AMS 2019 Day 3: Keynote Speaker Lars Neumann from TRUMPF Discusses 3D Printed Medical Devices
At last week’s second annual Additive Manufacturing Strategies (AMS) summit, held in Boston and co-hosted by 3DPrint.com and SmarTech Markets Publishing, there were several firsts, including an exhibition floor, a startup showdown, dedicated workshops, and two separate tracks for medical and dental 3D printing. During the official opening of AMS 2019, “The Future of 3D Printing in Medicine and Dentistry,” Lawrence Gasman, the President of SmarTech Markets Publishing, said that while last year’s event had participants from roughly 11 countries, this year 24 countries, along with 27 US states, were represented.
The first keynote speaker at AMS 2019 was Dr. Ali Tinazli, the Head of Healthcare and Life Sciences Strategy for HP; he discussed the democratization of medicine and the implications of this. On the final day of the summit, Lars Neumann from German machine tool supplier TRUMPF took the stage for the final keynote presentation, titled “Integrating Additive Manufacturing Into Medical Device Production” and centering around 3D printed instruments and implants.
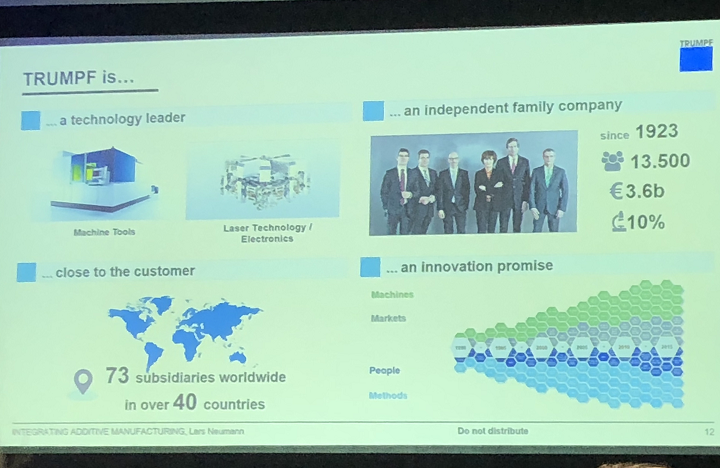
 Neumann, who works at the company’s south German location, explained that TRUMPF is a family business, and that after 90 years in the manufacturing business, it has “quite a bit of a track record” in the medical field, noting examples like using lasers to cut stents.
Neumann, who works at the company’s south German location, explained that TRUMPF is a family business, and that after 90 years in the manufacturing business, it has “quite a bit of a track record” in the medical field, noting examples like using lasers to cut stents.
“If there are any doctors here, typically I’m not talking to you…my presentation today is the production of these devices,” Neumann stated at the beginning, explained that he was mostly talking to the medical device manufacturers.
Neumann noted that in the previous days at the summit, attendees had seen and heard lots of numbers, and said that he was going to be “looking at growth, more than the actual numbers.”
Speaking of those numbers, he mentioned that growth rates for 3D printed medical devices were around 10-15%, which is “quite a significant growth year on year.” But when it comes to fusion devices, Neumann said that people in the industry believe that additive manufacturing will be used 100% in the future.
Some of the main things Neumann said we need to keep discussing to allow serial additive manufacturing to become economically viable for more implants and devices include system and process capability, cost per part, and quality assurance, as “driving up quality lowers cost.”
But how can we assure quality when it comes to 3D printing? Neumann said lots of input, such as CAD data, are necessary when attempting to fabricate a medical device that fulfills all of its defined specifications, since the regulations and standards (like ASTM and ISO) aren’t complete yet. While the lengthy old guard of quality assurance centered around manually maintaining the quality of inputs, like powder, during 3D printing and post-processing and then again checking the completed product, now that imaging equipment and sensors are being added to help ensure quality during the build, we can ideally intervene, if necessary, during the actual 3D printing process.
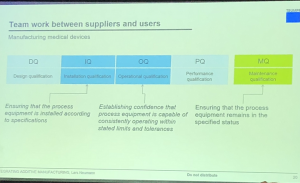 It’s equally as important to lower the cost per part. In manufacturing environments, such as factory floors, ideally the 3D printers should be working on builds around the clock, instead of having to take time for set-up and cleaning. Neumann said that to help ensure this notion, laser off times need to be reduced, and that all other processes, such as post-processing, should be moved to different locations so that the printers can just keep doing what they do.
It’s equally as important to lower the cost per part. In manufacturing environments, such as factory floors, ideally the 3D printers should be working on builds around the clock, instead of having to take time for set-up and cleaning. Neumann said that to help ensure this notion, laser off times need to be reduced, and that all other processes, such as post-processing, should be moved to different locations so that the printers can just keep doing what they do.
In terms of system and process capability, Neumann asked the room what the industry could be doing better to arrive at not only different implants, but also more of them. His personal impression is that, since the additive manufacturing field is developing so quickly, process chain integration is one of the main topics at the moment, along with software, and that machine technology will need to be pushed again a few years down the road.
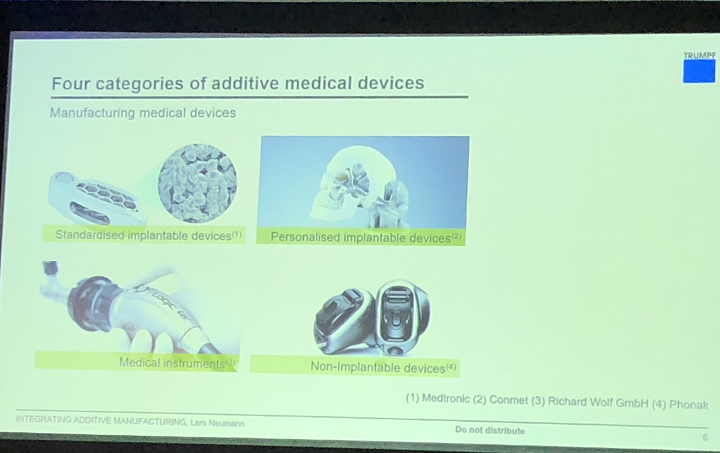
Neumann stated that in terms of additive manufacturing, the main medical device categories are:
- standardized implants
- personalized implants
- medical instruments
- non-implantable devices
He also noted what he called “three key advantages” for 3D printing in the medical field: mass personalization, which provides new treatment options; using porous structures to improve osseointegration; and cost-effective manufacturing, such as low- to mid-volume, less expensive materials, and the ability to create complex shapes. Neumann said that this last point is “slowly coming into focus,” because when it comes to medical 3D printing, hundreds of thousands of parts are not always needed, which can definitely help keep costs down.
Because of increased interest by medical device manufacturers to use 3D printing, Neumann believes that instrumentation as an application will definitely grow, and mentioned that about 100 3D printed medical devices are already FDA-approved.
Switching the focus to the metals used to 3D print many of these instruments and devices, Neumann said that while many people have been excited about titanium in recent years, new materials like cobalt chrome and stainless steel are the talk of the town at this point in time. Finally, when finishing up, Neumann said that aerospace companies are the only ones that possess guidelines to follow when installing metal 3D printers, and that it would be helpful if this would eventually spread to other sectors as well, such as the medical field.
“I hope someday this norm is valid for all industries equally,” Neumann stated.
Some of the questions asked at the end of Neumann’s keynote were quite interesting. One person approached the mic and asked his opinion on the currently available simulation tools, and Neumann said that the software is interesting and seeing a lot of investment at the moment, as many companies, such as OEMs, that use 3D printing are running simulations ahead of nearly every component they’re manufacturing in order to predict defects early on. But, he also noted that the data coming from these simulation solutions has yet to be validated.
Another attendee mentioned again the demand for new, exotic materials in medical instrument 3D printing, and asked Neumann for any specific examples. While it may not sound exotic, he said that stainless steel is one material that many manufacturers can use without having to change the production or post-processing methods, meaning that re-certification won’t be required, so lead times will likely decrease.
Plans have already been laid in motion for the third annual Additive Manufacturing Strategies summit, which will be held from January 29-30, 2020 and will include a metal 3D printing track. To keep up to date on registration information and everything else for AMS 2020, sign up for our newsletter here.
Discuss this news and other 3D printing topics at 3DPrintBoard.com or share your thoughts in the comments below.
[Images: Sarah Saunders]
Subscribe to Our Email Newsletter
Stay up-to-date on all the latest news from the 3D printing industry and receive information and offers from third party vendors.
Print Services
Upload your 3D Models and get them printed quickly and efficiently.
You May Also Like
Bambu Lab A1 Used to Directly 3D Print Copper Electroplated Parts
Maker Dzingof was doing tonnes of electroplating of desktop and other 3D prints years ago with his Metalizzr project. I’ve been playing, a lot less successfully, with electroplated 3D prints...
3D Printing News Briefs, October 1, 2025: Bambu Lab Store, Shape-Morphing Materials, & More
In today’s 3D Printing News Briefs, Bambu Lab has opened its first retail store, and Meltio is inaugurating its first international additive manufacturing reference site. A consortium has launched a...
3D Printing News Briefs, August 13, 2025: Public Utility, Rocket Engine, Brains, & More
In today’s 3D Printing News Briefs, AML3D’s large-scale metal 3D printing system is now online at the largest public utility in the U.S. Moving on, Bright Laser Technologies is 3D...
Soft Robotics is Finally Coming Together: Northwestern Researchers Make Strides Towards a Better Actuator
Soft robotics for a long time was a lot of nice videos, papers, and very little in the way of useful technology. It was kind of an engineering Esperanto: a...