A pharmaceutical test can be referred to as a clinical trial or a rigorously controlled test of a new drug or a new invasive medical device on human subjects. In the United States, it is conducted under the direction of the FDA before being made available for general clinical use. With the testing of various drugs it is important to understand their efficacy. The FDA approves drugs for the public use after said tests have gained FDA clearance. It is such a pivotal moment within the development of any drug and it costs a good amount of money to go through FDA clinical trials. Most of these trials typically involved the testing of a drug on an animal as well. Today we will analyze bioprinting and this particular sector of the healthcare industry and how it may change what is possible in years to come.
The field of pharmaceuticals focuses on the following: drug discovery as well as drug development. When one discovers a novel usage of a chemical in terms of a drug, it must then be tested thoroughly a number of times in order to have validity as a commonplace treatment to a specific pathology or ailment. This also allows for one to see how a drug may be developed in lieu of complications that arise when testing a drug. A lot of these tests can be very expensive when done. A way to reduce cost of said tests could be bioprinting. Currently, the technology is not at a scale where one can mass produce tissues or organs for the use of clinical trials on a large scale quantity. With time though, this could be a reality and it can help save time and materials for pharmaceutical companies. Bioprinting also allows pharmaceutical companies to have models of human organs that may provide more accurate test results than lets say a pig organ genetically.
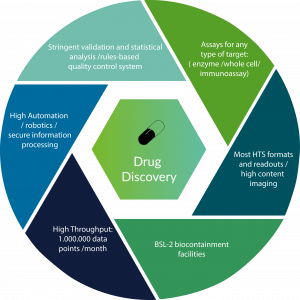 High Throughput Screening and Pharmaceuticals
High Throughput Screening and Pharmaceuticals
We briefly have talked about animal trials already, but let us take a closer look at animal trials. Some animal tests take months or years to conduct and analyze (e.g., 4-5 years, in the case of rodent cancer studies), at a cost of a lot of dollars per substance examined (typically $2 to $4 million per two-species lifetime of a cancer study). The inefficiency and exorbitant costs associated with animal testing makes it impossible for regulators to adequately evaluate the potential effects of the more than 100,000 chemicals currently in commerce worldwide, let alone study the effects of a myriad combinations of chemicals to which humans and wildlife are exposed, at low doses, every day throughout life. One may even look into the ethics behind an animal test overall, and they could argue that bioprinting methods can be a solution to solve ethical problems of using animals detrimentally, but in a manner to serve humans. With bioprinted organs and tissue used for pharmaceutical testing animal ethics may be detracted from pharmaceutical testing (even though ethics is still very apparent if we want to analyze stem cell use within bioprinting, but that is not the topic of discussion).
Bioprinting can truly be beneficial to pharmaceutical testing if high throughput screening is also integrated. High throughput screening is a method to automate and reduce the costs of drug testing. As mentioned previously in our bioprinting series, high-throughput screening (HTS) is a method for scientific experimentation especially used in drug discovery and relevant to the fields of biology and chemistry. Using robotics, data processing/control software, liquid handling devices, and sensitive detectors, high-throughput screening allows a researcher to quickly conduct millions of chemical, genetic, or pharmacological tests. With the combination of high-throughput screening and bioprinting, automation of pharmaceutical testing will cut down the time needed to conduct these type of tests, which also leads itself to better use of time and more money to be made for pharmaceutical companies large and small.
Overall there lies large potential with bioprinting and pharmaceutical testing. It still is far from a strong reality due to the following factors:
- The healthcare industry does not innovate or change methods quickly due to the standards being used.
- A lack of FDA clearance for bioprinting as a whole holds back development
- Not a lot of work done in terms of perfecting these technologies when applied to a large scale pharmaceutical test being done
Subscribe to Our Email Newsletter
Stay up-to-date on all the latest news from the 3D printing industry and receive information and offers from third party vendors.
You May Also Like
Air Force Awards Fortius Metals $1.25M to Qualify 3D Printing Wire for Hypersonic Applications
AFWERX, part of the US Air Force Research Laboratory (AFRL), awarded a Direct-to-Phase II Small Business Innovation Research (SBIR) contract worth $1.25 million to Colorado’s Fortius Metals, to accelerate qualification...
US Air Force Awards JuggerBot $4M for Large-format Hybrid 3D Printing
Large-format 3D printer manufacturer JuggerBot has received a $4 million grant to develop a large format 3D printer, courtesy of the Under Secretary of Defense, Research and Engineering Manufacturing Technology...
Where Have All AM’s Unicorns Gone?
In the rapidly evolving world of 3D printing, startups valued at over a billion dollars, known as unicorns, once seemed as fantastical as the mythical creatures themselves. While a few...
How My Childhood Fascination with Planes Led to Investing in 3D Printing
My fascination with aerospace started young, and I started studying planes–identifying them in the sky and learning everything I could about how they work. Fast forward to my first week...


 Animal Testing
Animal Testing




























