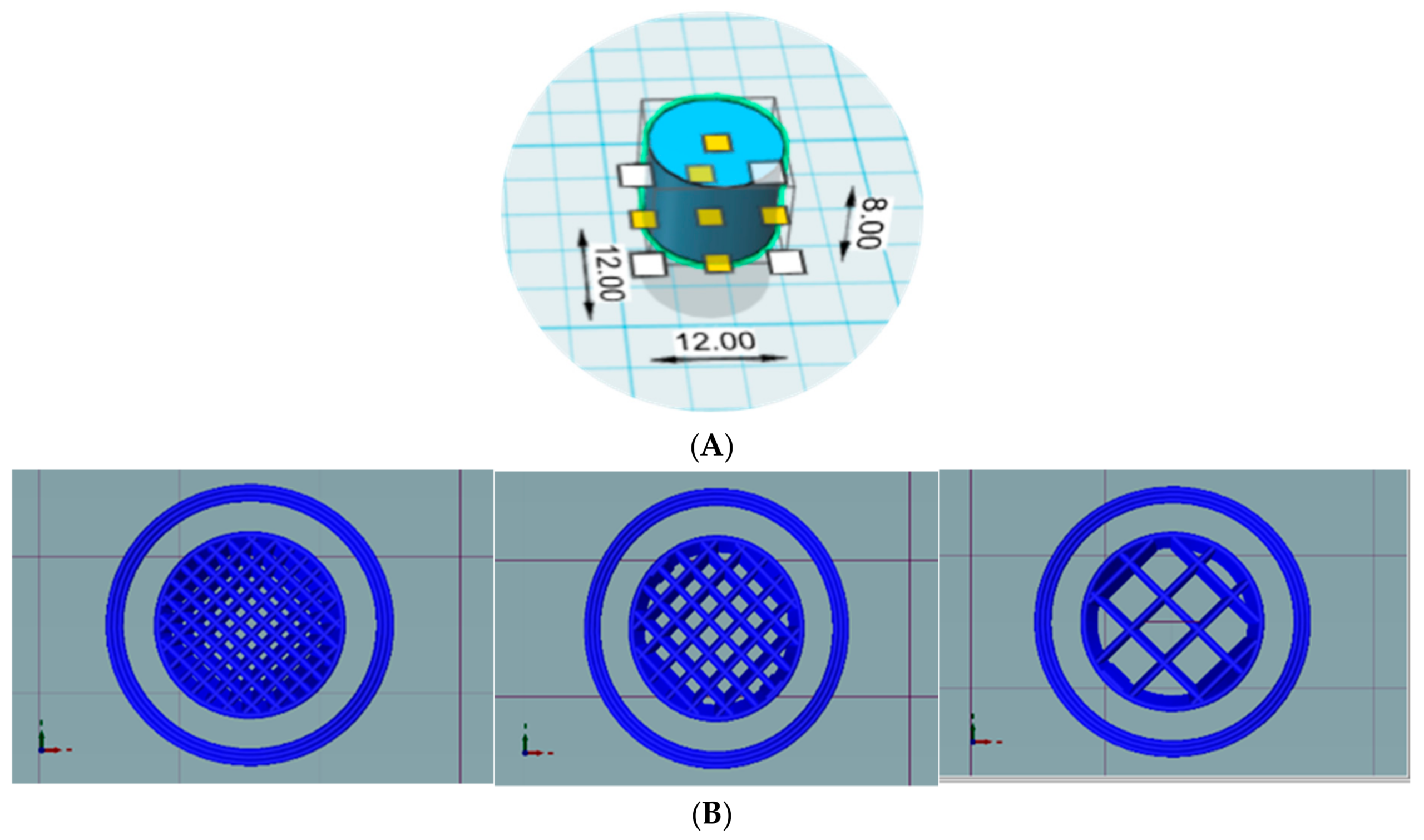
Design and internal structures of tablets. Design of preliminary cylindrical tablets (A) and slicing of tablets with infill percentages of 25%, 50% and 75%, left to right (B).
One of the greatest challenges in the use of pregabalin (approved by the FDA for managing not only epilepsy and diabetes but also other conditions like fibromyalgia and post-herpetic neuralgia) is the half-life of around six hours—meaning that it must be given two to three times a day; however, the researchers have noted that one of the best ways to override the need for more frequent dosing is to increase gastric retention of the medication.
“Floating gastro-retentive formulations can effectively minimize the risk of premature gastric emptying of swellable systems by floating above gastric juice and being away from the pylorus,” state the authors of the study. “Previously, an intragastric floating tablets of domperidone was formulated using 3D technology. As FDM 3D technology requires filaments as the main material, hydroxy-propyl cellulose filaments loaded with domperidone were prepared using hot melt extruder and hollow tablets were made using a 3D printer. Printed tablets were studied for in vitro and in vivo floating time and drug release profile, which demonstrated a promising application of FDM technology to reduce the frequency of administration and improve patient compliance.”

Prepared filaments and tablets. Drug loaded filaments (A), printed open system tablets with 25%, 50% and 75% infill left to right (B), and printed closed system tablet (C).
While tapping into previous research and combining information, the researchers created their own system featuring pregabalin in a floating tablet. Drug delivery is under controlled release. During the study, the researchers discovered that diameter uniformity affected weight variations. A partial crystalline structure was observed, also, during extrusion, with an endothermic peak of pure pregabalin at 194.81 °C. The team also noted a two to three percent mass loss in the hot melt extrudable (HME) zone, with only five percent in the printing zone. They attribute this to the possibility of high temperatures and degradation of material.
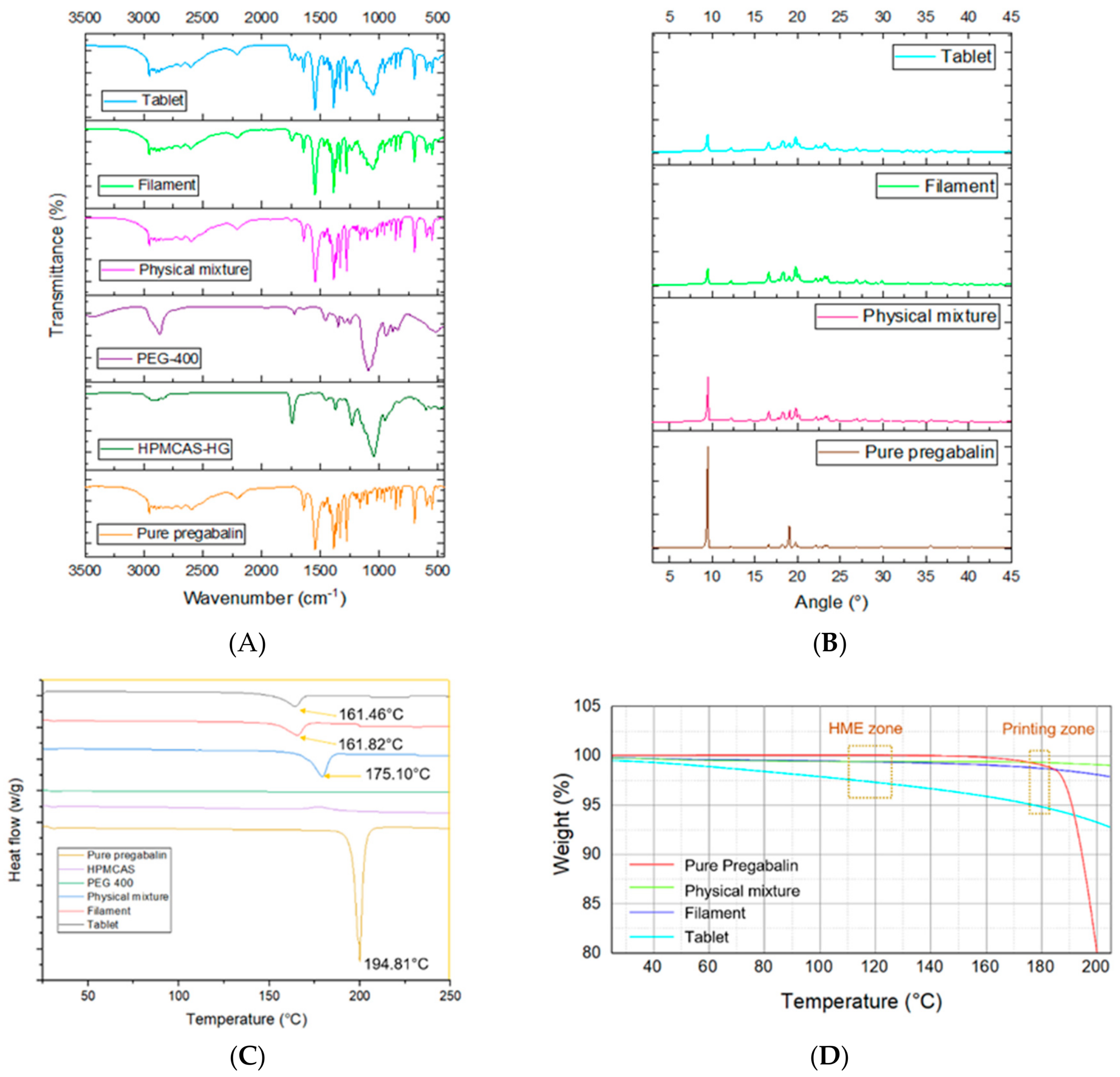
Physical characterization of printed tablets. Fourier-transform infrared spectroscopy (A), X-ray powder diffractometry (B), differential scanning calorimetry (C), and thermogravimetric analysis (D).
While previous research has found floating capacity and resulting duration to revolve around density of tablets, here, the researchers found a direct connection between the existence of both a top and bottom layer—and minimum dependency of density.

Floating study of prepared formulations. Open system (A), closed system (B), and optimized formulation (C) over 1, 8 and 24 h (left to right).
Floating mechanism of optimized formulation. The internal structure of tablet is composed of a grid infill with void space filled with air so that the tablet has low density, which helps in the buoyancy of the tablet in media.
“To understand the release properties better, drug release data were fitted to different mathematical kinetic models,” explained the researchers in conclusion. “A single model could not define the drug release pattern from tablets and rather seemed to have a combination of different mechanisms. The regression value was found to be higher for the Zero-order and Higuchi models, suggesting that the release and diffusion rates were constant. Drug release for HPMCAS polymers have been found to be regulated by the drug diffusion and erosion polymer from the surface of the system. Printed tablets did not show any changes in morphology and maintained their integrity during whole dissolution process.
“The results show that FDM printing is suitable for the formulation of floating dosage form with the desired drug release profile.”
What do you think of this news? Let us know your thoughts; join the discussion of this and other 3D printing topics at 3DPrintBoard.com.

Top view and side view of optimized formulation. Design of unique shaped optimized formulation (A), slicing of tablets with 25% infill (B) and printed tablets (C).
Subscribe to Our Email Newsletter
Stay up-to-date on all the latest news from the 3D printing industry and receive information and offers from third party vendors.
Print Services
You May Also Like
3D Printing Grows to $15.9B in 2024 Amid Shifting Industry Dynamics
The global additive manufacturing (AM) market reached $15.9 billion in calendar year 2024, according to “Q4 2024 3DP/AM Market Data and Forecast” from Additive Manufacturing Research (AM Research). Despite a...
Low-cost “Suzy” Polymer Powder 3D Printer is Faster and Cheaper than Past Models
Polish laser powder bed fusion (LPBF) firm Sinterit has released a follow-up to its predecessors, Lisa and Nils, called Suzy, a $19,490 printer equipped with a 30W fiber diode laser....
BellaSeno’s 3D Printed Breast Implants Keep Shape with 87% Fat Volume, Avoids Silicone Risks
At a medical conference in Austin this week, a new kind of breast implant took center stage. It is not made from silicone but from a 3D printed, fully resorbable...
Australia’s Untapped Potential as a Disruptive Innovator: SPEE3D CTO Steven Camilleri Explains Why He Wants to Make Stuff There
When I first met Steven Camilleri in 2023, the CTO of additive manufacturing (AM) OEM SPEE3D shared a quote with me from Clayton Christensen, the late Harvard professor who popularized...


























