Understanding cancer cells is a fundamental part of looking for ways to cure the disease, which has been around for a long time. Cancer is the second leading cause of death globally and was responsible for an estimated 9.6 million deaths in 2018. That means globally, about one in six people die from cancer. But even more troubling is knowing that between 30% and 50% of these deaths could have been prevented. According to the World Health Organization (WHO) public health measures, early detection, treatment, and new technologies could help stop patient suffering.
 Since 2009, French startup CTIBiotech has been moving fast to find ways to tackle the disease. With a strong, decades-old basis on cancer research, founders Nico Forraz and Colin McGuckin have perfected a technique using bioprinting to develop microtumors, which are human biological models of real tumors. The company, headquartered in the city of Lyon, released its microtumor initiative this year, mainly focusing on cells for breast, pancreatic, ovarian, prostate, colon and liver cancer. One of their main business lines is called CTIPharma, which revolves around oncology research developments with a mission to make predictive cell models and work on a National Biobank. 3DPrint.com recently interviewed Forraz, CEO of CTIBiotech, to learn how microtumors will improve drug development platforms and predictive medicine.
Since 2009, French startup CTIBiotech has been moving fast to find ways to tackle the disease. With a strong, decades-old basis on cancer research, founders Nico Forraz and Colin McGuckin have perfected a technique using bioprinting to develop microtumors, which are human biological models of real tumors. The company, headquartered in the city of Lyon, released its microtumor initiative this year, mainly focusing on cells for breast, pancreatic, ovarian, prostate, colon and liver cancer. One of their main business lines is called CTIPharma, which revolves around oncology research developments with a mission to make predictive cell models and work on a National Biobank. 3DPrint.com recently interviewed Forraz, CEO of CTIBiotech, to learn how microtumors will improve drug development platforms and predictive medicine.
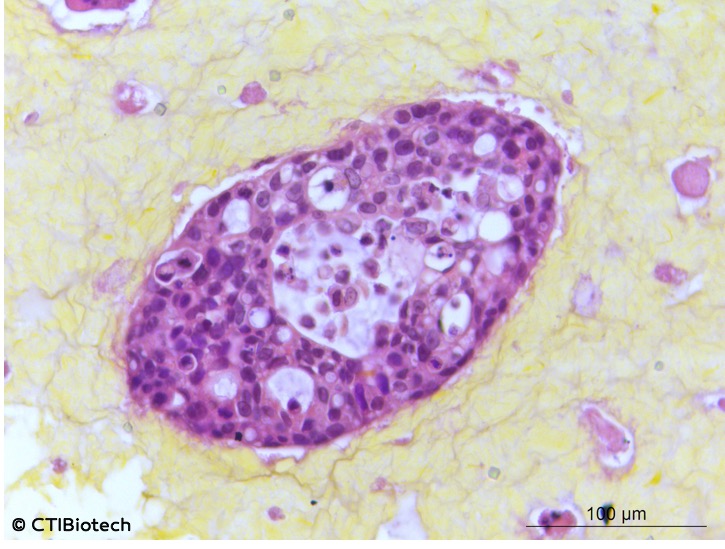
“For the last ten years, we have investigated ways to dissociate tumor samples, characterizing and sorting tumor initiating cells and cells within the tumor environment, such as, cancer stem cells, cancer-associated fibroblasts and endothelial cells. They are then expanded to amplify the number of cells available for study. Today, we have the technological know-how for the creation of models from primary tissues from the patient or derived tumor models.”
“Once we dissociate the cells of the tumor, we amplify them and package each cell type into a bioink cartridge. For example, we can combine the cancer cells with a special biomatrix into a cartridge and then the cancer associated fibroblasts into another one and the edothelial cells into a third one. Once we do that, we create an electronic design and print the cells accordingly to form a microtumor.”
“Until four years ago, we manually tissue engineered tumours. It would take an experienced scientist half a day to make 24 tumors. To accelerate the process, we began using bioprinting technology to make our bioassays, which means it now takes between 5 to 10 minutes for the same amount of tumors. Bioprinting is not just an interesting tool for productivity, it is also reprodcuible and brings added value to presicion, for instance, our specialists know that they can make several ‘copies’ of a tumour and be sure that the result will be identical to the original.”
At CTIBiotech, researchers use any of six CELLINK BIO X bioprinters, as well as a couple of proprietory printer designs that they developed using open source technology.
95% of new drug candidates fail cancer clinical trials, costing billions of dollars to the pharmaceutical industry and delaying new treatments for patients. Robust predictive human cell bioassays support faster development of preclinical trials thereby saving time and money. So using the latest tools in bioengineering, CTI’s researchers are now printing cancer tissues in 3D. These models can be used for highly significant preclinical trials and can be produced in sets of 24, 48 and 96 well plates for high throughput screening.
“At the lab, we test our designed microtumors to see if they work for drug development, subjecting them to intense research. We are noticing it is a technology that can fast track the preclinical development because it is tested on human cells in 3D, meaning it would make for a great universal drug screening and personalized medicine tool. Actually, I expect that in the future, an oncologist could collect tumour samples from a patient, bring it to our lab and in four weeks be able to know which drug combination works best.”
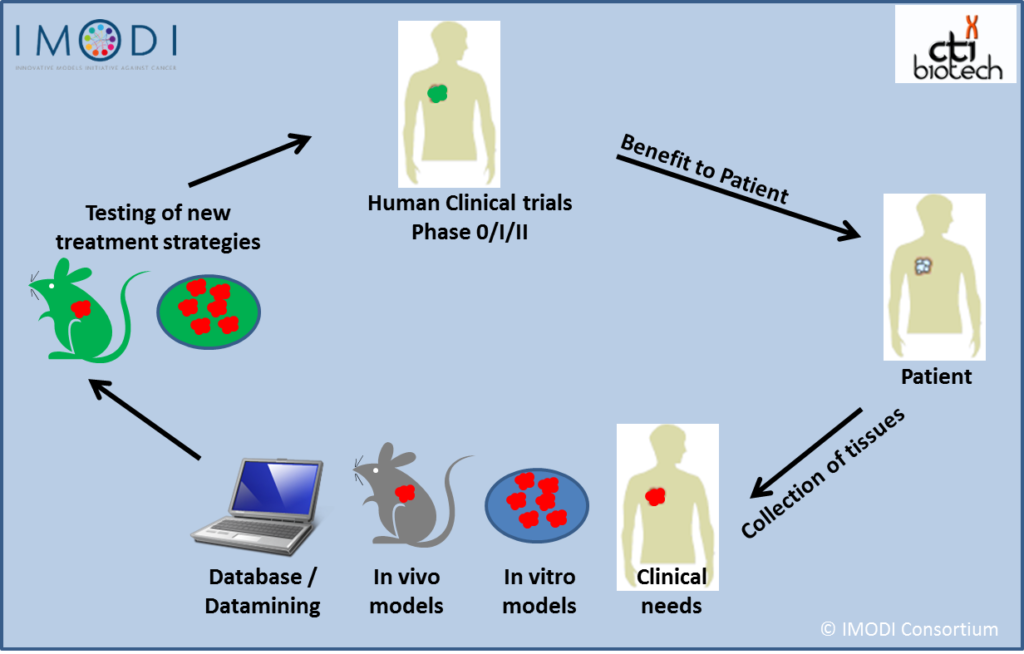
In fact, CTI has found a way to collect different tumors, transport them to the lab and use them to develop patient-derived cell models or cell-based assays for cancer research. For its research and development needs, the company has also set up a scientific biobank of cells and tissues which are the basic components of the cell-based assays developed by the company. It also hosts the scientific biobank of the IMODI initiative–a French consortium to develop new experimental models of cancer–which preserves and archives all tissue and cell samples developed by the consortium partners during the project. The company’s CTI Biosourcing unit is responsible for the National Biobank for cancer and the International Biobank for clients and partners, both with top-quality human cells, tissues and biological samples.
According to Forraz, the National Biobank has collected more than 2000 tumors from patients in collaboration with 11 major cancer hospitals in the country. IMODI was an initiative of former French president Francois Hollande to create the first national network dedicated to personalized medicine in oncology and initially received an investment of 13.4 million Euros under the Futures Program.
“All the samples are qualified. And although a lot of the samples are annonymized, we get the clinical data of the patient as well, and follow their development over a seven-year period to understand what therapy works. The focus of IMODI was to unite the public and private sectors, make useful tools and integrate platforms for cancer research, technical, in vitro, in vivo, as well as record all the clinical data associated with it. We know that if anyone, anywhere in the world needs tools for cancer resarch they can contact each platform, which offers different solutions for the same tumours.”
Located at Meyzieu, near Lyon, the National Biobank is available to all the project partners. Forraz suggests that Lyon is a great hub for cancer research. In a region with five million inhabitants, Lyon encompasses over 2 million. This population, exciting business center, and relevant adult and childhood cancer research network makes it an ideal place for CTI’s future.
And about the future, Forraz says they are increasing their 3D bioprinting capacity to make more microtumor models for their customers. They would also like to expand their company by launching production platforms in each continent so they can be closer to their clients.
With more than 60 customers from the pharmacy sector, biotech companies, academic centers, and hospitals, it is probably a good idea to expand. Close to 75% of the company’s turnover is from abroad, 35% comes from Asia (particularly Japan), other European countries and North America.
Up until now, CTI is the only company using 3D bioprinting for toxicology platforms and generating 3D microtumors in France. Having access to cancer cells is elemental for their work to thrive and ideal to create predictive cell models. The company, led by Forraz’s extensive experience with stem cells, regenerative medicine, and cell manipulation, is quickly building on its expertise in cell culture and tissue engineering in 3D combining cells, biomaterials, growth factors and bioreactors for the development of specialized cell-based assays. Moreover, the drive behind their oncology-related enterprise could help produce tomorrow’s drugs to treat cancer and other diseases.
[Images: CTIBiotech and IMODI]Subscribe to Our Email Newsletter
Stay up-to-date on all the latest news from the 3D printing industry and receive information and offers from third party vendors.
You May Also Like
3D Printed Heat Spreader Could Improve Efficiency of Electronics
The low-hanging fruit for decarbonization has long been improving the efficiency of existing systems, hence the justification for LED lights and ENERGY STAR certified appliances. While such minor moves are...
3D Printing News Unpeeled: Marine Gearboxes, 3D Printed Motors and $1.7 Million in Seed Funding
UK based Equipmake just released their Ampere-220 e-axle system. The system, which is meant for high performance electric cars, was similar to one released on the Ariel HIPERCAR. It has...
CEAD Unveils 36-Meter-Long 3D Printer for Abu Dhabi’s Al Seer Marine
CEAD, a Dutch original equipment manufacturer dedicated to large-format 3D printers, has unveiled what it claims to be the world’s largest robotic arm-based 3D printer. At 36 meters long and...
3D Printed Biocomposites Could Help Reduce Marine Plastic Pollution
Concerns about the impact of plastic litter and microplastics in the oceans are at the forefront of environmental study. For decades, the marine environment has suffered from the degradation of...